Abbreviation list
CAD: coronary artery disease
CV: cardiovascular
CVD: cardiovascular disease
HRT: hormonal replacement therapy
PCOS: polycystic ovary syndrome
NSAID: non-steroidal anti-inflammatory drugs
SCAD: spontaneous coronary artery dissection
Take-home messages
- Integrating cardiovascular prevention into obstetric and gynaecological care represents a promising opportunity for early intervention.
- Women require different approaches to CV care according to their stages of life.
- Prevention strategies include lifestyle interventions and gender-specific cardiovascular pharmacology.
Patient-oriented messages
- Effective cardiovascular prevention in women requires early risk identification with attention to female-specific risk factors and the implementation of guideline-directed lifestyle and pharmacological interventions.
- Compliance with therapy can be improved by reducing doses, and staggered drug administrations should be considered.
Impact on practice
- Aggressive and early control of cardiovascular risk factors, focusing particularly on smoking, diabetes, and obesity
- Integrated gynaecological and cardiological care for women with gynaecological conditions like endometriosis, polycystic ovary syndrome, or complicated pregnancies, which confer high cardiovascular and metabolic risk.
- Counselling and cardiology evaluation before or at the early phase of pregnancy should become common practice.
- Multidisciplinary management is the key to complex scenarios that are frequent with females.
- Providing women with dedicated ambulatory care for primary prevention, with the involvement and interconnection of several professional figures, may hopefully reduce the gender-related cardiovascular risk burden.
- Modes of conception should be reported in health records.
Introduction
An overview on comorbidities that specifically increase women’s cardiovascular (CV) risk was presented in Part 1 and shows how many conditions are interconnected and may carry a hidden yet high or very high CV risk. Clinical experience specifically dealing with these comorbidities in women has led me to employ a holistic and comprehensive approach in managing CV disease (CVD) prevention in my patients.
Gender symptoms and clinical presentation
The knowledge that the clinical presentation of CVD in women may include atypical symptoms is of paramount importance to avoid overlooking serious scenarios. Chest pain is the most common clinical presentation of coronary artery disease (CAD). However, up to a quarter of women report atypical symptoms such as jaw pain, shortness of breath, palpitations, dizziness, and fatigue. Electrocardiogram (EKG) alterations are less frequent and less marked in women with ST-segment elevation myocardial infarction (STEMI), and a pathological EKG does not always predict CAD in women. Also, stress tests may have lower sensitivity and specificity in women compared with men. Women should be educated to recognise and report symptoms, and doctors should proceed to further investigations when in doubt.
Figure 1. Life course model for cardiovascular primary prevention in women.
Abbreviations:
ART: assisted reproduction technologies; BP: blood pressure; CAC: coronary artery calcium score; CV: cardiovascular; RF: risk factors
Gender-specific life stages
Childbearing age
Oestrogens have anti-inflammatory, antiplatelet, and antioxidant actions, decrease procoagulant factors, and promote vasodilatation by increasing prostacyclin and nitric oxide, thus exerting a cardioprotective action in fertile women. Oestrogen protection typically delays the onset of CVD in women by 10 years. However, while age-adjusted CVD mortality has declined over recent decades, there are some signals of increasing cardiovascular mortality in younger women. There are several possible reasons for this occurrence.
Premature ovarian insufficiency and hypoestrogenism are increasing in young women. Stress-related chronic inflammation, pollution, sedentary behaviour, eating disorders, and malnutrition are all contributors. Although pill dosages are lower and the preservation of the oestrogen/progesterone equilibrium has improved, contraception is starting at a younger age than in the past. This often occurs without taking into account the patient’s risk profile. The only common recommendation for an adolescent starting birth control pills is to exclude those with a personal history of thrombosis or a family history of ovarian/breast cancer. However, no attention is paid to a family history of CV disease and/or the individual CV risk profile (i.e., lipids, smoking), nor is CV follow-up recommended. Conversely, oral contraception might cause secondary hypertension and worsen the lipid profile. Early detection is crucial as pill withdrawal may normalise these conditions.
