Keywords
advanced heart failure, advanced HF therapies, heart transplantation, left ventricular assist device, mechanical circulatory support
Abbreviations
GDMT: guideline-directed medical therapy
HF: heart failure
HT: heart transplantation
LVAD: left ventricular assist device
LVEF: left ventricle ejection fraction
Take-home messages
- Treatment with maximally tolerated GDMTs is important to be addressed before a diagnosis of advanced HF is established.
- To improve prognosis, early referral of eligible patients for advanced HF therapies in dedicated centres before end-organ damage is irreversible, is crucial.
- HT remains the gold-standard therapy for advanced HF, but durable LVADs are life-prolonging therapies for patients who are ineligible for HT.
- A complete evaluation to exclude reversible and potentially treatable causes of HF (arrhythmias, conduction disorders, coronary artery disease, valvular diseases, thyroid disease, and cardiotoxicity) is mandatory before considering the diagnosis of advanced HF.
Impact on current practice
This manuscript aims to offer contemporary information for general cardiologists and primary care physicians about durable advanced HF therapies (LVAD/HT), outlining the importance of early recognition of advanced HF and timely referral to advanced HF centres that can provide advanced therapies. Unfortunately, these patients are often referred too late, when irreversible end-organ dysfunction has developed, and futility overcomes any treatment benefit. Thus, a better understanding of the definition of advanced HF accompanied by insights into the appropriate selection of patients who need a referral to highly specialised centres with facilities, staffing, and expertise for advanced HF therapies can critically impact current clinical practice. The process of patient selection, the evaluation in specialised centres, and decision-making concerning either LVAD or HT are clinically relevant.
Epidemiology, definition, diagnosis criteria, prognosis
Despite the new pharmacotherapies, devices, or surgical therapies, heart failure [HF] has a long-term progressive trajectory, and given the aging population, is resulting in an increasing prevalence of advanced HF [1]. The prevalence is reported to be up to 15% of the overall population with HF [2]. The prognosis of advanced HF is poor and despite the great efforts made in well-conducted programs of life-prolonging therapies (left ventricular assist device [LVAD] and heart transplantation [HT]), the reported 1-year mortality remains close to 50% [2].
Advanced HF is the phase in the evolution of the disease characterised by persistent and progressive symptoms despite guideline-directed medical therapies (GDMTs). The definition of this stage was proposed by several professional societies that integrated different combinations of clinical parameters, objective data, and therapeutic interventions. Also, several overlapping terminologies have been used frequently, such as “end-stage”, “terminal”, and “advanced”, that created different thresholds for prognostic expectations. Therefore, in 2018 the Heart Failure Association of the HFA-ESC developed a set of diagnostic criteria to define advanced HF, that could be easily applicable in clinical practice. A new definition emerged out of a need for early recognition, early referral to HF centres, and subsequent early life-saving interventions that could improve survival rates [3].
The current HFA-ESC diagnostic criteria consist of four domains:
- severe and persistent symptoms (NYHA Class III or IV),
- severe cardiac dysfunction (LVEF ≤30%, isolated right ventricular (RV) failure, non-operable severe valve abnormalities, non-operable severe congenital abnormalities, persistently high natriuretic peptides values, and severe left ventricular diastolic dysfunction or structural abnormalities),
- hospitalisations due to acute decompensation of HF (>1 unplanned visit or hospitalisation in the last 12 months) and
- severe impairment of exercise capacity or low 6-minute walking test (6MWT) distance (<300 m) or pVO2 <12 mL/kg/min or <50% predicted value, estimated to be of cardiac origin.
The definition has been validated in the Assessment of the I Need Help markers in Heart Failure (HELP-HF) registry, which showed a stepwise increase in the severity of prognosis with the number of criteria. Patients meeting all four criteria of the current definition are those who need to be referred to advanced HF centres and to be evaluated for durable advanced therapies (1-year rate of all-cause mortality or first HF hospitalisation of 69.3% compared to patients with no criteria 34.3%) [4].
Criteria for referral to an advanced HF centre
Using the definition, in the presence of clinical markers for advanced HF, and when clinical stabilisation after intensive disease management is unobtainable, physicians are encouraged to refer the patient to an advanced HF centre. A mnemonic, “I-NEED-HELP” has previously been proposed to identify those patients who require an early referral (Table 1) [5].
