Doctor Dimitrios Miltiadou Farmakis
FESC, FHFA
Mr Dimitrios Papadopoulos
Doctor Georgios Papingiotis
Keywords
Cancer, risk factors, thrombosis, venous thromboembolism
Abbreviation list
CAT: cancer-associated thrombosis
DIC: disseminated intravascular coagulation
TMA: thrombotic microangiopathy
VTE: venous thromboembolism
Take-home messages:
- Venous thromboembolism (VTE) is common in cancer carrying a high risk of morbidity and mortality
- Several factors are associated with VTE in cancer including tumour-specific, patient-related and therapy-associated factors.
- Anticoagulation for VTE in active cancer may be challenging and decisions should be individualised based on thrombotic and bleeding risks, drug-drug interactions and patient preferences.
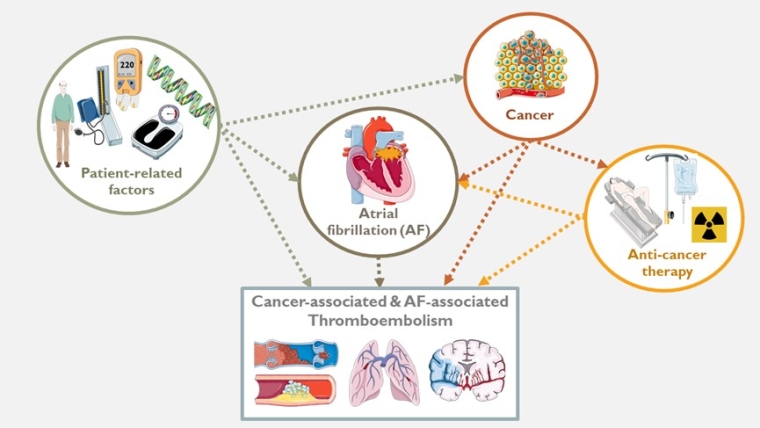
The burden and spectrum of cancer-associated thrombosis
The relationship between thrombosis and cancer was first described in 1865 by Armand Trousseau. Since then, it has been established through research that thrombosis is a common cancer complication [1]. Cancer patients are generally in a prothrombotic state with abnormalities that affect all components of Virchow’s triad (endothelial injury or dysfunction, hypercoagulability, blood stasis or turbulence). Cancer-associated thrombosis (CAT), affecting both the arterial and venous system, is the second-leading cause of death in patients with malignancies, accounting for 9% of deaths in a cohort of 4,466 patients [2]. The spectrum of CAT includes on one hand, the more frequent venous thromboembolism (VTE), which comprises deep vein thrombosis (DVT) and pulmonary embolism (PE), and on the other, the arterial thromboembolism, including coronary syndromes, stroke and peripheral arterial thromboembolism [3]. Additional manifestations include disseminated intravascular coagulation (DIC) and thrombotic microangiopathy (TMA) which are significantly less common and yet far more severe [1].
Venous thromboembolism
Venous thromboembolism often starts in the valve sinus, a site that is prone to thrombosis due to reduced and abnormal blood flow, hypoxia and reduced shear stress leading to dysfunctional endothelium. Moreover, leukocytes and platelets become trapped in valve pockets. In cancer patients, tumours can cause venous compression and stasis encouraging thrombosis. VTE is a serious complication of cancer with adverse effects on patient’s quality of life and overall survival rates. Annually, 0.5% of cancer patients experience thrombosis compared to 0.1% incidence rate of thrombosis in the general population [1]. This overview will focus on VTE since it is more frequently clinically observed in cancer patients.
Arterial thromboembolism
The incidence rate of arterial thrombosis at 6 months was 4.7% in cancer and 2.2% in matched controls, according to a large prospective matched cohort study [4]. The pathogenesis differs from venous thrombosis as it typically occurs with endothelial damage. An atherosclerotic plaque with a lipid-rich core and a thin fibrous cap is prone to rupture and thrombosis, however when there is an underlying malignancy, arterial thrombosis can occur in the absence of atherosclerotic plaque. In these patients, systemic hypercoagulation is induced by several secreted factors from cancer cells such as thrombin and endothelial growth factors [4]. Many chemotherapeutic agents have been associated with increased rates of arterial thrombosis. Platinum-based agents (cisplatin), vascular endothelial growth factor (VEGF) tyrosine kinase receptor inhibitors (sorafenib/sunitinib/pazopanib) and VEGF inhibitors (bevazicumanb) have been associated with an increased risk of arterial thrombosis [5]. Co-existing hypertension, atherosclerosis and vascular anomalies contribute to arterial thrombosis by affecting blood flow and provoking platelet adhesion which plays a pivotal role in pathogenesis. On the other hand, atrial fibrillation has been identified as an important contributor to cancer-associated thrombosis by increasing the risk of stroke and systemic thromboembolism while interacting at the same time with patient factors, cancer and anticancer therapies [6] (Central illustration).
