One of the main events at the last ESC congress in Paris was the launch of the new ESC Guidelines on Chronic Coronary Syndromes (CCS) [1]. This is a change of title from the last guidelines in 2013 [2] which were called the “Guidelines on stable coronary artery disease”. The new guidelines recognise that coronary artery disease (CAD) may have many different facets and is a dynamic process of plaque accumulation and functional changes of coronary circulation that can be modified by lifestyle changes, pharmacological therapies and revascularisation (Figure 1). To reflect the dynamic nature of the syndrome, the new guidelines have been named Chronic Coronary Syndromes (CCS) as opposed to the Acute Coronary Syndromes (ACS) guidelines.
All Figures are from the current guidelines, courtesy of the ESC and publisher.
Read the full version of the new CCS Guidelines here: https://academic.oup.com/eurheartj/advance-article/doi/10.1093/eurheartj/ehz425/5556137
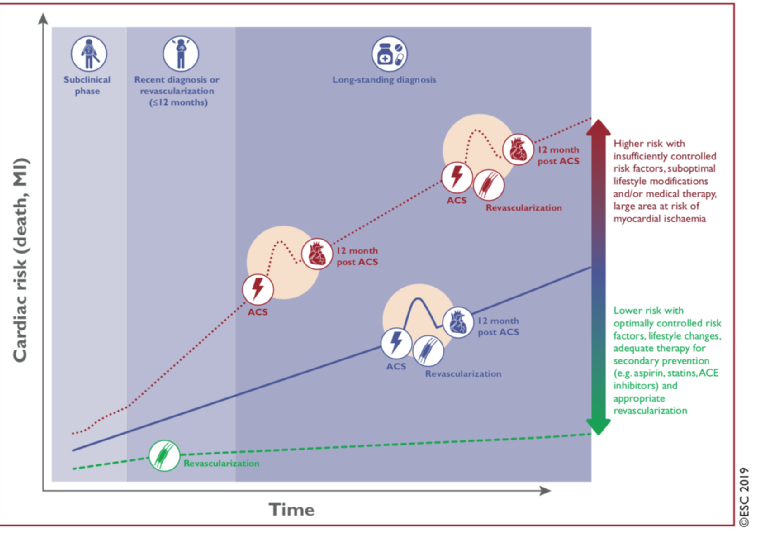
Figure 1. Natural history of chronic coronary syndromes.
With permission of Oxford University Press on behalf of the European Society of Cardiology
From reference [1]. Knuuti J, Wijns W, Saraste A, et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes: The Task Force for the diagnosis and management of chronic coronary syndromes of the European Society of Cardiology (ESC). Eur Heart J. 2019 Aug 31. [Epub ahead of print].
The new Guidelines, well written and clinically oriented, are built around the six major clinical scenarios of CCS:
- Patients with angina and/or dyspnoea, and suspected coronary artery disease.
- Patients with new onset of heart failure or reduced left ventricular function.
- Patients with a long-standing diagnosis of chronic coronary syndrome.
- Patients with angina without obstructive disease in the epicardial coronary arteries.
- Asymptomatic subjects who undergo screening for possible subclinical CAD.
- Chronic coronary disease in special subgroups such as hypertension, valve disease, refractory angina, very old persons and renal failure.
The guidelines underscore, like the previous ones, the importance of a thorough clinical history before further evaluation. They have also kept in the recommendation for an initial echocardiographic examination in all patients (Ia); as well as preferably also carotid ultrasound to detect subclinical atherosclerosis (IIa).
Pre-test probability and clinical likelihood
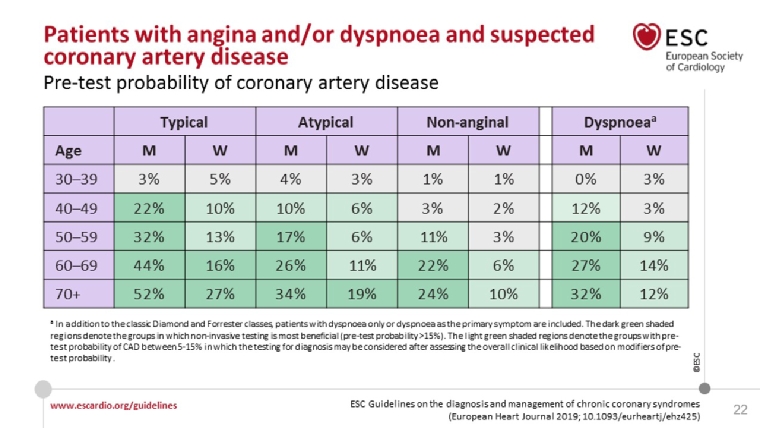
The previous guidelines [2] introduced the concept of pretest probability (PTP) based on variables such as sex, age and angina characteristics. However, recent publications [3] have shown that the probability figures in the previous GUIDELINES were grossly overestimated. The new GUIDELINES have an updated Table (Figure 2) showing numbers indicating probability of significant CAD which are around 1/3 of the previous Table.