Although not always straightforward, one of the most important steps in treating HF patients is establishing the optimal moment for the proper referral to an advanced HF team. The team is oriented towards the optimisation of therapy, comprehensive risk assessment, and facilitation of shared decision-making [6]. The concept of “shared care” was proposed to optimise the management of severe patients and patients with chronic diseases through improved communication between primary and speciality centres and knowledge sharing [7]. At this particular stage, markers of advanced HF can be useful for clinicians (Table 1).
Table 1. Markers of advanced heart failure. [3], [5]
“I NEED HELP” (2017 Criteria)
- I: Inotropes
- N: NYHA class, natriuretic peptides
- E: LVEF ≤25%
- E: End-organ dysfunction (renal, liver)
- D: Defibrillator shock
- H: Hospitalisation
- E: Oedema, escalating
- L: Low blood pressure, high heart rate
- P: Prognostic medications
Markers of Advanced Heart Failure (HFA-ESC 2021)
- Prior inotropic use
- NYHA III–IV despite optimal medical therapy (including ICD/CRT when indicated)
- LVEF <20%
- Worsening right ventricular function
- Worsening renal function or liver function
- Ventricular arrhythmias or ICD shocks
- More than one admission or unplanned visit to heart failure clinic within the last 12 months
- Need for escalating diuretic doses for persistent congestion
- Non-response to cardiac resynchronisation therapy
- Systolic blood pressure <90 mmHg and/or signs of peripheral hypoperfusion
- Intolerant to beta-blockers or ACEi/ARB/ARNI
ACE: angiotensin-converting enzyme; ARB: angiotensin II receptor blocker; ARNI: angiotensin receptor-neprilysin inhibitor; CRT: cardiac resynchronisation therapy; HF: heart failure; HFA-ESC: Heart Failure Association of the European Society of Cardiology; ICD: implantable cardioverter defibrillator; LVEF: left ventricular ejection fraction; NYHA: New York Hear Association; RV: right ventricle; SBP: systolic blood pressure
Treatment of advanced chronic HF – general considerations
Heart transplantation represents the gold standard for treating advanced HF, but a shortage of donors and HT contraindications are the main limitations of the therapy nowadays. In consequence, LVAD has emerged as one suitable option for treating patients with advanced HF, when traditional treatment is no longer effective. The recommended approach for management and patient selection is an organisational referral, based on a model using a “hub and spoke” network which should be regionally implemented [8].
Heart transplantation
HT is the treatment of choice for patients with advanced HF in the absence of contraindications, with an overall median survival of 12.5 years and conditional survival of 14.8 years for those who survive the first year [9]. However, less than 5,000 HT are performed annually, therefore the epidemiological impact is trivial [1]. Current guidelines recommend HT for patients with advanced HF, who are refractory to medical/device therapy, in the absence of absolute contraindication, and who have no other therapeutic options except for LVAD as a bridge to transplant [3].
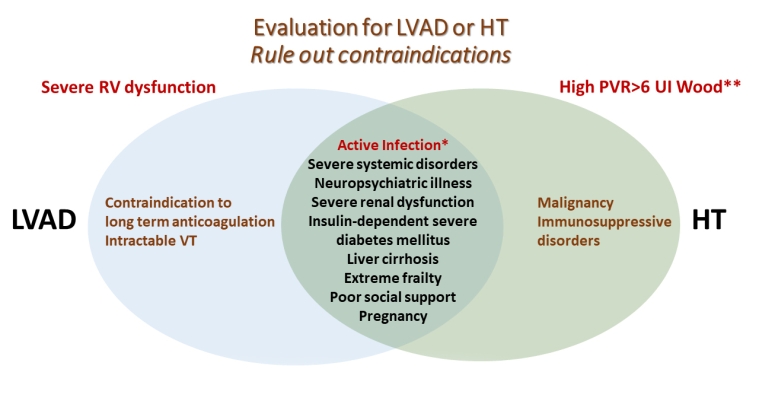
Contraindications include fixed pulmonary hypertension, severe illnesses with a life expectancy of less than 2 years, active infection (except LVAD-related infection which is a prioritised indication for HT), severe peripheral arterial or cerebrovascular disease, malignancy, irreversible liver dysfunction (cirrhosis) or irreversible renal dysfunction (liver-heart and kidney-heart transplantation can be considered), systemic disease with multiorgan involvement or another serious comorbidity with poor prognosis, alcohol or drug abuse, psychological instability that jeopardises proper follow-up and insufficient social support (Figure 1) [7]. Weight loss is recommended in obese patients to achieve a BMI <35 kg/m² [10].