Chronic disseminated intravascular coagulation
Chronic disseminated intravascular coagulation such as disseminated intravascular coagulation (DIC) and thrombotic microangiopathy (TMA), are more severe manifestations of the procoagulant state of cancer patients. DIC is a rare complication of cancer that manifests as a consumptive coagulopathy that leads to microvascular thrombosis, thrombocytopenia, severe bleeding and organ failure. According to one study, the incidence in solid tumours was 7% [7]. TMA manifests as haemolytic uremic syndrome and thrombotic purpura. It also leads to microvascular thrombosis with increased bleeding diathesis and organ failure. TMA differs from DIC in that it is initiated by marked platelet activation, consumption and subsequent vascular endothelial injury [8].
This overview will focus on venous thrombosis since it is much more frequently clinically observed in cancer patients.
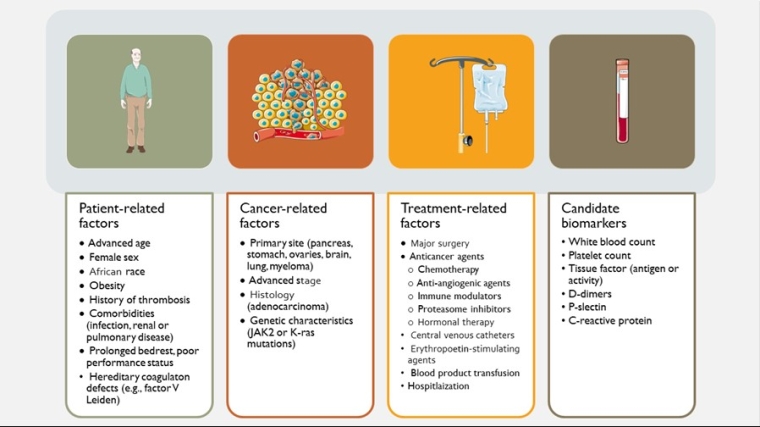
Risk factors for venous thromboembolism in cancer
The increased incidence of VTE has been attributed to the hypercoagulable state associated with cancer itself. However, the individual patient thromboembolic risk can vary significantly and depends on several factors, including tumour- and patient-specific factors, as well as anatomic and therapy associated factors (Figure 1).
Patient-related risk factors
- Older age is associated with higher risk of VTE both in the general population and in the cancer population. In retrospective cohort studies, cancer patients aged ≥65 years demonstrated a higher incidence of VTE compared to younger patients [9]. Moreover, in patients undergoing surgery for cancer treatment, the risk of VTE seems to increase with age irrespective of the presence of cancer. This is in part attributed to the fact that older age is accompanied by pre-coagulant factors including increased immobility and systemic activation of coagulation.
- Sex is another patient-related factor that has been investigated regarding the risk of developing VTE. Retrospective studies show that female patients are at higher risk for VTE while male patients develop arterial thromboembolism more frequently.
- Patient comorbidities, a high body mass index (BMI), a family history of VTE, coexisting cardiovascular disease, or acute illness, can all also increase the risk. Poor performance status and immobility are also recognised risk factors [9]. In the Coronavirus disease 2019 (COVID-19) era, patients with cancer may be at higher risk of developing VTE during a COVID-19 infection compared with non-cancer patients and thus, a risk assessment tool for VTE in cancer populations, (CoVID-TE risk) has been proposed [10].
- A previous history of VTE is a major risk factor for developing VTE. These patients have a 6- to 7-fold increased risk of VTE recurrence when compared with cancer patients with no history of VTE [11].