Besides pure contraception, other conditions that may require early hormone therapy in young women are polycystic ovary syndrome (PCOS) and endometriosis. Each condition may be present in up to 13% of reproductive-aged women, and both are adjunctive, non-traditional cardiometabolic risk factors [1]. Moreover, the presence of at least one of the common gynaecological disorders, such as PCOS, endometriosis, or heavy or irregular periods, confers a very high risk for developing cardiovascular and cerebrovascular disease as compared to women without these conditions [2]. Plausible biological pathways include systemic inflammation, systemic vascular dysfunction, and hormonal imbalances. There may also be an overlap between gynaecological and cardiovascular risk factors. Indeed, PCOS is often associated with metabolic syndrome and diabetes, often independent of body mass index.
The abuse of painkillers, including non-steroidal anti-inflammatory drugs (NSAIDs), in the treatment of these conditions may be another detrimental factor in CV health, confirming how impactful gynaecological conditions are on quality of life. In addition, stress, hormone therapy, hypothyroidism, fibromuscular dysplasia, and inflammatory or connective tissue disorders are all risk factors for spontaneous coronary artery dissection (SCAD). Despite all this evidence, too often women continue to be completely excluded from prevention strategies. This may be even more pronounced in low- and middle-income countries.
The complete absence of dedicated paths for young women needing potential cardiology attention creates confusion and further diagnostic delays, while it is in the childbearing years that the first cardiological evaluation should take place.
Pregnancy
The European Society of Cardiology Guidelines for the management of cardiovascular disease and pregnancy have recently been published [3]. As maternal age increases, the traditional risk factors for coronary artery disease are more pronounced and important, with pregnancy known to increase the risk of acute myocardial infarction 3- to 4-fold compared with age-matched non-pregnant women.
Pre-pregnancy counselling, including a cardiological evaluation, should be required. However, a study involving women with hypertrophic cardiomyopathy found that only 8% underwent an echocardiogram before pregnancy [4].
Pregnancy complications, including pre-eclampsia, eclampsia, gestational hypertension, gestational diabetes, pre-term delivery, placental abruption, and stillbirth are associated with increased CV risk. In particular, pre-eclampsia almost triples the risk of CVD [5], and gestational diabetes mellitus is associated with a 2-fold increase in CV risk, with the highest risk in the first postpartum decade, and is associated with the development of diabetes and hypertension. A history of spontaneous pre-term delivery is also associated with increased CV risk [6].
Most cases of acute coronary syndrome in pregnancy are due to SCAD, and this most commonly happens during the third trimester or postpartum. Though rare, this condition should be promptly recognised as it carries about a 7% risk of maternal and foetal mortality [7]. It is also important not to mistake postpartum cardiomyopathy for postpartum depression, so as not to slow down access to appropriate care.
Women with complicated pregnancies should be promptly taken in charge by a cardiologist and followed throughout their lives as recommended by current guidelines with regular cardiovascular and metabolic check-ups as well as lifestyle and/or pharmacological interventions [3]. Unfortunately, these women are often forgotten after delivery, and follow-up cardiovascular check-ups are not performed.
Menopause
Oestrogen withdrawal after menopause has many negative effects, including endothelial dysfunction, increased sympathetic nerve activity, higher insulin resistance, and alteration in body fat distribution. Indeed, myocardial infarction and stroke rates increase after menopause.
The incidence of premature menopause (<40-45 years) is increasing, and this reduces the timeline of hormonal protection, increasing CV risk by 50%. As perimenopause can last longer for these women, the extended duration of hormonal fluctuations during this period may add additional cardiovascular risk. Vasomotor symptoms might also indicate a higher CV risk [8] and create a vicious circle of insomnia, irritability, and depression. Notably, hormone replacement therapy (HRT) has not been proven to have cardioprotective effects.