LVAD: left ventricular assist device; HT: heart transplantation; PVR: pulmonary vascular resistance; RV: right ventricular; VT: ventricular arrhythmias.
*Active, persistent infection in LVAD-assisted patients is an indication of HT.
**Despite the use of vasodilators.
In cases of severe pulmonary hypertension (pulmonary vascular resistance [PVR] of 5-6 Wood units), LVAD should be considered as an option to completely or partially reverse elevated PVR to obtain eligibility for HT. The reason is that the healthy RV of the donor’s heart is not conditioned to handle pulmonary hypertension and may fail in the operating room or the early postoperative period [1]. In the same manner, multiorgan dysfunction could be reversed in LVAD-assisted patients.
LVAD: when and whom to refer?
In HT-ineligible patients, LVAD is recommended as a destination therapy (DT) to prolong and improve quality of life, or as bridge to transplant/bridge to candidacy (BTT/BTC) to reverse contraindications to HT [3]. The REMATCH trial was the first trial showing a clinically meaningful survival benefit and an improved quality of life after LVAD implantation as a DT [3], [11]. Since then, significant technological advances were achieved with each generation of LVADs for longer durability, lower device complications, and higher survival.
Nowadays, the use of the HeartMate 3 (HM3; Abbott), the newest-generation LVAD with a fully magnetically levitated centrifugal pump, has led to improved haemocompatibility, improved survival, and significantly reduced pump thrombosis [10]. Compared to the previous generation of axial pumps, the centrifugal flow pump used in the MOMENTUM 3 trial showed lower rates of disabling stroke, major bleeding, and gastrointestinal haemorrhage [12]. Nowadays, based on the MOMENTUM 3 results, HM3 LVAD therapy has emerged as a valuable therapeutic option in treating patients with advanced HF and is comparable to HT in terms of 1 and 2-year survival rates (86.6% and 82.3%, respectively) [8] [12] [13].
However, the reported incidence of bleeding events, thromboembolic events, driveline infections, and RV failure remained similar to those of the older devices [14]. Hospital readmissions are also prevalent, 1 out of 3 LVAD-assisted patients is readmitted in the first 3 months after LVAD implantation for complications or adverse events [15].
Contraindications to LVAD support usually overlap with those for HT (Figure 1). Unlike HT, contemporary LVADs need anticoagulation to avoid pump thrombosis. Although the HM3 has a lower risk of pump thrombosis as compared to previous-generation devices, the prothrombin time/international normalised ratio should be maintained between 1.8 and 2.3, thus a proper evaluation of the coagulation state and conditions predisposing to bleeding is mandatory, as they could contraindicate LVAD therapy.
Another important issue in considering LVAD therapy is RV dysfunction. RV failure is recognised as a major contributor to adverse outcomes and death following LVAD implantation [16]. A certain degree of RV dysfunction is present in patients undergoing LVAD implantation and changes in ventricular interdependence after LVAD implantation modifies RV haemodynamics with consecutive RV dysfunction. RV failure is the main concern in the perioperative period in LVAD implantation, with early RV dysfunction being a common complication in up to 40% of patients [17]. Strategies to prevent RV dysfunction have large institutional variability and are a matter of ongoing discussion.
Selecting patients for either LVAD or HT
HF patients unlikely to recover, who present aggravated symptoms despite aggressive attempts at medical, surgical, electric, and mechanical optimisation should be considered candidates for durable advanced HF therapies.