- Genetic predisposition to cardiovascular disease in general and more specifically to VTE is an important patient-related risk factor [12]. Factor V Leiden and other prothrombotic genetic variants, including prothrombin G20210A and variants in the ABO, FGG, and F11 genes have been shown to confer additional risk of VTE in patients with cancer. The susceptibility to cardiotoxicity from anticancer therapies, including thromboembolic complications, is also influenced by genetics [12]. In the era of precision medicine, the identification of patients at risk for cancer-associated thrombosis, based on genetic risk stratification, could help establish additional cardiovascular protection strategies.
Cancer-related risk factors
Cancer itself represents a procoagulant state. Cancer-associated hypercoagulability involves abnormalities in the coagulation and fibrinolytic pathway and platelet activation. Moreover, the anatomic location of the tumour may promote thrombosis by compressing externally or invading major veins, accelerating their obstruction.
The cancer site is one of the most important risk factors for cancer-associated thrombosis. The highest risk of VTE are observed in pancreatic, stomach, brain, colorectal, lung, and ovarian cancers [13]. Respectively, in the Khorana risk score, pancreas and stomach cancers are considered very high risk and are assigned 2 points, while lung, lymphoma, gynaecologic, bladder and testicular cancer are assigned 1 point [9,14].
- The cancer stage also plays an important role. In oncologic surgery, advanced stages have been associated with almost 3-fold higher rates of VTE [15]. Patients with metastatic disease showed 1.4- to 21.5-fold higher risk of VTE [16]. On the other hand, an advanced stage could also be a surrogate of poor performance status, also associated with increased risk of VTE.
- Histological type and grade further influence the risk of VTE. Adenocarcinomas are known to carry a higher risk of thrombosis, while high-grade tumours have been associated with an almost 2-fold higher incidence of VTE, indicating a link between tumour differentiation and thrombosis pathogenesis [9].
- Time since diagnosis plays a role in the risk for VTE. Specifically, the risk for VTE is highest during the initial period after diagnosis. The active disease status, the intensity of treatments received, and associated hospitalisation and procedures may account for this risk.
Treatment-related risk factors
Cancer therapy related factors include systemic therapy (chemotherapy, targeted and immunotherapy), supportive therapy, cancer surgery, central venous catheters, and hospitalisation.
- Anticancer drugs have been identified as an important independent risk factor for VTE in cancer, increasing the risk of VTE by 3-fold compared with no treatment and 24-fold compared with matched controls without cancer [13]. The thrombotic risk varies with different agents. Platin-based regimens, especially with cisplatin, have been associated with higher risk for VTE [5]. Tamoxifen, a selective oestrogen receptor modulator used in breast cancer, is associated with an increased risk of VTE, especially in the first years of treatment. Anti-vascular endothelial growth factor (VEGF) targeted therapies, including the monoclonal antibody bevacizumab, have been mostly associated with arterial thrombosis. The association with venous thrombosis remains inconsistent among studies [17]. Other targeted and immunological therapies that have been associated with increased thrombotic risk include cyclin-dependent kinase inhibitors (palbociclib, abemaciclib), immunomodulatory drugs (thalidomide, lenalidomide), immune check point inhibitors and CAR-T cell therapy [5].
- Indwelling vascular access devices (catheters or ports), hospitalisation and cancer surgery also increase the risk of VTE. The rate of thrombosis associated with central catheters ranges from 27 to 66%. Hospitalisations significantly increase the risk of thrombosis, while cancer surgery itself does less, as the rates of compliance with thromboprophylaxis are higher in the surgical setting [9,14].
- Cancer surgery increases the risk of postoperative DVT 2-fold and that of fatal PE greater than 3-fold when compared to similar procedures in non-cancer patients [18]. Historically, the observed incidence of VTE has been greatest in cancer patients admitted to hospital for surgery or acute medical illness. This risk has been reduced over the years due to early mobilisation following surgery, improvement of prophylaxis and perioperative care [18].
Therapeutic challenges
Proper antithrombotic therapy of VTE in cancer patients faces several challenges. The higher prothrombotic and, at the same time, bleeding risk and the frequent drug-drug interactions with anticancer and supportive therapies require a tailored management of each individual case [3]. It has been proposed that decision-making for anticoagulation therapy for either VTE or atrial fibrillation in patients with active cancer is based on four main pillars that are summarised in the acronym TIBP: T, risk of thrombosis; B, risk of bleeding; I, drug-drug interactions; P, patient preferences [19]. This structured approach for anticoagulation in active cancer has also been adopted by the recent ESC Guidelines on Cardio-Oncology [5]. In addition, any therapeutic decision should be re-evaluated periodically, as both thrombosis and bleeding are dynamic conditions that may alter significantly over the course of cancer.
