Keywords
cancer, treatment, venous thromboembolism
Abbreviation list
DOAC: direct oral anticoagulants
DVT: deep venous thrombosis
LMWH: low-molecular-weight heparins
GI: gastrointestinal
GU: genitourinary
PE: pulmonary embolism
VTE: venous thromboembolism
Take-home message:
- In patients with cancer-related VTE, anticoagulation should be individualised and based on the characteristics of the cancer and treatment as well as the patient’s characteristics and preferences.
- Incidental VTE should be treated as symptomatic (VTE) because of risk of recurrence.
- Anticoagulant treatment should be continued for as long as the cancer is active.
Introduction
Venous thromboembolism (VTE), including pulmonary embolism (PE) and deep venous thrombosis (DVT), is one of the major causes of morbidity and mortality in cancer patients. We aim to review the current knowledge and recommendations for treatment of cancer-associated VTE
Epidemiology
The risk of VTE is 4-to-7-fold higher in cancer patients, according to the type of cancer (the highest risk is for stomach and pancreatic cancer), as well as the stage of cancer (metastatic >locally advanced>primary site). In addition, VTE is the second cause of death after cancer itself (>60% within the first year following the VTE diagnosis). Cancer patients also have a high risk of VTE recurrence (up to 20% within 12 months after the stop of anticoagulant therapy) with a case fatality rate of 14.8% (95% confidence interval [CI]: 6.6-30.1) [1].
Even the risk of bleeding is increased in cancer patients with a 12 month cumulative incidence rate of major bleeding of 12.4% (95% CI: 6.5-18.2) and a case fatality rate of 8.9% (95% CI: 3.5-21.1) [1]. The risk is highest for colorectal and prostate cancer [2].
Anticoagulant options
Over the last two decades, low-molecular-weight heparin (LMWH) has been the first-line therapy for VTE. The CLOT study showed that LMWH (i.e., dalteparin) was more effective than vitamin K antagonists (VKA) in reducing the risk of VTE recurrence in cancer patients (risk ratio [RR] 0.51, 95% CI: 0.33–0.79] without significant differences in major bleeding events [3]. Several meta-analyses confirmed the superiority of LMWH (including dalteparin, tinzaparin, enoxaparin, nadroparin) over VKA [4-5]. Long-term treatment with LMWH can be problematic due to both the burden of daily injections (leading patients to prematurely stop the anticoagulant therapy) as well as the risk of haematoma at site of injection, osteoporosis, and heparin-induced thrombocytopenia.
The direct oral anticoagulants (DOAC) represent an alternative to LMWH. The three anti-Xa inhibitors: edoxaban, rivaroxaban, and apixaban, have been evaluated for the treatment of cancer-associated VTE (Table 1).
Table 1. Randomised clinical trial with DOAC.
| Clinical trials | Patients |
Primary endpoint |
Secondary endpoint |
|---|---|---|---|
|
HOKUSAI-VTE study [6] |
1046 |
|
|
|
SELECT-D pilot study [7] |
406 |
|
|
|
CARAVAGGIO study [8] |
1,155 |
|
|
CRNMB: clinically relevant non-major bleeding; DOAC: direct oral anticoagulants; HR: hazard ratio; LMWH: low-molecular-weight heparin; MB: major bleeding; VTE: venous thromboembolism
Compared to LMWH (dalteparin), both edoxaban [6] and rivaroxaban [7] showed a non-inferiority for VTE recurrence. However, the incidence of bleeding (major bleeding and clinically relevant non-major bleeding) was significantly higher for both DOACs (Table 1) although primarily confined to patients with gastrointestinal cancer [6, 7]. In the Caravaggio study, apixaban showed non-inferiority compared to dalteparin for VTE recurrence at 6 months (Table 1) [8]. However, and conversely to previous studies, the incidence of major bleeding and clinically relevant non-major bleeding events was similar in both treatment groups [8]. In a further sub-analysis, the incidence of bleeding remained similar in both groups, irrespective of cancer localisation (Table 2) [9]. The highest incidence of bleeding was observed in patients with gastrointestinal (GI) and genitourinary (GU) cancer, primarily if the cancer was not resected [9]. It should be noted that patients with primary or metastatic cerebral cancers and acute leukaemia were excluded from the Caravaggio study.