A complete clinical evaluation is the first step in approaching every patient, with an assessment of clinical and haemodynamic stability, systemic perfusion, and multi-organ dysfunction [10]. The evaluation process should be structured to establish the patient’s eligibility for either LVAD and/or HT, rule out any contraindications, and the benefit-risk ratio of either LVAD and/or HT should evolve in parallel until the final decision-making by the Heart Team. At the end of the evaluation process, physicians should have a clear perspective on the severity of the HF condition and the presence of comorbidities that might impact survival or quality of life after HT or LVAD implantation [18].
Identifying absolute contraindications for LVAD and HT is the second step in assessing patients with advanced HF (Figure 1). After excluding ineligibility based on severe associated comorbidities, the patient should be referred to an advanced HF centre. It should be noted that some of the conditions, such as reversible severe pulmonary hypertension, that represent contraindications for an HT listing at the time of evaluation may improve after haemodynamic support by LVAD. Thus, the patient may have a chance to be reassessed and listed after a certain period of assistance (BTC) [19]. In cases of ineligibility for either LVAD or HT, palliative care should be considered.
The set of mandatory investigations for decision-making in a specialised centre is listed below:
Clinical evaluation and defining advanced HF: an objective evaluation of HF symptoms and signs focused on congestion and hypoperfusion is mandatory along with demonstrating a lack of improvement of current HF status despite maximal pharmacological and non-pharmacological treatment.
Biomarkers and laboratory testing: testing for multiple organ dysfunction should be carefully assessed. Severe renal and liver dysfunction due to haemodynamic deterioration could be a contraindication for HT, but these patients can be candidates for LVAD support as BTC. Anaemia, thrombocytopenia, and other coagulation abnormalities should be evaluated and treated. A prothrombotic state must be excluded to minimise the risk of pump thrombosis after LVAD implantation and conditions that predispose to severe bleeding should be well known before the decision is made, due to the need for anticoagulation therapy in LVAD-assisted patients and the high risk of haemorrhagic events.
Cardiac imaging: (including transthoracic/transoesophageal echocardiography, coronary angiography). RV and LV function, valvular dysfunction (particularly aortic regurgitation [AR]), concomitant coronary artery disease, and other structural abnormalities may need to be addressed before or at the time of LVAD implantation. RV dysfunction and AR are particularly important. AR creates a circulatory shunt or a ‘closed circulatory loop’ between the pump, valve, LV, and back to the pump, thus concomitant aortic valve procedures at the time of LVAD implantation are recommended in patients with moderate or severe AR [20].
Right heart catheterisation (RHC): for assessing filling pressures, PVR, transpulmonary gradient (TPG), and cardiac output (CO). RHC is recommended in all patients evaluated for HT or LVAD. An elevated TPG, a reflection of passive or reactive high pulmonary arterial pressures due to persistently elevated LV filling pressures can be improved after a proper decongestion and the augmentation of the CO.
RHC is of great importance in the RV function evaluation and proper risk stratification before LVAD implantation. Central venous pressure (CVP), the ratio of CVP to pulmonary capillary wedge pressure (PCWP), the pulmonary artery pulsatility index (PAPi), and the RV stroke work index are some of the parameters used for predicting early and late RV failure. In the settings of RV dysfunction, pulmonary vasodilators might be considered but their beneficial effects were not demonstrated [21].
Functional studies: (cardiopulmonary stress testing [CPET]). CPET is a risk stratification test, providing parameters impacted by cardiac, pulmonary, peripheral, and psychological factors and it is recommended as a part of the evaluation for both HT and/or LVAD [3].
Multidisciplinary teams (pulmonary, neurology, gastroenterology, oncology): are often needed for evaluating associated comorbidities and future impact upon outcomes after LVAD or HT.
Eligibility for each of the options may change according to the particular conditions of each patient, which may also change over time. As such, periodical reassessment in cases of LVAD-assisted patients as BTT or BTC therapy should be considered for future HT listing. In these patients, LVAD therapy may offer a transplant opportunity to the patient when eligible, as being the gold-standard therapy for advanced HF [8].