Regarding anticoagulation choices for VTE in active cancer, a minority of patients will not be eligible for anticoagulation due to very high bleeding risk, while for many, low-molecular-weight-heparins (LMWH) will be the preferred strategy due to increased bleeding risk or drug-drug interactions [5,20]. For the remaining patients, there is the possibility to choose between LMWH and direct oral anticoagulants [5,20].
It has become apparent that cancer patients, particularly those with an active malignancy, are a special population in whom individual risk factors, the type and stage of cancer and the type of treatment and management they receive play a great role in the risk of developing thrombosis. All of the above issues regarding VTE in cancer patients are covered in a comprehensive and practical manner in the following chapters of the current issue of the Journal.
Conclusions/Impact on Practice statement
Patients with active cancer have a high risk of thromboembolic complications. Venous thromboembolism (VTE) is a common and serious complication in cancer patients. Healthcare providers should promptly diagnose the underlying condition and initiate proper antithrombotic therapy taking into consideration the risk of thrombosis, the risk of bleeding, the interactions between anticoagulants and cancer-treatment agents and patients’ preferences.
Note to editors
Authors:
Dimitrios Farmakis1,2, MD, PhD, FESC, FHFA; Eirini Solomou3, MD, PhD; Georgios Benetos3, MD, PhD; Victoria Evagorou1; Dimitrios Papadopoulos1; Georgios Papingiotis4, MD; Konstantinos Toutouzas3, MD, PhD, FESC
- University of Cyprus Medical School, Nicosia, Cyprus;
- Second Department of Cardiology, National and Kapodistrian University of Athens Medical School, Attikon University Hospital, Athens, Greece;
- First Department of Cardiology, National and Kapodistrian University of Athens Medical School, Hippokration General Hospital, Athens, Greece;
- Department of Cardiology, Evangelismos General Hospital, Athens, Greece
Address for correspondence:
Prof. Dimitrios Farmakis, University of Cyprus Medical School, Shakolas Educational Center for Clinical Medicine, Palaios dromos Lefkosias Lemesou No.215/6, 2029 Aglantzia, Nicosia, Cyprus
Twitter handle: @FarmakisD
Author disclosures:
D. Farmakis reports speaker honoraria and consultation fees from Bayer and Leo. The other authors have no conflicts of interest to declare.
References
- Sud R, Khorana AA. Cancer-associated thrombosis: risk factors, candidate biomarkers and a risk model.Thromb Res. 2009;123 Suppl 4:S18-21.
- Khorana AA, Francis CW, Culakova E, Kuderer NM, Lyman GH. Thromboembolism is a leading cause of death in cancer patients receiving outpatient chemotherapy.J Thromb Haemost. 2007;5:632–4.
- Farmakis D, Papakotoulas P, Angelopoulou E, Bischiniotis T, Giannakoulas G, Kliridis P, Richter D, Paraskevaidis I. Anticoagulation for atrial fibrillation in active cancer.Oncol Lett. 2022;23:124.
- Navi BB, Reiner AS, Kamel H, Iadecola C, Okin PM, Elkind MSV, Panageas KS, DeAngelis LM. Risk of Arterial Thromboembolism in Patients With Cancer.J Am Coll Cardiol. 2017;70:926–38.
- Lyon AR, López-Fernández T, Couch LS, Asteggiano R, Aznar MC, Bergler-Klein J, Boriani G, Cardinale D, Cordoba R, Cosyns B, Cutter DJ, de Azambuja E, de Boer RA, Dent SF, Farmakis D, Gevaert SA, Gorog DA, Herrmann J, Lenihan D, Moslehi J, Moura B, Salinger SS, Stephens R, Suter TM, Szmit S, Tamargo J, Thavendiranathan P, Tocchetti CG, van der Meer P, van der Pal HJH; ESC Scientific Document Group. 2022 ESC Guidelines on cardio-oncology developed in collaboration with the European Hematology Association (EHA), the European Society for Therapeutic Radiology and Oncology (ESTRO) and the International Cardio-Oncology Society (IC-OS).Eur Heart J. 2022;43:4229–361.
