Hypertension after menopause: an unmet preventive imperative
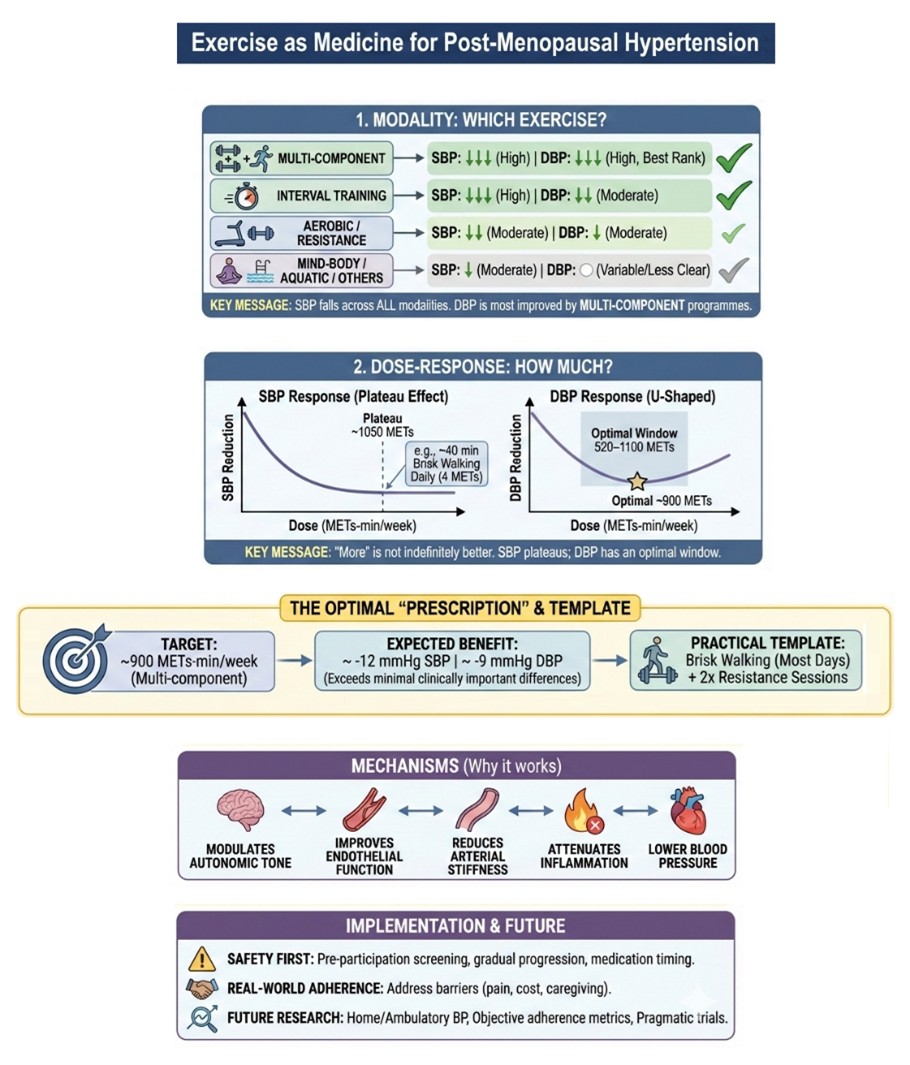
No cardiologist taking care of women can deny that raised blood pressure (BP) often begins its stealthy ascent after menopause, adversely impacting on cardiovascular risk precisely when prevention should be most assertive and attentive.(1) Yet, despite broad endorsement of physical activity, we still lack clear, life-stage–specific guidance on which exercise to choose and how much is enough. In their timely synthesis of the available evidence base recently published in the European Journal of Preventive Cardiology, Zhang and colleagues tackle this practical dilemma by applying a dose–response network meta-analytic framework, moving beyond simple “exercise versus control” comparisons to jointly weigh modality and dose (Figure 1).(2)
Indeed, capitalising on as many as 79 randomised trials including a total of 3,628 post-menopausal women, they standardised training exposure using metabolic equivalents of task per minute (METs-min)/week, enabling fairer contrasts across aerobic, resistance, interval, multi-component, mind–body/stretching, aquatic, and whole-body vibration programs. The key message is quite favorable: systolic blood pressure (SBP) uniformly falls across modalities, with estimates frequently overlapping, suggesting that the key point is exercising, irrespectively of the chosen type of exercise. At the same time, the more the merrier, but with a degree of common sense. Indeed, more work is not necessarily increasingly beneficial, as some plateau effect is plausible.