Risk stratification models
Various attempts to stratify risk before LVAD implantation have been proposed. The most longitudinal and widely used classification is the Interagency Registry for Mechanically Assisted Circulatory Support (INTERMACS) scale, which divides advanced HF patients into 7 different risk levels based on clinical severity and intensity of treatment (Figure 2) [3] [22]. Although useful in better defining the prognosis and urgency of intervention for patients receiving the treatment, it does not include variables related to clinical history [23]. INTERMACS is also utilised to gauge morbidity, mortality, and health‐related quality of life outcomes. Initially, LVADs have been used in INTERMACS profiles 1–3 but increased attention has turned to the INTERMACS profiles 4–7 as a potential candidate for LVAD therapy, to mitigate post-implantation morbidity and mortality by timing intervention before critical clinical decline.
Recently, the investigators from the MOMENTUM 3 trial proposed a risk score that includes six commonly available pre-implantation variables: age, prior coronary or valvular surgery, serum sodium, blood urea nitrogen, LV end-diastolic diameter, and haemodynamic findings (RAP/PCWP ratio). The score accurately determined individualised 1- and 2-year survival after HM3 LVAD implantation in patients with advanced HF and can be used to provide individual survival estimates and to discuss the degree of increase in expected survival when compared with medical therapy alone [24].
However, prognostication is complex and although helpful for selection, it is not required for referral to an advanced HF centre [8].
NYHA: New York Heart Association
Palliative care
Although it is not the purpose of this document, we must be reminded that based on current guidelines and recommendations, all patients in the advanced stages of HF and those considered for LVAD or HT should receive a palliative care consultation before intervention [3]. It is an important step, as it provides information about realistic expectations and goals of both the patients and their families/caregivers, regardless of their eligibility for advanced therapies, as future considerations should be based on their acceptance of long-term commitment to either therapy [8] [25]. Patients with contraindications to mechanical circulatory support or HT should be considered for palliative care.
Conclusions
The triage of patients with advanced HF and the appropriate timing of referral to advanced HF centres is still a challenge for general clinicians. A useful multiparametric algorithm has been proposed by the HFA-ESC, to help clinicians identify patients with advanced HF in need of advanced therapies. Raising awareness about the poor prognosis associated with this stage of the disease is important in the context of the larger number of patients in need of advanced HF therapies who are referred too late to have the chance for a better outcome.
Note to editors
Authors:
Oliviana Geavlete1, MD, PHD; Laura Antohi1, MD, PHD; Binyamin Ben-Avraham2, MD; Tuvia Ben Gal2, MD
- “Prof. Dr. CC. Iliescu” Emergency Institute of Cardiovascular Diseases, “Carol Davila” University of Medicine and Pharmacy, Bucharest, Romania;
- Heart Failure Unit, Cardiology Department, Rabin Medical Center, Sackler Faculty of Medicine, Tel Aviv University, Tel Aviv, Israel
Address for correspondence:
Dr Oliviana Geavlete, 258 Fundeni Street, 022238, Bucharest, Romania
Author disclosures:
The authors have no conflicts of interest to declare regarding this article.
References
- Guglin M, Zucker MJ, Borlaug BA, Breen E, Cleveland J, Johnson MR, Panjrath GS, Patel JK, Starling RC, Bozkurt B; ACC Heart Failure and Transplant Member Section and Leadership Council. Evaluation for Heart Transplantation and LVAD Implantation: JACC Council Perspectives.J Am Coll Cardiol. 2020;75):1471-87.
- Dunlay SM, Roger VL, Killian JM, Weston SA, Schulte PJ, Subramaniam AV, Blecker SB, Redfield MM. Advanced Heart Failure Epidemiology and Outcomes: A Population-Based Study.JACC Hear Fail. 2021;9:722-32.
- McDonagh TA, Metra M, Adamo M, Gardner RS, Baumbach A, Böhm M, Burri H, Butler J, Čelutkienė J, Chioncel O, Cleland JGF, Coats AJS, Crespo-Leiro MG, Farmakis D, Gilard M, Heymans S, Hoes AW, Jaarsma T, Jankowska EA, Lainscak M, Lam CSP, Lyon AR, McMurray JJV, Mebazaa A, Mindham R, Muneretto C, Francesco Piepoli M, Price S, Rosano GMC, Ruschitzka F, Kathrine Skibelund A; ESC Scientific Document Group. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure.Eur Heart J. 2021;42:3599-726.