- Farmakis D, Parissis J, Filippatos G. Insights into onco-cardiology: atrial fibrillation in cancer.J Am Coll Cardiol. 2014;63:945–953.
- Sallah S, Wan JY, Nguyen NP, Hanrahan LR, Sigounas G. Disseminated intravascular coagulation in solid tumors: clinical and pathologic study. Thromb Haemost. 2001;86:828–33.
- Wada H, Matsumoto T, Suzuki K, Imai H, Katayama N, Iba T, Matsumoto M. Differences and similarities between disseminated intravascular coagulation and thrombotic microangiopathy.Thromb J. 2018;16:14.
- Khorana AA, Connolly GC. Assessing risk of venous thromboembolism in the patient with cancer.J Clin Oncol. 2009;27:4839–47.
- Li A, Kuderer NM, Hsu C-Y, Shyr Y, Warner JL, Shah DP, Kumar V, Shah S, Kulkarni AA, Fu J, Gulati S, Zon RL, Li M, Desai A, Egan PC, Bakouny Z, Kc D, Hwang C, Akpan IJ, McKay RR, Girard J, Schmidt AL, Halmos B, Thompson MA, Patel JM, Pennell NA, Peters S, Elshoury A, Lima Lopes G de, Stover DG, Grivas P, Rini BI, Painter CA, Mishra S, Connors JM, Lyman GH, Rosovsky RP; CCC19 consortium. The CoVID-TE risk assessment model for venous thromboembolism in hospitalized patients with cancer and COVID-19.J Thromb Haemost. 2021;19:2522–32.
- Connolly GC, Khorana AA. Emerging risk stratification approaches to cancer-associated thrombosis: risk factors, biomarkers and a risk score.Thromb Res. 2010;125 Suppl 2:S1-7.
- Farmakis D, Papingiotis G, Filippatos G. Genetic Predisposition to Cardiovascular Disease in Patients With Cancer.JACC: CardioOncology. 2023. Epublished.
- Mulder FI, Horváth-Puhó E, van Es N, van Laarhoven HWM, Pedersen L, Moik F, Ay C, Büller HR, Sørensen HT. Venous thromboembolism in cancer patients: a population-based cohort study.Blood. 2021;137:1959–69.
- Tuzovic M, Herrmann J, Iliescu C, Marmagkiolis K, Ziaeian B, Yang EH. Arterial Thrombosis in Patients with Cancer.Curr Treat Options Cardiovasc Med. 2018;20:40.
- Agnelli G, Bolis G, Capussotti L, Scarpa RM, Tonelli F, Bonizzoni E, Moia M, Parazzini F, Rossi R, Sonaglia F, Valarani B, Bianchini C, Gussoni G. A clinical outcome-based prospective study on venous thromboembolism after cancer surgery: the @RISTOS project.Ann Surg. 2006;243:89–95.
- Chew HK, Wun T, Harvey D, Zhou H, White RH. Incidence of venous thromboembolism and its effect on survival among patients with common cancers.Arch Intern Med . 2006;166:458–64.
- Herrmann J, Yang EH, Iliescu CA, Cilingiroglu M, Charitakis K, Hakeem A, Toutouzas K, Leesar MA, Grines CL, Marmagkiolis K. Vascular Toxicities of Cancer Therapies: The Old and the New--An Evolving Avenue.Circulation. 2016;133:1272–89.
- Khorana AA, Dalal M, Lin J, Connolly GC. Incidence and predictors of venous thromboembolism (VTE) among ambulatory high-risk cancer patients undergoing chemotherapy in the United States.Cancer. 2013;119:648–55.
- Farmakis D. Anticoagulation for atrial fibrillation in active cancer: what the cardiologists think.Eur J Prev Cardiol. 2021;28:608–10.
- Falanga A, Ay C, Di Nisio M, Gerotziafas G, Jara-Palomares L, Langer F, Lecumberri R, Mandala M, Maraveyas A, Pabinger I, Sinn M, Syrigos K, Young A, Jordan K; ESMO Guidelines Committee. Venous thromboembolism in cancer patients: ESMO Clinical Practice Guideline.Ann Oncol. 2023:S0923-7534(22)04786-X.

