Gender and cancer
Breast and ovarian cancer also create a high cardiovascular risk, because of overlapping risk factors and the detrimental prothrombotic effect of treatment. In particular, chest radiotherapy increases the risk of developing CAD. Hysterectomy with or without oophorectomy is associated with an increased risk of cardiovascular events, partly because it may accelerate the onset of menopause or compromise blood supply to the ovaries.
Although some of these variables have been mentioned in recent guidelines [3-9], these risk factors have yet to be implemented into cardiovascular risk calculators. In these women, correction of modifiable risk factors should be complete, including psychological support when needed.
Cardiovascular risk assessment in women
Conventional risk calculators often underestimate risk in women, especially younger women and those with sex-specific major risk enhancers. Conversely, accurate cardiovascular risk assessment in women should incorporate reproductive and pregnancy history, comorbidities, and psychosocial conditions. In addition, coronary artery calcium scoring, biomarkers (i.e., high-sensitivity C-reactive protein >2 mg/L, lipoprotein(a) >50 mg/dL), and subclinical atherosclerosis may improve risk stratification [9].
Prevention strategies
Lifestyle interventions
Lifestyle modifications are the cornerstone of CVD prevention. At all ages, the main strategies for CVD prevention in women are healthy habits, physical activity, and management of negative emotional stress.
A heart-healthy diet rich in fruits, vegetables, cereals, lean proteins, and unsaturated fats while limiting salt (<2-3 mg/day), saturated fat, and added sugar is strongly recommended.
Smoking cessation is critical, and targeted interventions (such as behavioural counselling and pharmacotherapy) may be particularly beneficial for women who often face unique barriers to quitting, including the fear of gaining weight.
Physical activity improves blood pressure, lipid profiles, insulin sensitivity, and overall cardiovascular and mental health. It should be daily, regular, and progressive without prolonged stops followed by peaks, which can endorse SCAD. Mindfulness and yoga may help modulate the autonomic nervous system response since sympathetic nervous activity increases with age and is even more pronounced in females. Indeed, distress may trigger SCAD and Takotsubo [10], both of which are more frequent in women. Notably, women may achieve greater cardiovascular benefit than men with similar or even lower amounts of physical activity [11].
Weight management through sustainable dietary and physical activity strategies rather than short-term strategies is essential. Adequate sleep and stress management (including the ability to say “no” and refuse to be overwhelmed both at work and in private life) are of paramount importance. Educational strategies, increased awareness of individual CV risk, and the development of communication skills to adequately report symptoms, which are often hidden, behind sociocultural barriers may truly enhance female CV prevention.
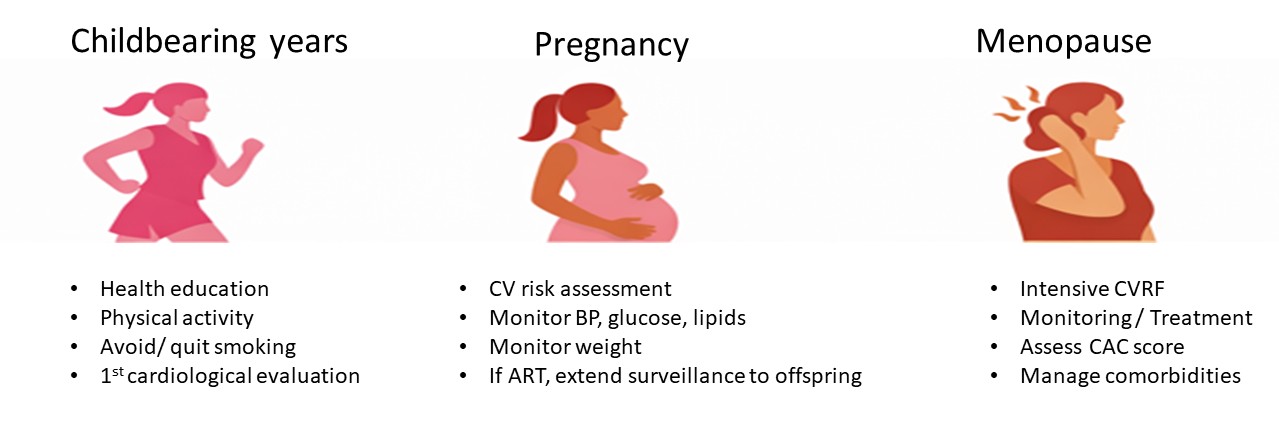
A useful framework for CVD prevention according to the phases of a woman’s life is depicted in Figure 2. For those with gynaecological problems and complicated pregnancies, adding a CV evaluation is recommended.