- Pagnesi M, Lombardi CM, Chiarito M, Stolfo D, Baldetti L, Loiacono F, Tedino C, Arrigoni L, Ghiraldin D, Tomasoni D, Inciardi RM, Maccallini M, Villaschi A, Gasparini G, Montella M, Contessi S, Cocianni D, Perotto M, Barone G, Merlo M, Cappelletti AM, Sinagra G, Pini D, Metra M. Prognostic impact of the updated 2018 HFA-ESC definition of advanced heart failure: results from the HELP-HF registry.Eur J Heart Fail. 2022;24:1493-1503.
- Baumwol J. “I Need Help”-A mnemonic to aid timely referral in advanced heart failure.J Heart Lung Transplant. 2017;36:593-4.
- Saeed D, Feldman D, El Banayosy A, Birks E, Blume E, Cowger J, Hayward C, Jorde U, Kremer J, MacGowan G, Maltais S, Maybaum S, Mehra M, Shah KB, Mohacsi P, Schweiger M, Schroeder SE, Shah P, Slepian M, Tops LF, Alvarez P, Arabia F, Aslam S, Benson-Louis L 4th, Birati E, Buchholz HW, Cedars A, Christensen D, Ciarka A, Coglianese E, Cogswell R, Cook J, Copeland J, Costello JG, Drakos SG, Eghtesady P, Elliot T, Estep JD, Eulert-Grehn JJ, Fabrizio R, Garbade J, Gelow J, Guglin M, Hernandez-Montfort J, Horstmanshof D, John R, Kanwar M, Khaliel F, Kim G, Kumar S, Lavee J, Leache M, Leprince P, Lim S, Loforte A, Maly J, Najjar S, Netuka I, Pamboukian SV, Patel SR, Pinney S, Pluym CV, Potapov E, Robson D, Rochlani Y, Russell S, Sandau K, Sandoval E, Sayer G, Schettle S, Schibilsky D, Schlöglhofer T, Schmitto J, Siddique A, Silvestry S, Slaughter MS, Sun B, Takayama H, Tedford R, Teuteberg JJ, Ton VK, Uriel N, Vierecke J, Zimpfer D, D'Alessandro D. The 2023 International Society for Heart and Lung Transplantation Guidelines for Mechanical Circulatory Support: A 10-Year Update.J Heart Lung Transplant. 2023;42:e1-e222.
- Cameli M, Pastore MC, Mandoli GE, Landra F, Lisi M, Cavigli L, D'Ascenzi F, Focardi M, Carrucola C, Dokollari A, Bisleri G, Tsioulpas C, Bernazzali S, Maccherini M, Valente S. A multidisciplinary approach for the emergency care of patients with left ventricular assist devices: A practical guide.Front Cardiovasc Med. 2022;9:923544.
- Crespo-Leiro MG, Metra M, Lund LH, Milicic D, Costanzo MR, Filippatos G, Gustafsson F, Tsui S, Barge-Caballero E, De Jonge N, Frigerio M, Hamdan R, Hasin T, Hülsmann M, Nalbantgil S, Potena L, Bauersachs J, Gkouziouta A, Ruhparwar A, Ristic AD, Straburzynska-Migaj E, McDonagh T, Seferovic P, Ruschitzka F. Advanced heart failure: a position statement of the Heart Failure Association of the European Society of Cardiology.Eur J Heart Fail. 2018;20:1505-35.
- Khush KK, Cherikh WS, Chambers DC, Harhay MO, Hayes D Jr, Hsich E, Meiser B, Potena L, Robinson A, Rossano JW, Sadavarte A, Singh TP, Zuckermann A, Stehlik J; International Society for Heart and Lung Transplantation. The International Thoracic Organ Transplant Registry of the International Society for Heart and Lung Transplantation: Thirty-sixth adult heart transplantation report - 2019; focus theme: Donor and recipient size match.J Heart Lung Transplant. 2019;38:1056-66.
- Truby LK, Rogers JG. Advanced Heart Failure: Epidemiology, Diagnosis, and Therapeutic Approaches.JACC Heart Fail. 2020;8:523-36.