Figure 2. Updated Pre-test probability (PTP) of obstructive coronary artery disease based on sex, age and angina characteristics.
With permission of Oxford University Press on behalf of the European Society of Cardiology
From reference [1], Table 5. Knuuti J, Wijns W, Saraste A, et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes: The Task Force for the diagnosis and management of chronic coronary syndromes of the European Society of Cardiology (ESC). Eur Heart J. 2019 Aug 31. [Epub ahead of print].
Thus, a 38-year-old man with atypical angina has only a 4% PTP of having obstructive CAD and further diagnostic testing - besides a good history, basal blood tests, an ECG and possibly an echo exam - is not necessary. Some CAD patients have dyspnoea as the only symptom and in this new version of the Table, there is a new column for this group.
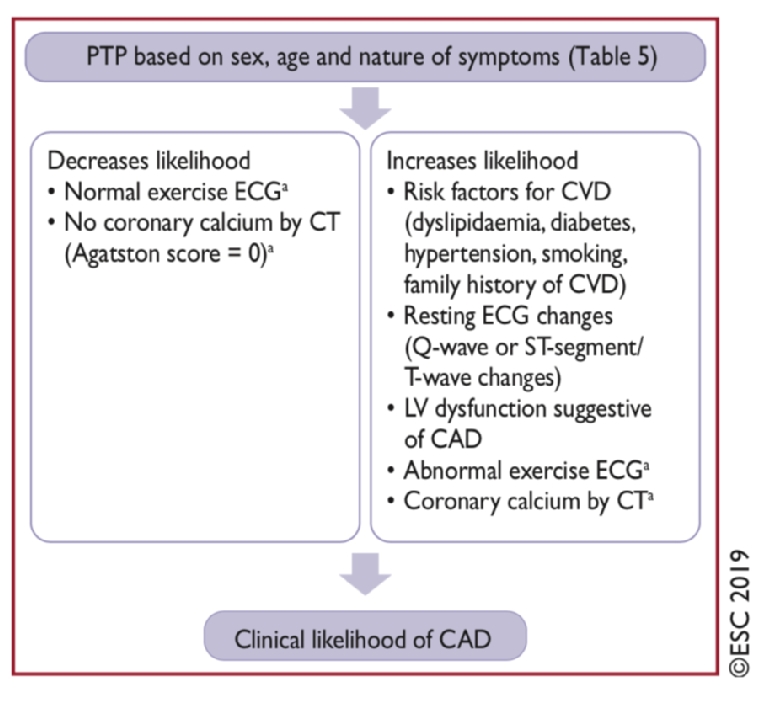
This pre-test probability can be further modified by applying other known clinical risk markers such as smoking and lipid status, ECG changes, coronary CT, carotid ultrasound, etc., and can be correspondingly diminished or augmented to give a clinical likelihood ratio (Figure 3) for obstructive CAD.
This likelihood ratio is then further applied to the various diagnostic tests to rule in or rule out obstructive CAD.
Figure 3. Introducing the clinical likelihood ratio in the diagnostic sequence.
With permission of Oxford University Press on behalf of the European Society of Cardiology
From reference [1]. Knuuti J, Wijns W, Saraste A, et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes: The Task Force for the diagnosis and management of chronic coronary syndromes of the European Society of Cardiology (ESC). Eur Heart J. 2019 Aug 31. [Epub ahead of print].
Which tests offer the best and worst performance?
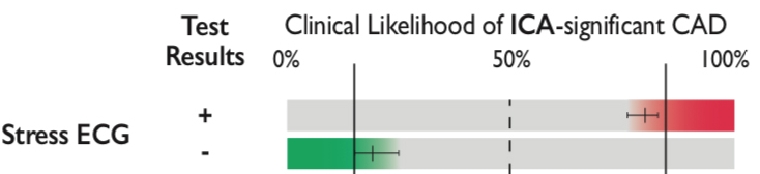
The traditional exercise electrocardiogram (ECG) test was kept in the diagnostic algorithm in the previous GUIDELINES; however, it is now no longer among the tests recommended for the diagnosis of obstructive CAD. The diagnostic performance of the traditional exercise ECG is so poor that it can only reliably rule out CAD in patients with very low PTP and rule in CAD in patients with very high PTP. Thus, an exercise ECG is not generally recommended in the clinical diagnostic workout (IIb) provided that other and more precise diagnostic methods are available, keeping in mind the very high risk of false-negative and false-positive test results. Accordingly, exercise ECG has been downgraded to a class IIb recommendation with respect to CAD diagnosis.
