Singe Photon Emission Computed Tomography (SPECT) allows the simultaneous assessment of blood flow distribution and wall motion, but many of the problems that interfere with optimal image quality have not yet been resolved. The application of attenuation correction is suboptimal and the ambitious target of absolute blood flow is still within the PET (Positron emission tomography) domain - thus still far from clinical cardiology.
Nevertheless, the recent introduction of Cadmium Zinc Telluride (CZT) detectors have enhanced the diagnostic spectrum of SPECT via a better temporal and spatial resolution, and have allowed an important reduction in both dose and scan time - by 50-75%. Also, the CZT technique has the theoretical “imaging power” needed for the measurement of absolute blood flow.
1 - Background
Anger camera imaging (1) has been the only system available in Nuclear Medicine for the last 40 years despite important advances in technology in general. Spatial resolution is one of the camera’s limitations. Its hampered imaging power effects sophisticated protocol results such as the Quantitative Perfusion Score, which is routinely used to assist with decision making in patients with coronary artery disease. Collimators used in Anger cameras enhance spatial resolution but reduce count rates. This poor resolution does not fully reflect the true myocardial thickening and degrades image quality, introducing attenuation artifacts especially in the inferior and anterior walls.
Similarly, a temporal resolution of minutes limits all those quantitative approaches that are possible with PET, and thus making the PET measurement of MBF a standard in cardiology. The relatively recent introduction of attenuation correction obtained with hybrid systems is not widespread enough among nuclear cardiology specialists (2). Strong evidence in favour of artifacts using gated SPECT, and a heterogeneity of application among manufacturers are the reasons for which the technique has not made itself available enough. Thus, most of the limitations of the Anger camera persist with intrinsic conflicts between resolution and sensitivity.
Furthermore, image quality requires high doses of radiotracers and prolonged imaging times when compared to alternative techniques. SPECT offers the best compromise of cost-benefit ratio, and provides simultaneous information on perfusion and function, but imprerfect spatial and temporal limitations have prevented SPECT to spread in cardiology in favour of alternative techniques.
2 - High-Speed SPECT
An enterily new technology has been developed for high-speed SPECT which uses a bank of independently controlled detectors with large-hole tungsten collimators and multiple cadmium zinc telluride crystal arrays. Compared to the standard gamma camera, this system provides an 8-fold increase in count rates, thereby reducing imaging times significantly, while achieving a 2-fold increase in spatial resolution as well(3), thus enabling high quality scans (Fig. 1) with a significant reduction in imaging time and radioisotopes doses.
Recently, Sharir et al, (4) compared a high-speed SPECT system with a standard Anger camera in a series of patients undergoing a Technetium-99m sestamibi rest/stress protocol. Although imaging times decreased 4-fold for stress and 6-fold for rest acquisitions, high-speed SPECT provided images that were comparable or better in resolution to those achieved with conventional SPECT. In this study, the overall diagnostic interpretation with qualitative and semi-quantitative analyses was the same.
3 - The High-Speed SPECT revolution
The use of high-speed SPECT has revolutionarised instrumentation, with unique characteristics that have improved both resolution and sensitivity when compared to the latest Anger cameras. But will high-speed SPECT revolutionarise the field of nuclear cardiology? It is quite possible, but beyond these initial observations more work is needed to enter into this of High-speed imaging.
The cardiological impact will be powerful from several points of view:
- Patients will benefit from a significant dose reduction. Dosimetry is increasingly important in the choice of cardiac imaging technique and high-speed SPECT offers a 4-fold decrease for acquisition time that can be translated to dose. Looking for a compromise, a high-speed SPECT could be completed in 50% of the time with a 50% decrease in the dosage DOSE used in standard spect
- Higher resolution images than those achieved with Anger cameras, allow an exact identification of ischemia and scar with more transmural resolution than previous SPECT systems, and hopefully in the future with a more accurate identification of multivessel coronary artery disease;
- The improved resolutions will reduce the occurrence of motion and attenuation artifacts;
- Smaller and faster cameras will offer the advantage of increased patient throughput that will reduce costs and waiting lists. Such considerations could have a strong impact on future appropriateness of high-speed SPECT thereby determining a new usefulness and clinical value
4 - The revolution inside the revolution: toward myocardial blood flow
Since SPECT is the most wide-spread stress imaging technique in cardiology, research should focus on the recent development of high-speed technology in order to obtain a “cheap” tool to investigate myocardial blood flow. In fact, the quantitative assessment of coronary flow reserve would be useful for the functional evaluation of coronary stenosis, for tracking the effects of medical treatment or revascularization and to follow-up on the progression of the disease. High-speed SPECT would represent the best solution for its physical characteristics of temporal and spatial resolution and efficiency.
Preliminary investigation should be targeted at patients to define the real possibility to be a full PET alternative in all its aspects, with the added value of standard volume quantification and regional wall motion already available with gated SPECT, and with the limits of available 99mTc tracers that exhibit a plateau in myocardial uptake at high flow rates.
5 - Quantitative evaluation of myocardial blood flow and coronary reserve
The quantitative assessment of coronary flow reserve could be useful for the cardiologist in many clinical conditions. In patients with coronary artery disease the measurement of coronary reserve could enhance the detection of stenoses in patients with balanced as well as unbalanced multivessel disease. In these patients each coronary territory could be characterised as an absolute value, leading to a more accurate characterisation of flow reserve with high-speed SPECT similarly to cardiac PET imaging with a lower intra and interobserver variability. In patients with intermediate lesions coronary reserve rather than percent stenosis could be used to assist in decision making, e.g. medical treatment or revascularisation.
In all these conditions a SPECT marker of absolute flow could be used to verify the effect of treatment and to follow-up the progression of the disease.
Finally, the limitations of all “relative” stress imaging techniques such as standard SPECT, stress echo and MRI suggest that quantitative measurements of coronary reserve could enhance patients’ care, especially in the field of asymptomatic subjects with multiple cardiac risk factors, hypertension or diabetes in whom coronary vasodilator capacity has been shown to be abnormal even in absence of coronary lesions. In these patients an invasive assessment of coronary reserve is contraindicated, the available non-invasive tools show a drop in specificity and the use of PET results prove to be too expensive for a routine application.
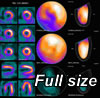
Figure 1. High speed Gated SPECT
Obtained in a 56 years old male patient with a clinical history of hypertension, dyslipidemia and angina on effort. Diagnosis is multivessel coronary artery disease with normal wall motion, EF=50%, EDV 126 ml. Scan time was 4 minutes at rest and 2 at stress, cumulative dose 1480 MBq. Note the clear-cut identification of reversible perfusion defects in the vascular beds of left anterior descending and right coronary arteries.
Conclusion:
Whether high-speed SPECT will deeply revolutionarise cardiology, and in a cost-effective manner, will only be decided with studies performed in significant groups of patients with a known coronary anatomy and in comparison with PET. However, it certainly has the potential to do so.
The content of this article reflects the personal opinion of the author/s and is not necessarily the official position of the European Society of Cardiology


 Our mission: To reduce the burden of cardiovascular disease.
Our mission: To reduce the burden of cardiovascular disease.