- Rose EA, Gelijns AC, Moskowitz AJ, Heitjan DF, Stevenson LW, Dembitsky W, Long JW, Ascheim DD, Tierney AR, Levitan RG, Watson JT, Meier P, Ronan NS, Shapiro PA, Lazar RM, Miller LW, Gupta L, Frazier OH, Desvigne-Nickens P, Oz MC, Poirier VL; Randomized Evaluation of Mechanical Assistance for the Treatment of Congestive Heart Failure (REMATCH) Study Group. Long-Term Use of a Left Ventricular Assist Device for End-Stage Heart Failure.N Engl J Med. 2001;345:1435-43.
- Mehra MR, Goldstein DJ, Uriel N, Cleveland JC Jr, Yuzefpolskaya M, Salerno C, Walsh MN, Milano CA, Patel CB, Ewald GA, Itoh A, Dean D, Krishnamoorthy A, Cotts WG, Tatooles AJ, Jorde UP, Bruckner BA, Estep JD, Jeevanandam V, Sayer G, Horstmanshof D, Long JW, Gulati S, Skipper ER, O'Connell JB, Heatley G, Sood P, Naka Y; MOMENTUM 3 Investigators. Two-Year Outcomes with a Magnetically Levitated Cardiac Pump in Heart Failure.N Engl J Med. 2018;378:1386-95.
- Ben Avraham B, Crespo-Leiro MG, Filippatos G, Gotsman I, Seferovic P, Hasin T, Potena L, Milicic D, Coats AJS, Rosano G, Ruschitzka F, Metra M, Anker S, Altenberger J, Adamopoulos S, Barac YD, Chioncel O, De Jonge N, Elliston J, Frigeiro M, Goncalvesova E, Grupper A, Hamdan R, Hammer Y, Hill L, Itzhaki Ben Zadok O, Abuhazira M, Lavee J, Mullens W, Nalbantgil S, Piepoli MF, Ponikowski P, Ristic A, Ruhparwar A, Shaul A, Tops LF, Tsui S, Winnik S, Jaarsma T, Gustafsson F, Ben Gal T. HFA of the ESC Position paper on the management of LVAD-supported patients for the non-LVAD specialist healthcare provider Part 1: Introduction and at the non-hospital settings in the community.ESC Heart Fail . 2021;8:4394-408.
- Milicic D, Ben Avraham B, Chioncel O, Barac YD, Goncalvesova E, Grupper A, Altenberger J, Frigeiro M, Ristic A, De Jonge N, Tsui S, Lavee J, Rosano G, Crespo-Leiro MG, Coats AJS, Seferovic P, Ruschitzka F, Metra M, Anker S, Filippatos G, Adamopoulos S, Abuhazira M, Elliston J, Gotsman I, Hamdan R, Hammer Y, Hasin T, Hill L, Itzhaki Ben Zadok O, Mullens W, Nalbantgil S, Piepoli MF, Ponikowski P, Potena L, Ruhparwar A, Shaul A, Tops LF, Winnik S, Jaarsma T, Gustafsson F, Ben Gal T. Heart Failure Association of the European Society of Cardiology position paper on the management of left ventricular assist device-supported patients for the non-left ventricular assist device specialist healthcare provider: Part 2: at the emergency department.ESC Heart Fail . 2021;8:4409-24.
- Mehra MR, Nayak A, Desai AS. Life-Prolonging Benefits of LVAD Therapy in Advanced Heart Failure.JACC Heart Fail. 2023 May 24:S2213-1779(23)00249.
- Wang TS, Cevasco M, Birati EY, Mazurek JA. Predicting, Recognizing, and Treating Right Heart Failure in Patients Undergoing Durable LVAD Therapy.J Clin Med. 2022;11:2984.
- Hulde N, Koster A, von Dossow V. Perioperative management of patients with undergoing durable mechanical circulatory support.Ann Transl Med. 2020;8:830.
