Take-home messages
- 2023 ESC Guidelines for the management of cardiovascular disease in patients with diabetes.
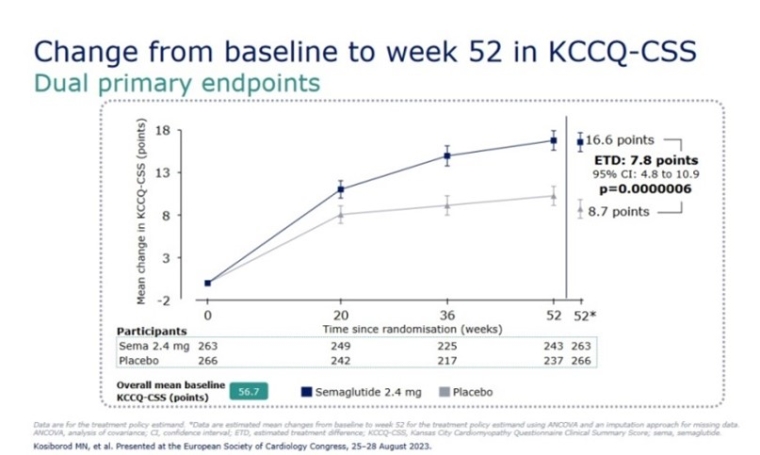
- The STEP-HFpEF Trial: once-weekly semaglutide for patients with HFpEF and obesity.
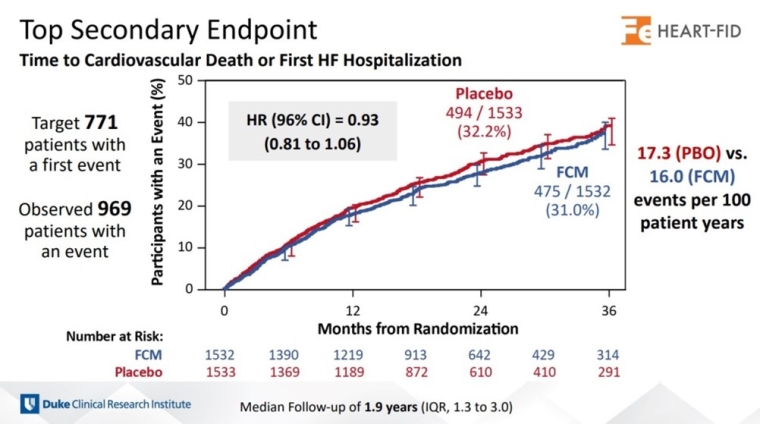
- The HEART-FID Trial: disappointing results from the trial targeting iron deficiency in HF.
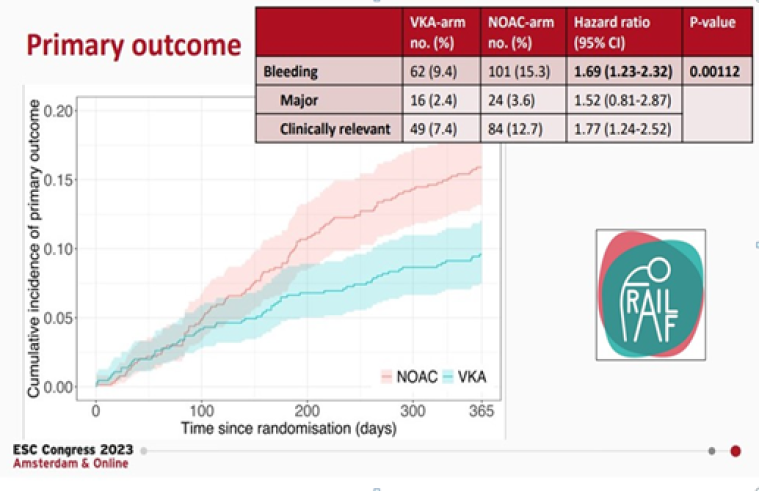
- The FRAIL-AF Trial: your frail patient well tolerates an INR-guided VKA treatment - don't switch to a NOAC!