Which exercise and how much?
Overall, few interventions in preventive cardiology are as universally recommended (and beneficial) as physical activity, yet clinicians still struggle with a deceptively simple question: which exercise, and how much, should we recommend to lower BP?(3) In the work by Zhang and colleagues, diastolic blood pressure (DBP) emerged as the most discriminating endpoint, with multi-component programmes ranking highest, interval training close behind, and several other modalities not being separated clearly from control. Instead, SBP improved across the board of exercise types, but not indefinitely, with the meta-analytic dose-response curve suggesting a clinically meaningful benefit from relatively modest doses, followed by a plateau once weekly volumes approached roughly 1050 METs-min/week. This translates, assuming brisk walking (associated with an average 4 METs of effort), into roughly 40 minutes of brisk walking every day.
Responses in DBP were even more nuanced, with a U-shaped pattern and a optimal window around 520–1100 METs-min/week. Strikingly, a multi-component dose near 900 METs-min/week delivered the most consistent combined reductions (about -12 mmHg in SBP and -9 mmHg in DBP), comfortably exceeding minimal clinically important differences. Importantly, when multi-component training is impractical, aerobic and resistance-based options remain viable, provided dose is respected and progression is gradual. We may tentatively recommend a template—brisk walking on most days plus two resistance sessions, which may help patients and clinicians operationalize this dose. This 900-METs target also appears quite appropriate in terms of contemporary global activity recommendations.
It is useful to reinstate that, while no clinician would dispute that exercise is among the most potent, low-cost tools to protect cardiovascular health in women, prescribing and implementing it with the same rigor as drugs remains uncommon in today’s routine care.(4) The dose–response meta-analysis by Zhang and colleagues strengthens biological plausibility and reinforce the message that exercise is crucial to prevent hypertension (Figure 1): combining aerobic and resistance components can favorably modulate autonomic tone and neurohumoral pathways, improve endothelial function, reduce arterial stiffness, and attenuate inflammation—mechanisms that converge on lower blood pressure.
Safety, screening, and the limits of the evidence
Still, “more” must also be “safe”: pre-participation screening, attention to medication timing, and gradual progression are essential, particularly for previously sedentary hypertensive women, and the sparse reporting of adverse events across trials should temper complacency.(5) Crucially, while this meta-analysis provides convincing evidence in support of the beneficial effects of exercise on BP, it does not directly link these changes to patient-important outcomes (e.g. major adverse events accrued over several years of follow-up). Similarly, safety endpoints were not systematically captured across the included trials, and these limitations evidently limit external validity. Indeed, exercise can be accompanied by transient BP surges during activity, which could theoretically carry safety implications in selected high-risk patients.(6) Such premises further underscore the key value of pre-participation screening, especially before more intense efforts. In addition, it is paramount to adopt careful progression of intensive exercise and thoughtful medication timing, especially in previously sedentary hypertensive women, in the context of limited adverse-event reporting.(5)
Moreover, the real dose is the dose patients can sustain: barriers such as pain, caregiving demands, cost, and access make implementation science as critical as physiology. Future trials should therefore move beyond office readings to home and ambulatory blood pressure, quantify adherence and intensity with objective metrics, and define who benefits most and for how long, and test pragmatic, scalable multi-component programs against single-modality comparators. With better reporting and higher-certainty evidence, guideline writers can translate exercise “ranges” into blood-pressure–targeted prescriptions for post-menopausal women.
Figure 1. Navigating the evidence base on exercise effects on blood pressure (BP) in post-menopausal women. DBP=diastolic blood pressure; MET=Metabolic equivalent of task; min=minutes; SBP=systolic blood pressure.
Acknowledgement: This manuscript was drafted and illustrated with the assistance of artificial intelligence tools, such as ChatGPT (OpenAI, San Francisco, CA, USA), and Gemini (Alphabet, Mountain View, CA, USA), in keeping with established best practices (Biondi-Zoccai G. ChatGPT for Medical Research. Torino: Minerva Medica; 2024). The final content, including all conclusions and opinions, has been thoroughly revised, edited, and approved by the authors. The authors take full responsibility for the integrity and accuracy of the work and retain full credit for all intellectual contributions. Compliance with ethical standards and guidelines for the use of artificial intelligence in research has been ensured.



