- Morris AA, Khazanie P, Drazner MH, Albert NM, Breathett K, Cooper LB, Eisen HJ, O'Gara P, Russell SD; American Heart Association Heart Failure and Transplantation Committee of the Council on Clinical Cardiology; Council on Arteriosclerosis, Thrombosis and Vascular Biology; Council on Cardiovascular Radiology and Intervention; and Council on Hypertension. Guidance for Timely and Appropriate Referral of Patients with Advanced Heart Failure: A Scientific Statement from the American Heart Association.Circulation. 2021;144:e238-e250.
- Ono M, Yamaguchi O, Ohtani T, Kinugawa K, Saiki Y, Sawa Y, Shiose A, Tsutsui H, Fukushima N, Matsumiya G, Yanase M, Yamazaki K, Yamamoto K, Akiyama M, Imamura T, Iwasaki K, Endo M, Ohnishi Y, Okumura T, Kashiwa K, Kinoshita O, Kubota K, Seguchi O, Toda K, Nishioka H, Nishinaka T, Nishimura T, Hashimoto T, Hatano M, Higashi H, Higo T, Fujino T, Hori Y, Miyoshi T, Yamanaka M, Ohno T, Kimura T, Kyo S, Sakata Y, Nakatani T; JCS/JSCVS/JATS/JSVS Joint Working Group. JCS/JSCVS/JATS/JSVS 2021 Guideline on Implantable Left Ventricular Assist Device for Patients With Advanced Heart Failure.Circ J.. 2022;86:1024-58.
- Bouabdallaoui N, El-Hamamsy I, Pham M, Giraldeau G, Parent MC, Carrier M, Rouleau JL, Ducharme A. Aortic regurgitation in patients with a left ventricular assist device: A contemporary review.J Heart Lung Transplant. 2018;37:1289-97.
- Kirklin JK, Pagani FD, Goldstein DJ, John R, Rogers JG, Atluri P, Arabia FA, Cheung A, Holman W, Hoopes C, Jeevanandam V, John R, Jorde UP, Milano CA, Moazami N, Naka Y, Netuka I, Pagani FD, Pamboukian SV, Pinney S, Rogers JG, Selzman CH, Silverstry S, Slaughter M, Stulak J, Teuteberg J, Vierecke J, Schueler S, D'Alessandro DA. American Association for Thoracic Surgery/International Society for Heart and Lung Transplantation guidelines on selected topics in mechanical circulatory support.J Thorac Cardiovasc Surg. 2020;159:865-96.
- Kirklin JK, Naftel DC, Stevenson LW, Kormos RL, Pagani FD, Miller MA, Ulisney K, Young JB. INTERMACS database for durable devices for circulatory support: first annual report.J Heart Lung Transplant. 2008;27:1065-72.
- Metra M, Dinatolo E, Dasseni N. The New Heart Failure Association Definition of Advanced Heart Failure.Card Fail Rev. 2019;5:5-8.
- Mehra M, Nayak A, Morris A, Lanfear DE, Nemeh H, Desai S, Bansal A, Guerrero-Miranda C, Hall S, Cleveland JC Jr, Goldstein DJ, Uriel N, Chen L, Bailey S, Anyanwu A, Heatley G, Chuang J, Estep JD. Prediction of Survival After Implantation of a Fully Magnetically Levitated Left Ventricular Assist Device.JACC Hear Fail. 2022;10:948-59.
- Gustafsson F, Ben Avraham B, Chioncel O, Hasin T, Grupper A, Shaul A, Nalbantgil S, Hammer Y, Mullens W, Tops LF, Elliston J, Tsui S, Milicic D, Altenberger J, Abuhazira M, Winnik S, Lavee J, Piepoli MF, Hill L, Hamdan R, Ruhparwar A, Anker S, Crespo-Leiro MG, Coats AJS, Filippatos G, Metra M, Rosano G, Seferovic P, Ruschitzka F, Adamopoulos S, Barac Y, De Jonge N, Frigerio M, Goncalvesova E, Gotsman I, Itzhaki Ben Zadok O, Ponikowski P, Potena L, Ristic A, Jaarsma T, Ben Gal T. HFA of the ESC position paper on the management of LVAD-supported patients for the non-LVAD specialist healthcare provider Part 3: at the hospital and discharge.ESC Hear Fail. 2021;8:4425-43.


















