N.J. Samani – Genome-wide association analysis of coronary artery disease
Samani presented the results of a British/German genome-wide association analysis of coronary artery disease (CAD). Until today the strong influence of genetic aspects on the development of CAD has been extensively described; however, the genetic basis remained insufficiently defined.
New genetic techniques allowing the analysis of hundreds of thousands of genetic polymorphisms in a systematic, non-biased approach, promise new insights into complex inherited diseases.
In this study, variations in 500,568 Single Nucleotide Polymorphisms (SNPs) were determined in the Wellcome Trust Case Consortium Study (1,926 cases with premature CAD and 2,938 controls) and the German MI Family Study (875 cases and 1,644 controls). These SNPs represent small genetic changes or variations that occur within a person's DNA sequence. SNPs occur more than 1 % of the time but in only 3-5 % of a person's DNA sequence codes for the production of proteins. Therefore, most SNPs are found outside of the "coding sequences". SNPs found within the coding sequence are of particular interest because they are likely to alter the biological function of a protein. Interestingly, in both study populations, the chromosome 9p21.3 (which means chromosome 9, position p (p for petit= the chromosome's short arm), position on the arm: band 21, sub-band 3) demonstrated the strongest signal in SNP analysis. Approximately 22% of the studied population was homozygeous for the allele with an additional 50% carrying a single copy. 6 more loci, with highly significant associations with CAD were identified in both studies including 1p13.3, 6q25.1, 10q11.21, 2q36.3, 15q22.33, and 1q41.
The aggregate attributable risk for the loci of 3 chromosomes was nearly 40% in this population of patients with premature CAD and underlines the important clinical relevance of the associations (figure 1).

N. Rizzi – Generation of a mouse model of catecholaminergic polymorphic ventricular tachycardia
Cardiac arrhythmias and sudden death have been attributed to genetic variations. The autosomal recessive variant of catecholaminergic polymorphic ventricular tachycardia (CPVT) is characterised by increased susceptibility to arrhythmias caused by mutations in the Calsequestrin (CASQ2) gene. CASQ2 buffers Ca2+ of the sarcoplasmic reticulum (SR) and determines the functional size and stability of the SR which controls contraction-relaxation cycles. To study the molecular mechanisms linking mutant CASQ2 to clinical manifestations, a knock-in mouse model was generated carrying a missense mutation (R33Q) previously identified in a patient with severe arrhythmias. Homozygous (Casq2 R33Q/R33Q) mice are viable but display recurrent episodes of ventricular bigemini and spontaneously polymorphic VT in 100%. The Casq2 protein is significant reduced (40%) and "Casq2-like proteins" could not been detected. Like in the wildtype mice, the mutant protein is targeted to the SR. Upon exposure to psychological stress, all Casq R33Q/R33Q mice present sustained bidirectional VTs identical to patients. Heterozygous Casq2 WT/R33Q mice do not develop arrhythmias. However, isolated cardiomyocytes from Casq2 WT/R33Q mice developed delayed after-depolarization (DAD, 18/32 cells; 56%) and triggered beats (9/31; 29%) upon pacing. Epinephrin perfusion increased the development of DADs (15/16; 93%) and triggered beats (8/17; 47%). The generation of such mouse models allows the dissection of the molecular and electrophysiological basics of inherited arrhythmogenic diseases.
G. Pasterkamp – Results from the Athero-Express study
Gene polymorphisms within the coding sequence are likely to alter the biological function of a protein. Therefore, in addition to genomic correlation studies, protein expression analyses are performed to identify proteins involved in atherogenesis.
Endothelial dysfunction and atherosclerotic lesion formation are often followed by plaque rupture with concomitant myocardial ischemia. The mechanisms leading to plaque rupture remain unclear. The Athero-Express study aims to investigate the role of plaque characteristics and future cardiovascular events.
In two Dutch hospitals, all patients undergoing carotid endarterectomy were included into the study and followed for cardiovascular events. 800 patients were enrolled in this ongoing study and 650 underwent follow-up for 1 to 3 years. 23% (153 patients) suffered from a combined endpoint consisting of myocardial infarction, revascularisation, cardiovascular death, stroke, cerebral bleeding, aneurysm surgery, and peripheral artery surgery. 667 plaque specimens were analysed for cellular content, calcification, and thrombus formation. Only the presence of thrombus formation correlated with cardiovascular outcome. An unbiased approach was performed using Proteomic analysis in pooled patients with cardiovascular events and matched controls. 90 proteins were identified which differed between the two groups. One protein identified is osteopontin. Event rate in the highest quartile of osteopontin expression within the plaque was approximately 40%. Further proteins have not been validated yet.
Again, clinical relevance and mechanistical insights remain to be elucidated. However, this proteomic approach may be an effective method to identify proteins involved in plaque rupture and will allow us to learn more about mechanistics of plaque rupture.
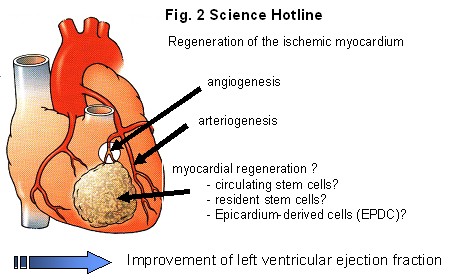
E.M. Winter – Epicardium-derived cells in myocardial regeneration
Myocardial infarction is an important endpoint of atherosclerotic disease and associated with increased morbidity and mortality especially in patients with impaired ventricular function post-AMI.
