Remote Ischemic Conditioning in Lymphoma Patients Receiving Anthracyclines (RESILIENCE)
The project

This will also allow the consortium to study gender differences in anthracyclines-induced heart failure. A phase II randomised clinical trial enrolling 608 patients undergoing anthracyclines chemotherapy will be done. The primary endpoint will be based on serial cardiac magnetic resonances exams. Taking advantage of the recruited population and data gathered, the project will further validate 2 novel cardiac magnetic resonance imaging methods: a novel early marker of cardiotoxicity, and a new sequence allowing a massive reduction of acquisition time.
The consortium will also study a personalised strategy to empower patients in clinical trial execution, which includes Patient-Reported Outcome and Experience Measures (PROMs and PREMs). The final goal is to reach the patient level by implementing the novel strategy at the clinical level while paving the way for a future large phase III trial.
RESILIENCE consortium is a multidisciplinary group, including experts in cardiology, haematology, cardio-oncology, and medical imaging, together with nurses and patients’ associations. The consortium is composed of 11 partners, coordinated by Centro Nacional De Investigaciones Cardiovasculares Carlos III (CNIC) and the Lymphoma Coalition Europe. The ESC Council of Cardio-Oncology will be involved in the dissemination and exploitation of results.
Work packages
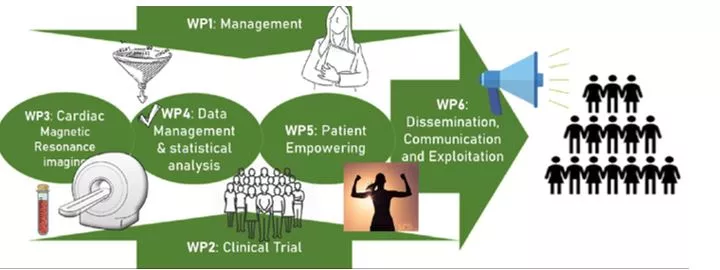
RESILIENCE is a five-year project organised around six distinct and closely related work packages (WPs):
- WP3: Cardiac Magnetic Resonance Imaging
- WP4: Data Management and statistical analysis
- WP5: Patient empowering.
These form a sound base for conducting the central RESILIENCE work package:
- WP2: Clinical trial
- WP1: Management - ensures the right project management and governance whereas
- WP6: Dissemination, communication and exploitations - provides a strategy for the communication and dissemination activities as well as effective exploitation of the project’s results.
Resources
- Press release: Kick-off meeting press release
- Website: the website of the project is under preparation.
Partners in the RESILIENCE Consortium
- CENTRO NACIONAL DE INVESTIGACIONES CARDIOVASCULARES CARLOS III” (CNIC)
- AMSTERDAM UMC
- EUROPEAN SOCIETY OF CARDIOLOGY
- INSTITUTO INVESTIGACION SANITARIA FUNDACION JIMENEZ DIAZ
- AARHUS UNIVERSITETSHOSPITAL
- INSTITUTO PORTUGUES DE ONCOLOGIA DE LISBOA FRANCISCO GENTIL EPE
- PHILIPS MEDICAL SYSTEMS NEDERLAND BV
- HEINRICH-HEINE-UNIVERSITAET DUESSELDORF
- CENTRE REGIONAL DE LUTTE CONTRE LE CANCER HENRI BECQUEREL ROUEN
- CONSORCIO CENTRO DE INVESTIGACION BIOMEDICA EN RED M.P.
- HOSPITAL DA LUZ LEARNING HEALTH (GLSMED)
- LYMPHOMA COALITION EUROPE
Funding Acknowledgement
The project has received funding from the European Union's Horizon 2020 Research and Innovation Programme under grant agreement 945118.
Consortium ESC Council of Cardio-Oncology

Doctor Riccardo Asteggiano
Past-Chairperson of the ESC Council of Cardio-Oncology Principal Investigator

Doctor Teresa Lopez Fernandez
Secretary of the ESC Council of Cardio-Oncology Co-Principal Investigator