Table 2. Bleeding events in Caravaggio study according to cancer site [9].
| Gastrointestinal cancer (n=375) |
Genitourinary cancer (n=139) |
|||||
| Major bleeding | 4.8% (9)* | 4.8% (9)* | ‒0.0003 (0.1009- 0.1009) | 6.1% (4)* | 8.2% (6)* | ‒0.0216 (‒0.1866- 0.1450) |
| CRNMB | 10.1% (19) | 3.7%(7) | 0.0636 (‒0.0372- 0.1642) | 7.6% (5) | 11% (8) | ‒0.0338 (‒0.1982- 0.1330) |
| Lung cancer (n=200) | Breast cancer (n=155) | |||||
| Apixaban (105) | Dalteparin (95) | Risk difference (95% CI) | Apixaban (79) | Dalteparin (76) | Risk difference (95% CI) | |
| Major bleeding | 3.8% (4) | 3.2% (3) | 0.0065 (‒0.1319- 0.1447) | 3.8% (3) | 3.9% (3) | ‒0.0010 (‒0.1574- 0.1572) |
| CRNMB | 12.4% (13) | 7.4% (7) | 0.0501 (‒0.0891- 0.1880) | 6.3% (5) | 2.6% (2) | 0.0370 (‒0.1193-0.1949) |
| Gynaecological cancer (n=119) | Haematological cancer (n=85 | |||||
| Apixaban (60) | Dalteparin (59) | Risk difference (95% CI) | Apixaban (33) | Dalteparin (52) | Risk difference (95% CI) | |
| Major bleeding | 3.3% (2) | 3.4% (2) | ‒0.0006 (‒0.1781- 0.1781) | 0 | 0 | |
| CRNMB | 5.0% (3) | 13.6% (8) | ‒0.0856 (‒0.2594- 0.0954) | 9.1% (3) | 3.8% (2) | 0.0524 (‒0.1655- 0.2660) |
Apixaban (188)Dalteparin (187)Risk difference (95% CI)Apixaban (66)Dalteparin (73)Risk difference (95% CI)
*Unresected cancer.
CI: confidence interval, CRNMB: clinically relevant non-major bleeding
How to choose the best anticoagulant for my patient
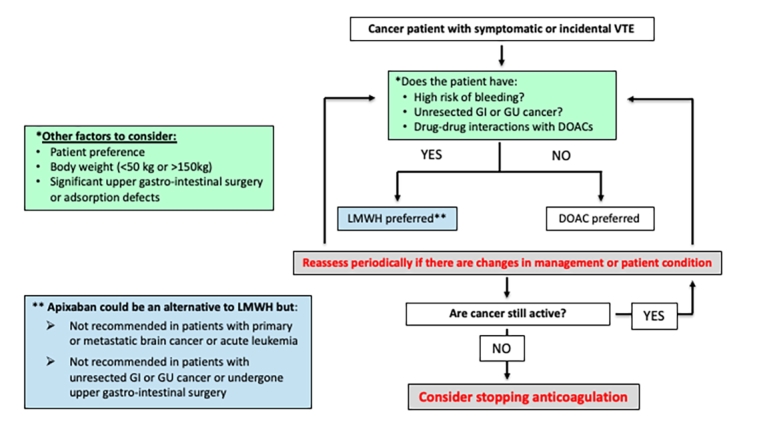
The algorithm in Figure 1 is intended as a guide on how to choose the best anticoagulant according to the cancer’s characteristics and treatment as well as patient’s characteristics and preferences. Several guidelines and consensus documents have been published in the last three years about the treatment of VTE in cancer patients, all with similar recommendations [10-13]. Edoxaban and rivaroxaban should be considered as an alternative to LMWH in patients without gastrointestinal or genitourinary cancer and, in general, for those patients with low risk of bleeding. Caution should be made for any potential drug interaction with anti-cancer therapy.
Figure 1. Algorithm for the treatment of cancer-associated venous thromboembolism.
DOAC: direct oral anticoagulants; GI: gastrointestinal; GU: genitourinary; LMWH: low-molecular-weight heparin; VTE: venous thromboembolism
Apixaban should be considered as an alternative to LMWH for initial and long-term treatment in patients without primary or metastatic brain cancer or acute leukaemia. Caution should be made for any potential drug interaction with anti-cancer therapy and in patients with unresected cancer.