Gender-specific cardiovascular pharmacology
Pharmacological therapy in women may be challenging and needs dedicated surveillance. No difference between women and men in the efficacy of antihypertensive drugs or statins in reducing CVD has been shown, and yet women remain less likely to receive statins than men. Chronic undertreatment in women is mostly due to therapeutic inertia, but also to higher therapy discontinuation by women. This is due to a reduced perception of risk on one hand and a high incidence of side effects on the other.
Women have lower body weight, higher fat/water balance, lower clearance, and different hormonal composition than men, which can ultimately affect pharmacokinetics and pharmacodynamics. Dose reduction may be beneficial. Also, drug-drug interactions are more frequent in women. In addition, as for other substances, allergies to drugs are more frequent in women, and therapeutic alternatives should be identified.
The use of aspirin for primary prevention in young women is not routinely recommended and should be individualised, balancing potential cardiovascular benefits against bleeding risk [12]. For example, the use of <100 mg of acetylic acid may have less impact on anaemia and metrorrhagia, preserving efficacy.
Refracted doses of beta blockers during the day may be better tolerated. A shift to another type of beta blocker is to be considered in case of alopecia and weight gain, which can occur and can be highly painful for women. Potassium-sparing diuretics, specifically spironolactone, may be considered as an early intervention in hypertensive women with PCOS or androgenetic alopecia due to its dual action as a diuretic and an anti-androgen.Low doses or alternate-day statins may be better tolerated; nutraceuticals, now a class III drug [4], have not been selectively studied in women. Personalised therapies guided by individual risk profiles with careful drug selection may be helpful for women to reduce side effects and maintain therapeutic compliance.
Also, chronic abuse of NSAIDs and other painkillers should be considered, as well as the selection of less cardiotoxic drugs, and limiting doses and duration of administration. Current evidence does not evenly support routine HRT for CVD prevention and suggests potential harm when initiated late after menopause.
Pre-primary prevention
Global maternal physical and mental health, lifestyle, and drugs during pregnancy impact maternal prognosis as well as that of the child. Specific data on the CV outcome of men and women born to complicated pregnancies are missing. However, a study showed that adolescents born to pregnancies conducted with assisted reproduction technologies had an incidence of hypertension 6-fold higher than in the control group [13]. Cryopreservation is increasing, both for voluntarily delayed pregnancies or after immunosuppressive or antineoplastic therapies responsible for infertility; this opens the possibility for future scenarios for CVD prevention, further underlining the need to record the mode of conception, which today is often, for various reasons, not available.
Conclusions
Biological differences, structural and social inequities, and still-persistent socioeconomic disadvantages significantly influence cardiovascular risk and outcomes in women. Cardiovascular disease prevention in women is complex and covers a wider range of clinical scenarios and comorbidities. This complexity requires dedicated and more comprehensive paths with the involvement of a wide range of medical professionals. Cardiovascular prevention should start during the childbearing years and continue throughout a woman’s life. Tailored and age-specific prevention programmes are needed, and a broader inclusion of women in cardiovascular clinical trials with sex-specific reporting of outcomes remains critical.