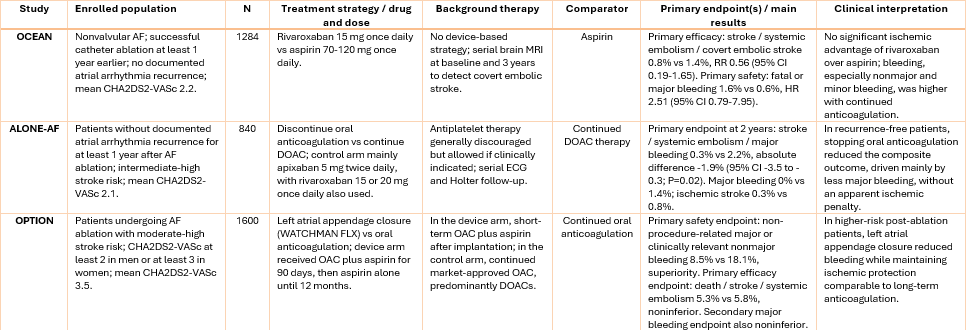
OCEAN was a randomised, open-label, blinded-outcome trial evaluating whether continued anticoagulation with rivaroxaban was superior to aspirin after apparently successful catheter ablation for atrial fibrillation in patients at risk of stroke.
A total of 1,284 patients were enrolled at least 1 year after ablation, without documented atrial tachyarrhythmia recurrence, and randomised to rivaroxaban 15 mg once daily or aspirin 70–120 mg once daily. Serial brain MRI was used to detect covert embolic stroke. The enrolled population had relatively low thromboembolic risk, with a mean CHA₂DS₂-VASc score of 2.2 and only 31.9% having a score ≥3. At 3 years, the primary efficacy endpoint – stroke, systemic embolism, or new covert embolic stroke – occurred in 0.8% of patients receiving rivaroxaban and 1.4% receiving aspirin, without a significant ischaemic benefit of continued DOAC therapy. Fatal or major bleeding occurred in 1.6% versus 0.6%, respectively, while minor bleeding was clearly increased with rivaroxaban.
The trial was stopped early for futility and excess nonmajor bleeding. Overall, OCEAN suggests that, after durable ablation success and in the absence of recurrent atrial arrhythmia, selected patients with low-to-intermediate CHA₂DS₂-VASc profiles have a very low residual ischaemic event rate, limiting the absolute benefit of prolonged anticoagulation. Together with ALONE-AF, these findings support cautious antithrombotic de-escalation in carefully selected low-risk patients. In contrast, OPTION extends this concept to higher-risk patients, suggesting that left atrial appendage closure may be a more attractive alternative than indefinite DOAC continuation when simple anticoagulation withdrawal is not appropriate.