While clear criteria for diagnosis have been recently established and a standardised guideline-based approach is increasingly being used, the results of therapy management for syncope in clinical practice, despite advances in recent years, are largely unknown and are still not satisfactory. Syncope recurrence rates remain high after diagnostic evaluation and new validated therapeutical strategies are warranted.
The status quo
Syncope recurrence rates remain high despite modern treatment in population studies. In the recent Evaluation of Guidelines in Syncope Study 2 (EGSYS 2) follow-up study (1), diagnostic evaluation was standardised and followed the established algorithm of the guidelines of the European Society of Cardiology (2), but the decision regarding whether and how to treat patients was left to the clinical practice of the individual physicians.
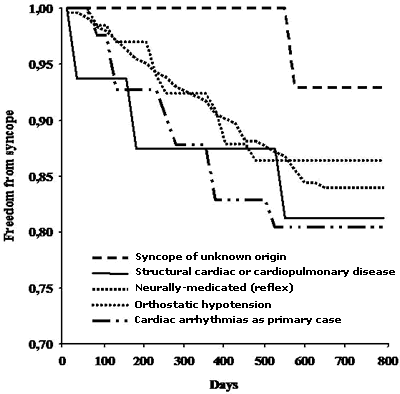
Treatment therapy as prescribed at the end of the diagnostic evaluation period are listed in Table 1. Syncope recurred in 16.5 % of patients during a mean of 614 days. The incidence of syncope recurrence was similar irrespective of the mechanism (Figure 1). Syncope rate was 12.5 per 100 patient-years in patients with syncope due to primary cardiac arrhythmia; among these, the rate was 9.1 per 100 patient years in those who received some specific treatment (pace-maker, ablation or ICD) and 20.0 per 100 patient years in those who did not (p=0.28). The recurrence rate was 14.9 per 100 patient years in patients with structural cardiac (or cardiopulmonary) syncope, 9.8 per 100 patient years in patients with neurally-mediated syncope, 8.8 per 100 patient years in patients with orthostatic syncope and 4.1 per 100 patient years in patients with unexplained syncope.
To put these data into perspective, it is noteworthy to note that, in population studies of untreated patients with similar baseline characteristics, a history of 1 or 2 episodes of syncope during life predicted a recurrence of 15% and 20% after 1 and 2 years respectively, whereas a history of 3 episodes of syncope during life predicted a recurrence of 36% and 42% after 1 and 2 years respectively (3).
In brief, the outcome of treated patients does not appear to be much better than untreated patients and syncope recurred in a substantial number of patients, even in those who received specific treatment (i.e., cardiac pacing or ICD). Moreover, recurrence rates of the Evaluation of Guidelines in Syncope Study 2 were not much better than those observed in studies performed several years ago, when many modern treatments were unknown or underused. For example, in prospective studies of patients with syncope referred to the Emergency Room, ambulatory clinics or admitted into the hospital, Kapoor observed a 20% recurrence rate in 1 year (4,5).
Conclusion and perspective
The above data show that abolition of syncopal recurrence is very difficult to achieve in the real world even with mechanism-specific treatments. There are multiple reasons that may explain recurrences: misdiagnoses, inappropriate indications, inefficacy of some proposed therapies, lack of therapy for some forms of syncope, multifactorial mechanism of syncope, etc. What can we do to improve outcomes? Some of the above factors are obviously not modifiable or difficult to improve.
The most efficient solution most likey is to adopt strict standardised criteria for treatment administration based on the recommendations contained in the guidelines. Results should be verified with large prospective registries. While randomised controlled trials are the strongest tools for demonstrating the efficacy of new therapies, registries seem to be more appropriate for the evaluation of effectiveness of management strategies. Should careful application of this evidence-based strategy for therapy be carried out, it is unlikely that complete abolition of recurrences would be achieved.. A more reasonable goal would be the reduction in the number and severity of recurrences in order to limit the risk of secondary trauma and improve quality of life. Thus, new trials should have as primary end-point the reduction in the total burden of syncope and related trauma rather than time to first syncopal recurrence.
Secondly, we must recognise that, typically patients are asymptomatic at the time of evaluation and the opportunity to capture a spontaneous event during diagnostic testing is rare. Even when they are guideline-based, most diagnoses are presumptive as they are based on laboratory test results and physiological states. Knowledge of what occurs during a spontaneous syncopal episode is ideally the gold standard for syncope evaluation and treatment. For this reason, it is likely that prolonged ECG monitoring (external and implantable), will become increasingly important in the assessment of the syncope patient. Initial results are promising. In a randomised prospective population study (6) patients who had received a therapy guided by documentation of syncope by means of an implantable loop recorder had a significantly lower recurrence rate than controls. The ISSUE 2 study (7), of the 102 patients with a symptom-rhythm correlation, 53 underwent loop recorder guided therapy, predominantly pacing for asystole. These patients experienced a striking reduction in recurrence of syncope compared with non-loop recorder guided therapy (10% vs 41%, p=0.002). However, whether a strategy of extensive utilisation of prolonged ECG monitoring will be able to substantially improve the outcome of patients in clinical practice is as well a matter of future researches.
Table 1: Prescribed treatment and measures in a population observational cohort
| Neurally-mediated (reflex) *- n | 243 |
| Education, reassurance & avoidance of triggers alone– n (%) | 190 (78) |
| Modification or discontinuation of hypotensive drugs – n (%) | 27 (11) |
| Physical manœuvres (tilt training or counterpressure manœuvres) – n (%) | 5 (2) |
| Vasoconstrictor drugs – n (%) | 2 (1) |
| Pacemaker – n (%) | 11 (5) |
| Orthostatic hypotension *- n | 66 |
| Education & avoidance triggers – n (%) | 30 (45) |
| Modification or discontinuation of hypotensive drugs – n (%) | 25 (40) |
| Physical manœuvres (counterpressure manœuvres, elastic stockings) – n (%) | 2 (3) |
| Volume expansion – n (%) | 9 (14) |
| Vasoconstrictor drugs – n (%) | 2 (3) |
| Cardiac Arrhythmias as primary cause *- n | 41 |
| Cardiac pacing – n (%) | 27 (66) |
| Antiarrhythmic drug therapy – n (%) | 8 (20) |
| Cardioverter-defibrillator implant – n (%) | 2 (5) |
| Catheter ablation – n (%) | 2 (5) |
| Structural cardiac or cardiopulmonary disease - n | 16 |
| Coronary revascularization (angioplasty, by-pass) – n (%) | 5 (31) |
| Valvular surgery – n (%) | 2 (12) |
| Drug therapy – n (%) | 5 (31) |
| Syncope of unknown origin – n | 14 |
| No specific therapy – no (%) | 3 (21) |
* More than 1 treatment was assigned to some patients of this group
Figure 1: Actual freedom from syncope recurrence curves in the different syncope forms (p=ns).



 Our mission: To reduce the burden of cardiovascular disease.
Our mission: To reduce the burden of cardiovascular disease.