Currently, no therapeutic strategy is available to replace dysfunctional myocardium or even scar tissue.
Various studies in the past and at the ESC 2007 have addressed this issue and evaluated different stem-cell based strategies after AMI and in ischemic heart disease. However, the identification of the optimal stem cell for tissue regeneration is ongoing.
Embryonic epicardium-derived cells (EPDCs) may be a new and attractive cell source for regeneration of myocardium. EPDCs are crucial for cardiogenesis and seem to be most important for coronary vessel formation and the generation of the compact myocardium. The effect of transplanted EPDCs isolated from human adult atrium and transduced with an eGFP lentiviral vector or vehicle in ischemic myocardium after permanent LAD ligation was evaluated. At week 6 ejection fraction and stroke volume was significantly higher in the EPDC group compared to the control group (EPDC: 34.0±2.5% vs control: 17.8±2.3%, p<0.001 and 27.7±1.3 μl vs 22.5±1.3 μl, p=0.008). End-systolic volume and end-diastolic volume were significantly smaller in the EPDC group. Histological evaluation showed no differences in scar composition but wall thickness of the infarcted area was higher in the EPDC group. Only a few eGFP positive EPDCs were found in the border zones of the infarct. Data suggest that EPDC may be a source for regenerating cells (figure 2). However, as in most published studies of stem cell research, tracking and fate of cells remain difficult. The obtained data suggest like many other studies that mostly paracrine effects of stem cells account for the improvements in ventricular function. The focus of future research should be the evaluation of these mechanisms.
M. Hermann – CYP4A polymorphism correlates with coronary endothelial function
Endothelial dysfunction is one of the earliest pathomorphological correlates for atherosclerotic disease and serves as a risk predictor for cardiovascular events. On a molecular basis, endothelial dysfunction is characterised by reduced nitric oxide bioavailability.
Furthermore, cytochrome P450 (CYP450) metabolizes arachidonic acid into vasoactive epoxyeicosatrienoic acid (EET) and 20-hydroxyeicosatetraenoic acid (20-HETE), substances which are involved in the regulation of the vascular tone.
Various polymorphisms involved in CYP pathways and their correlation with coronary endothelial function were analyzed in 758 patients of the ENCORE 1 and 2 trials (Evaluation of Nifedipine and Cerivastatin on the Recovery of Endothelial Function).
Only variations in CYPA4 turned out to be associated with impaired coronary endothelial function. In 2.04% of patients (n=15), a substitution of phenylalanine 434 by serine (434SS) in CYP 4A11F434 was significantly associated with augmented endothelial dependent vasorelaxation compared to patients with 434FS (n=193, 26.29%) and 434FF (n=526, 71.66%).
Interestingly, blood pressure was significantly higher in 434SS patients while creatinine and creatinine clearance was higher in the 434FF group. These correlation analyses do not provide insights in the underlying mechanisms of disease development.
The results underline the problems of a candidate-based genetic approach (more than 10 candidates were analyzed but only one turned out to be of potential relevance) compared to systematically unbiased approaches.
C. Parsai – Toward understanding response to cardiac resynchronisation therapy: LV dyssynchrony is only one mechanism among multiple
The final endpoint of the cardiovascular continuum is congestive heart failure. Several drug- and deviced-based therapeutic strategies have improved cardiovascular morbidity and mortality. However, these therapies are expensive and not all patients show equal improvement of clinical and functional parameters. Effective strategies are warranted to identify predictors for effective response to Cardiac Resynchronisation Therapy (CRT).
The PROSPECT study has addressed this problem prospectively in a large study and is discussed in the “Clinical Trial Update Sessions”. In another prospective, multi-center study, mechanisms predictive of a response in patients undergoing CRT for standard clinical indications were evaluated. 88 patients recruited from the UK and France (65±1y, EF 25±1%, QRS>120ms) were investigated before and 5±1 months after CRT. Endpoints included LV-remodeling (end-systolic volume reduction ≥10%) and a reduction in NYHA-class ≥1.
Based on ultrasound assessment, five subgroups with different pathological mechanisms were identified: i) Patients with LV dyssynchrony showing a pre-ejection "septal flash" (43 patients, 49%). Eliminating the "septal flash" by CRT was 100% predictive of LV remodelling. ii) Short AV delay with an absent/truncated A-wave (9pts, 10%). Correction of short AV delay was associated with remodelling in 8/9 patients. iii) Long AV delay with E/A fusion +/- diastolic mitral regurgitation (8 patients, 9%). Correction of the AV delay was achieved in 6/8 patients, but AV filling normalised only in 4/8 patients. iv) Exaggerated RV-LV interaction (9pts, 10%). Reduced RV-LV interaction was observed post CRT associated with clinical response and significant fall in PA pressure. v) unknown mechanisms (19pts, 22%). These patients failed to respond to CRT.
The study was able to identify 4 subgroups of patients, each with a different pathophysiological mechanism. An identified mechanism was in most cases associated with a response whereas unknown mechanisms were in 100% associated with a non-response. The study indicates that measurement of systolic LV dyssynchrony alone will miss patients with potential to respond to CRT.
The content of this article reflects the personal opinion of the author/s and is not necessarily the official position of the European Society of Cardiology.

 Our mission: To reduce the burden of cardiovascular disease.
Our mission: To reduce the burden of cardiovascular disease.