Mechanical cardiac asynchrony worsens prognosis and CRT helps to correct it. Electrical asynchrony is not a good response predictor to CRT while the presence of mecanical asyncrony is. However, current guidelines indicate CRT in electrical asynchrony, with no mention of mechanical asynchrony. Mechanical asynchrony can be found with echography at the atrioventricular, interventricular and intraventricular level, but there is no consensus yet on what the best marker for it is, if any. An individualised optimisation of both atrio-ventricular and VV intervals could potentially contribute to increase the benefit obtained from CRT.
Indeed, echocardiography is the preferred technique for studying cardiac mechanical synchrony. In this analysis, the main clinical application is cardiac resynchronisation therapy (CRT). Although current guidelines indicate CRT only if there is electric asynchrony (wide QRS in the ECG), the echocardiographic demonstration of cardiac asynchrony can be useful to identify patients who would also benefit from CRT yet who do not have conductance disturbances as well as to help in individualising CRT device programming.
I - Mechanical cardiac asynchrony : definition
Mechanical cardiac asynchrony is defined as the presence of a delayed contraction of certain myocardial segments. It usually appears at the end stage of a number of different cardiac diseases, together with left ventricular (LV) dilatation and systolic dysfunction.
Mechanical cardiac asynchrony may or may not be associated with electrical asynchrony which is present in up to 30% of patients with heart failure, expressed mainly as a left bundle branch block (LBBB) in the surface ECG. The presence of mechanical intraventricular asynchrony contributes to aggravating LV hemodynamics , favors the apparition of functional mitral regurgitation and consequently, worsens the patient’s prognosis.
II - Mechanical and electrical asynchrony. Helping patient selection for CRT
Due to its huge availability, high temporal resolution and low-cost, echocardiography has been the most widely used imaging technique to study the presence of mechanical asynchrony.
Some studies based on echocardiographic data have shown that up to 30% of patients with a wide QRS (electrical asynchrony) do not have mechanical asynchrony (1) whereas the same proportion - around 30% of patients with narrow QRS-, have mechanical asynchrony (2). This data is interesting, because identifying these patients, - with advanced heart failure and a normal surface ECG- , can extend the indication of CRT seeing that some studies have already demonstrated the benefit of CRT in patients with narrow QRS if mechanical asynchrony can be demonstrated.
Furthermore, most studies agree that the absence of mechanical asynchrony has an important role in a non-response to CRT (3,4) . This data could be useful to improve the selection of patients undergoing CRT if an easy echocardiographic marker to define the presence or absence of mechanical synchrony could be determined, and for this reason, CRT indication continues if electrical asynchrony is present, regardless of the presence or not of mechanical asynchrony.
III - CRT devices programming optimisation
Most CRT trials have shown the benefit of CRT that uses devices with simultaneous biventricular pacing (5), however all of the studies have systematically shown around 30% of non-responders. In order to improve these results, CRT devices are now equipped with sequential pacing capability, in other words, devices that permit the stimulation of both ventricles either simultaneously or sequentially, with a specific delay (VV interval).
Given that one specific electrical pattern does not always entail the same specific mechanical delay, LV stimulation is currently performed from the epicardium while the right ventricle is stimulated from the endocardium, lead positioning may also affect the transmission of the stimulus and the ventricular activation sequence, causing interventricular and intraventricular delays.
It is reasonable to think that an individualised optimisation of both atrio-ventricular and VV intervals would potentially contribute to increasing the benefit obtained from CRT, as some studies have begun to suggest (6). Echocardiography has an important role in this field since it allows for the non-invasive measurement of all these delays thus permitting, at the same time, the evaluation of the acute hemodynamic effect of different programming.
IV - Mechanical asynchrony classes
Mechanical asynchrony can be found at the following three levels: atrioventricular, interventricular and intraventricular level. They can coexist and may be measured by echography.
- Atrioventricular asynchrony
Atrioventricular (AV) asynchrony consists of the decoordination of auricular contraction regarding LV filling due to an electrical conductance disturbance.