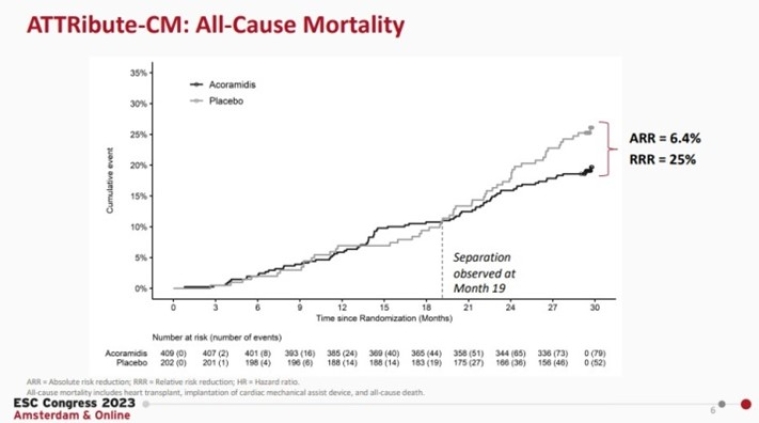
- The ATTRibute-CM Trial: acoramidis shows promising results for transthyretin amyloid cardiomyopathy treatment.
- The NOAH-AFNET 6 Trial: atrial high-rate sub-clinical episodes - oral anticoagulation increases the risk of bleeding without benefit on thrombotic events.
2023 ESC Guidelines for the management of cardiovascular disease in patients with diabetes
The 2023 ESC Guidelines for the management of cardiovascular disease (CVD) in patients with diabetes were released during ESC Congress 2023 in Amsterdam, and are of particular importance for our daily practice.
The first significant change from the previous guidelines, published in 2019, is that prediabetes is not discussed, due to lack of clear evidence.
The second important consideration is that all patients with diabetes should be assessed for the presence of CVD and severe target organ damage, defined by the estimated glomerular filtration rate (eGFR), urinary albumin–to-creatinine ratio (UACR) and the presence of microvascular disease in at least three different sites.
A novel algorithm, SCORE2-Diabetes, is proposed for patients with type 2 diabetes and no atherosclerotic cardiovascular disease (ASCVD) nor target organ damage. It is a 10-year CVD risk score for the classification of patients as low, moderate, high or at very high CV risk.
In patients with diabetes and ASCVD, treatment with sodium-glucose co-transporter-2 inhibitors (SGLT2-i) and glucagon-like peptide-1 receptor agonists (GLP1-RA) is recommended, independent of glucose control and in addition to standard of care.
A systematic search for heart failure (HF) is recommended at each clinical encounter. All diabetic patients with HF, regardless of left ventricular ejection fraction (LVEF), should be treated with SGLT2i to reduce HF hospitalisation and CV death.
Opportunistic screening for atrial fibrillation (AF) by pulse taking or electrocardiogram ECG is recommended in patients with diabetes aged >65 years.
In the presence of chronic kidney disease (CKD), patients should receive SGLT2i and finerenone to reduce CV events and kidney failure risk.
If additional glucose control is needed, metformin should be considered in patients with diabetes mellitus type 2 and ASCVD (Class IIa, Level of Evidence [LoE] C) or pioglitazone (Class IIb, LoE B).
Metformin alone is recommended to be used in low and moderate risk patients.
Metformin in combination with SGLT2I or GLP1RA, or SGLT2I and GLP1RA alone should be used in diabetic patients at high risk (SCORE2-Diabetes >10%).
Only SGLT2I and GLP1RA are recommended for very high-risk patients with diabetes.
Watch the session 2023 ESC Guidelines overview.
(Please note that the ESC will make these resources free of charge for all readers between the 6 October and 1 December 2023. After that time, from 2 December onward, they will only be available for ESC Professional Members.)