Figure 4. Demonstrating the poor performance of exercise ECG for the exclusion or confirmation of significant CAD based on the clinical likelihood ratio.
With permission of Oxford University Press on behalf of the European Society of Cardiology
From reference [1]. Knuuti J, Wijns W, Saraste A, et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes: The Task Force for the diagnosis and management of chronic coronary syndromes of the European Society of Cardiology (ESC). Eur Heart J. 2019 Aug 31. [Epub ahead of print].
Still, the guidelines recognise that an exercise ECG provides a lot of other valuable information such as exercise tolerance and capacity, which has a strong impact on long-term prognosis, heart rate and blood pressure response and information regarding exercise-induced arrhythmias in selected patients. Thus, the time has not come to throw the ergometer or the treadmill away. However, assessing a patient with suspected angina purely by exercise testing is not recommended.
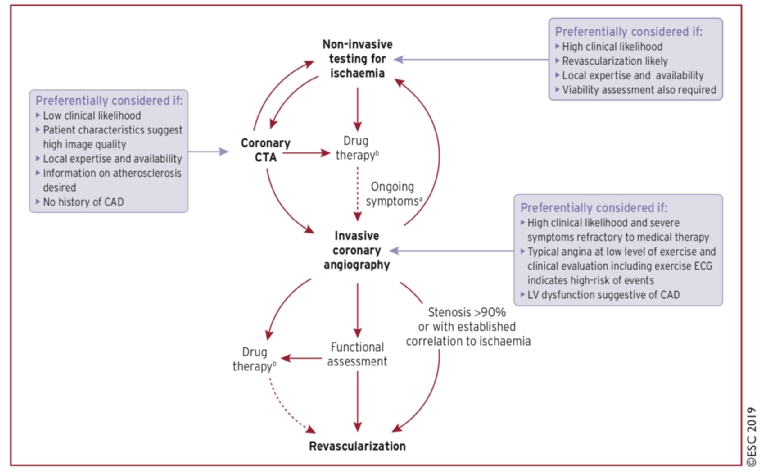
Computed tomography (CT) angiography (CTA) is now the recommended initial test for patients with a low to moderate clinical likelihood of CAD. Patients with a moderate to high likelihood should be triaged to a non-invasive test for ischaemia such as stress echo, single photon emission computed tomography (SPECT), stress magnetic resonance imaging (MRI) perfusion or positron emission tomography (PET), or directly to invasive angiography in patients with a very high likelihood.
Figure 5. Illustration of the recommended diagnostic algorithm with coronary CTA in a central position.
With permission of Oxford University Press on behalf of the European Society of Cardiology
From reference [1]. Knuuti J, Wijns W, Saraste A, et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes: The Task Force for the diagnosis and management of chronic coronary syndromes of the European Society of Cardiology (ESC). Eur Heart J. 2019 Aug 31. [Epub ahead of print].
Assessment of risk in secondary prevention
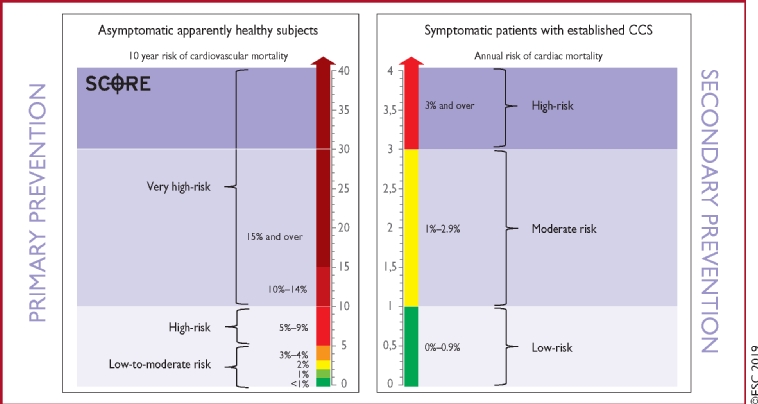
The guidelines recommend that every patient with suspected or documented chronic stable coronary artery disease should undergo an assessment of the future risk of death or a cardiac event. They define low risk as a yearly mortality rate of below 1% and high risk as a yearly mortality rate of above 3%.