LMWH is preferred over DOACs in cancer patients with unstable clinical situations including low platelet count, nausea and vomiting, risk of expected drug-drug interaction, patients undergoing surgery involving the upper gastrointestinal tract or with unresected GI or GU cancers.
Two elements should always be considered in the choice of an anticoagulant: the patient’s bleeding risk and any drug interactions with cancer therapy. Concerning bleeding risk, several bleeding scores have been evaluated but none have shown sufficient predictive accuracy to be recommended in routine clinical practice. In addition, bleeding scores specifically developed for the cancer population are lacking. In the evaluation of bleeding risk in cancer patients several elements should be considered:
- GI or GU cancer
- Brain cancer
- Recent surgery or life-threatening bleeding
- Concomitant cancer therapy associated with high risk of bleeding (i.e., bevacizumab, agents provoking GI mucosal-toxicity)
- Severe thrombocytopenia (<50,000)
- Renal or hepatic failure
- Concomitant antiplatelet therapy
Concerning drug-drug interactions, apixaban, edoxaban, and rivaroxaban are all substrates for P-glycoprotein (P-gp). Therefore, interactions with strong P-gp inducers or inhibitors may lead to a decrease or an increase in DOAC concentrations, respectively. In addition, apixaban and rivaroxaban are also dependent on cytochrome CYP3A4 for part of their metabolism. So, strong CYP3A4 inducers and inhibitors will also potentially alter their efficacy or safety.
There are different ways to evaluate a drug-drug interaction. First of all, this can be discussed with the local pharmacist or found in product monographs or package inserts of drugs. There are also different databases which are constantly updated, notably the Lexicomp Drug Interaction tool and the drug interactions checker at the University of Liverpool (https://cancer-druginteractions.org/). This last-one is free of charge.
Incidental VTE
Up to 50% of VTE diagnosed in oncology centres are incidentally detected during imaging performed for cancer staging, evaluation of treatment response and routine follow-up. Although VTE diagnosis in unsuspected in these patients, approximately one-half of patients report symptoms of PE (or DVT). However, as these symptoms are non-specific, they are often attributed to cancer or the side effects of treatment.
Recent studies showed that incidental PE (as well as subsegmental PE) carries the same risk of VTE recurrence and mortality as symptomatic PE [14, 15]. Therefore, patients with active cancer and incidental PE should always receive anticoagulant treatment. Before starting treatment, it is recommended to carefully review the images with an experienced radiologist, especially in cases of isolated subsegmental PE. In addition, compression ultrasonography of the lower limbs to detect concomitant incidental DVT is also suggested. In the same way, in patients with incidental iliofemoral DVT on computed tomography of the abdomen and pelvis are recommended to have a a venous ultrasonography of the pelvis and lower limbs.
Duration of anticoagulant treatment
The duration of anticoagulant therapy is typically 3-6 months and usually for as long as the cancer is active. According to clinical trials, cancer is considered active if metastatic or if cancer treatment (either chemotherapy and/or radiotherapy) is ongoing. Conversely, cancer is considered non-active if it has been in remission for more than 6 months.
Two recent studies showed that if anticoagulation is stopped too early, when cancer is still active, the risk of recurrence is significantly higher (up to 6-fold) [16, 17]. A safer alternative to stop anticoagulation could be to reduce the dose. To this end, a large randomised clinical trial, comparing the efficacy and safety of a reduced dose of apixaban (2.5 mg bid) vs a standard dose (5 mg bid) for the prevention of recurrent VTE in cancer patients who have completed at least 6 months of anticoagulant therapy, is currently ongoing [18].
In addition, it is important to periodically assess the risk of recurrence and bleeding as well as the cancer status and patient’s preferences.
Conclusions
Treatment of cancer-associated VTE remains a challenge. In these patients, anticoagulant therapies should be individualised, based on the type and stage of cancer, cancer treatment and potential drug-drug interactions, bleeding risk, and the patient’s preference.
Patients should be treated for three-six months and for as long as the cancer is active.
Incidental VTE should be treated as a symptomatic form because of the risk of recurrence.