OCEANIC-STROKE was a randomised, placebo-controlled, phase 3 trial enrolling patients with recent non-cardioembolic ischaemic stroke or high-risk TIA treated with background antiplatelet therapy (n=12,327).
Participants were assigned to asundexian or placebo on top of standard care. Asundexian reduced the incidence of recurrent ischemic stroke (6.2% vs. 8.4%), corresponding to a 26% lower hazard (HR 0.74, 95% CI, 0.65 to 0.84). Notably, ISTH major bleeding rates were similar between groups (1.9% vs. 1.7%; HR 1.10, 95% CI 0.85–1.44). These findings suggest that targeting factor XIa may provide additional protection against thrombotic events in this population, while maintaining a favorable bleeding profile.
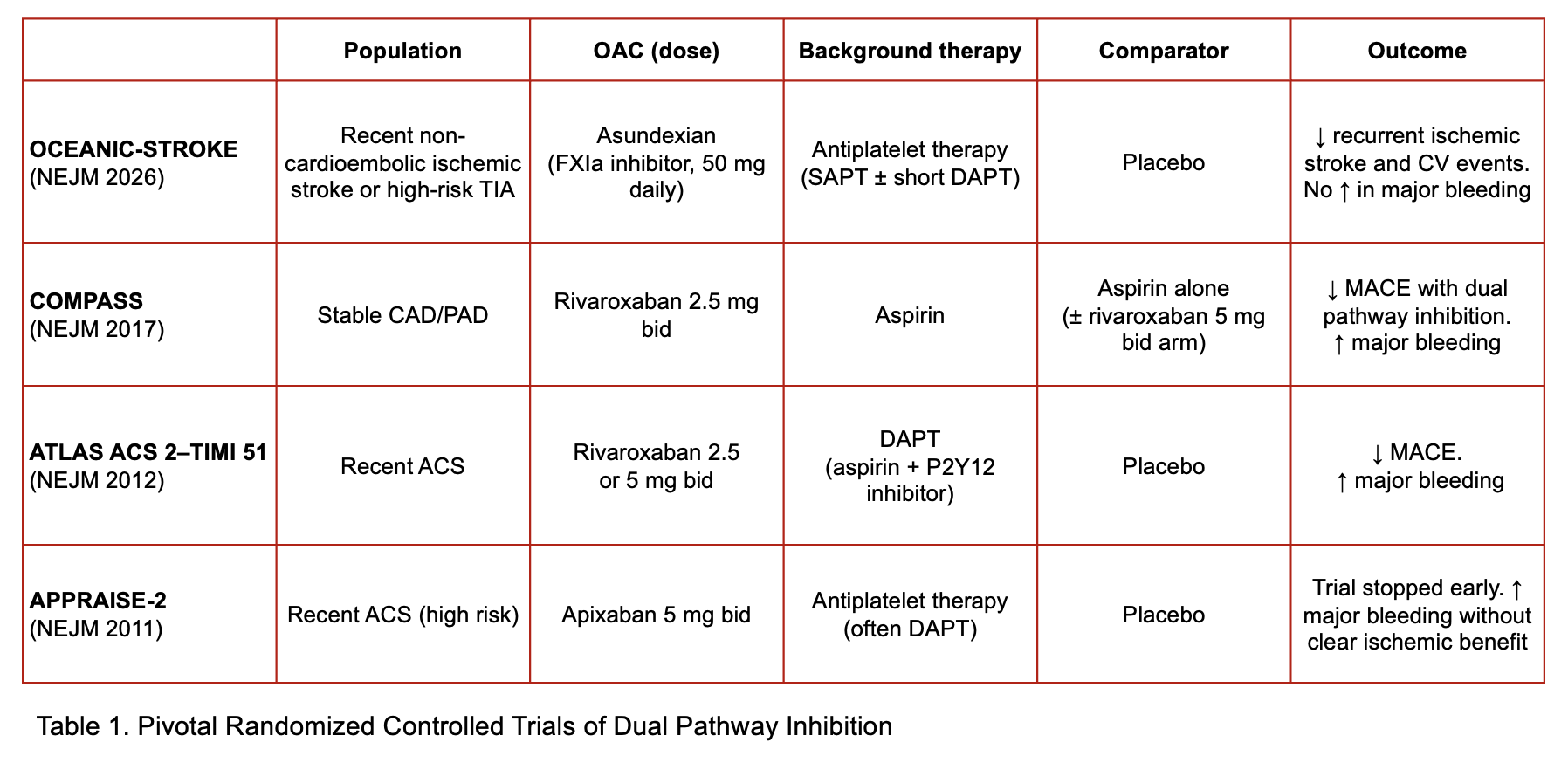
The concept underlying this approach is dual pathway inhibition, namely the combination of antiplatelet therapy with modulation of the coagulation cascade. This strategy has previously been evaluated in the COMPASS randomised controlled trial, where low-dose rivaroxaban added to aspirin reduced major cardiovascular events in patients with stable atherosclerotic vascular disease, albeit at the cost of increased bleeding risk. Notably, COMPASS relied on a factor Xa inhibitor, acting downstream in the coagulation cascade, whereas factor XIa inhibitors target an upstream component of the intrinsic pathway, a difference that may allow partial dissociation between antithrombotic efficacy and bleeding risk. Conversely, the early termination of LIBREXIA-ACS suggests that dual pathway inhibition strategies may not be uniformly effective across all the spectrum of cardiovascular diseases (Table 1).
The ongoing phase 3 LIBREXIA-STROKE randomised controlled trial (NCT05702034), evaluating milvexian versus placebo on top of standard antiplatelet therapy in patients with recent ischemic stroke or TIA, will further clarify the role of this strategy and help determine whether these findings can be generalized to broader stroke populations.