Figure 6. Risk classes in stable CAD.
With permission of Oxford University Press on behalf of the European Society of Cardiology
From reference [1]. Knuuti J, Wijns W, Saraste A, et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes: The Task Force for the diagnosis and management of chronic coronary syndromes of the European Society of Cardiology (ESC). Eur Heart J. 2019 Aug 31. [Epub ahead of print].
The left ventricular (LV) function is recognised as the strongest predictor of long-term survival and a patient with an LVEF <50% is already at high risk of cardiovascular death (annual mortality rate >3%), even without accounting for additional event risk factors such as those outlined in Figure 7.
Figure 7 & 8. Recommendations for risk assessment.
With permission of Oxford University Press on behalf of the European Society of Cardiology
From reference [1]. Knuuti J, Wijns W, Saraste A, et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes: The Task Force for the diagnosis and management of chronic coronary syndromes of the European Society of Cardiology (ESC). Eur Heart J. 2019 Aug 31. [Epub ahead of print].
The various components are discussed in more detail in the online supplementary data [1]. Ejection fraction (EF) is important and should be assessed in every patient with suspected or established CAD suspected; however, 50% of deaths in stable CAD patients are sudden and most of these have an EF of >50% [1]. One of the new recommendations is the global longitudinal strain (GLS) and this has been included in the guidelines with a class IIb recommendation. A GLS which is reduced by >2 standard deviation (SD) from the lower normal reference value gives an incremental value in the risk assessment of patients with CCS, particularly in those with an EF >35% [4].
Pharmacological treatment
There have been no landmark trials necessitating a major change regarding the drugs recommended for angina treatment since the 2013 guidelines. The class Ia recommendation is still to use beta-blockers and/or calcium channel blockers to control heart rate and symptoms with long-acting nitrates as the first second line addition. Newer second-line drugs such as nicorandil, ranolazine, ivabradine and trimetazidine, have class IIa recommendations when symptoms are not adequately controlled by first-line drugs and nitrates; these are discussed in detail in the supplement paper [1].
Regarding medication for event prevention, 75-100 mg aspirin is recommended (class Ia) life-long in patients with a previous myocardial infarction (MI) or revascularisation with 75 mg clopidogrel as an alternative. For patients without an MI or previous revascularisation, aspirin only has a class IIb recommendation.
Adding a long-term second antithrombotic drug to aspirin should be considered in those with a high risk of ischaemic events and a low bleeding risk and may be considered in those with a moderately increased risk of ischaemic events. The options are clopidogrel, prasugrel, ticagrelor or low-dose rivaroxaban. In highly selected patients with AF and a history of MI and a high risk of recurrent ischaemic events, antiplatelet therapy in addition to oral anticoagulation may be considered. Regarding dual antiplatelet therapy, the recommendations are in line with the recent ESC focused update [5].
Lifestyle modifications and event preventing therapy
Emphasis is placed on the various lifestyle modifications as outlined in the recent lipids [6] and preventive [7] guidelines. Regarding low-density lipoprotein (LDL) targets, it is surprising that these new stable CAD guidelines continue with an LDL target of <1.8 mmol/l (<70 mg/dL) or at least 50% reduction, while the lipid guidelines [6] now recommend secondary prevention in very high-risk patients, an low-density lipoprotein cholesterol (LDL-C) goal of <1.4 mmol/L (<55 mg/dL); for those with a second coronary event within two years while on maximally tolerated statin-based therapy, a target of <1 mmol/l may be considered.
Angiotensin-converting enzyme (ACE) inhibitors are recommended for those with other indications and should otherwise be considered in CCS patients at very high risk of cardiovascular events. Likewise, beta-blockers are recommended in heart failure (besides as an anti-anginal therapy) and should be considered in those with previous ST-elevation myocardial infarction (STEMI).
Revascularisation
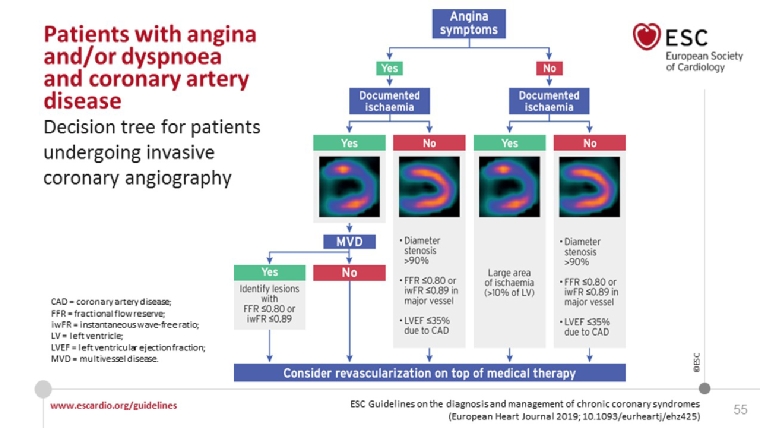
Revascularisation by percutaneous coronary intervention (PCI) or coronary artery bypass grafting (CABG) has previously mainly been regarded as a symptomatic treatment unless there is reduced LV function and/or severe proximal CAD. This is discussed in detail in the recent guidelines on myocardial revascularisation [8]. The new CCS guidelines place emphasis on several recent studies indicating a prognostic benefit of revascularisation and they promote a less restrictive indication for revascularisation in CCS, looking at the specific anatomy [e.g., left main (LM)] or extended ischaemia (>10%) and using physiologic measures of stenosis significance such as fractional flow reserve (FFR). However, the results of the recent ISCHEMIA trial [9] suggest that an optimal medical management in CAD is still a successful road to pursue, and that revascularisation should be clinically and symptomatically guided (https://www.ischemiatrial.org). A practical approach is given in Figure 9 which also underscores the importance of angina.
Figure 9. A practical approach to revascularisation in CCS.
With permission of Oxford University Press on behalf of the European Society of Cardiology
From reference [1]. Knuuti J, Wijns W, Saraste A, et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes: The Task Force for the diagnosis and management of chronic coronary syndromes of the European Society of Cardiology (ESC). Eur Heart J. 2019 Aug 31. [Epub ahead of print].
Angina without coronary stenosis, vasospastic angina, microvascular angina and chronic refractory angina
The diagnostic and therapeutic challenges of these entities are discussed. Coronary flow resistance can be evaluated by transthoracic echo looking at left anterior descending (LAD) artery flow, by MRI perfusion or by PET scan, while arteriolar dysregulation requires assessment of endothelial function with intracoronary acetylcholine infusion; treatment approaches are outlined. For refractory angina, several of the non-pharmacological treatments have now been included in the new guidelines with class IIb recommendations: enhanced external counter pulsation, coronary sinus constriction and spinal cord stimulation. However, based on the available data, autologous cell therapy is not recommended.
Long-term follow-up of patients with CCS
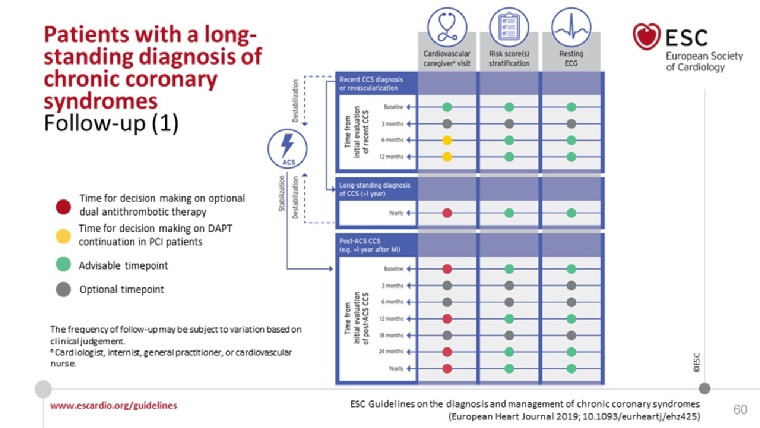
This is a new chapter of special interest to the office-based practicing cardiologist. An annual evaluation by a cardiovascular practitioner is warranted, even if the patient is asymptomatic. It may be beneficial to evaluate LV function and test for ischaemia every 3-5 years, even in asymptomatic patients. The recommendations are nicely illustrated in nice figures (Figure 10 and 11)
Figures 10 & 11. Recommendations for long-term follow-up in CCS.
With permission of Oxford University Press on behalf of the European Society of Cardiology
From reference [1]. Knuuti J, Wijns W, Saraste A, et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes: The Task Force for the diagnosis and management of chronic coronary syndromes of the European Society of Cardiology (ESC). Eur Heart J. 2019 Aug 31. [Epub ahead of print].