It can be echocardiographically evaluated by measuring the LV filling pattern with Pulsed-Wave (PW) Doppler, which obtains the E wave, that represents the passive LV filling, and the A wave, that shows the LV filling due to the atria contraction.
Atrioventricular asynchrony is defined as the presence of a LV diastolic filling time shorter than 40% of the cardiac cycle as shown in Figure 1. The maximal expression of AV asynchrony is the presence of atria fibrillation, when atria contraction, on the other hand very reduced, occurs independently of LV mechanics.
- Interventricular asynchrony
Interventricular asynchrony refers to the decoordination between right and LV contraction. Physiologically LV contraction occurs 10 to 20 ms later than the right ventricle; when this time difference is superior to 40 ms, this indicates interventricular asynchrony.
This delay between the contractions of both ventricles can also be measured with echo by calculating the time difference between both, pulmonary and aortic pre-ejective times, which are again obtained with PW - Doppler applied on the pulmonary and aortic valves as it is shown in Figure 2.
- Intraventricular Asynchrony
The decoordination of the contraction sequence in the different LV segments determines the presence of intraventricular asynchrony, which can be evaluated with different echocardiographic modalities permitting, with varying degrees of accuracy, the evaluation of segment contraction sequence. Until now, although no parameter on its own has demonstrated itself to be good and reproducible enough to extend its use, the combination of the different echo modalities can detect, in most cases, the presence of intraventricular asynchrony.
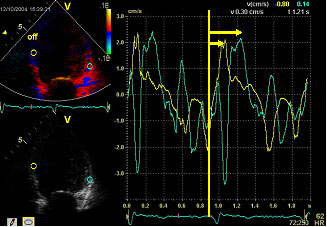
Among the different echo-measurements, the easiest one to obtain is the septum to posterior wall delay, described by Pitzalis et al using the M-mode (Figure 3). Later, more sophisticated determinations applying Doppler Tissue Imaging derived parameters as described by the group of Bax (Figure 4) and the studies of Yu et al, also demonstrated their usefulness in determining LV intraventricular asynchrony. More recently, some parameters derived from Speckle Tracking Imaging and three-dimensional echo have also shown their usefulness in determining the presence of intraventricular asynchrony, although the degree of complexity in obtaining and interpreting these images increases.
Conclusions
Atrioventricular, interventricular and intraventricular asynchrony can be measured with different echo techniques and this is particularly true for determining LV intraventricular synchrony. Although this knowledge would be useful to select patients, to extend CRT indications and to help in CRT programming optimisation, no consensus exists regarding which parameters are the best ones to use. This is the reason why patients continue to be selected by demonstrating the presence of electrical asynchrony.
Figure 1
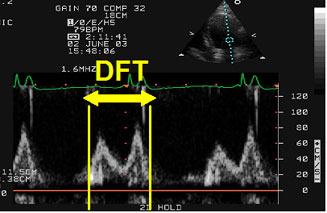
Atrioventricular asynchrony assessment. DFT: diastolic filling time measured with PW - Doppler in the 4-chamber apical view as the time passed from the beginning of the E wave to the end of the A wave. Atrioventricular asynchrony is described if the DFT is < 40% of the cardiac cycle.
Figure 2
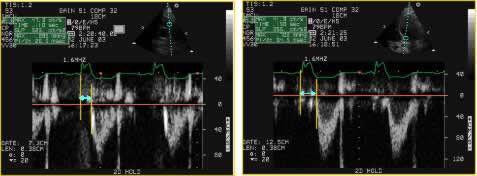
Interventricular asynchrony is assessed with PW - Doppler applied at the aortic and pulmonary valves measuring the difference between the aortic pre-ejection time and the pulmonary pre-ejection time defined as the time passed from the beginning of the QRS complex to the beginning of the pulmonary or aortic ejection.
Figure 3
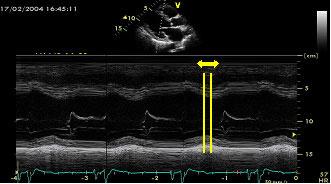
Intraventricular asynchrony determined by M-mode scans in the long axis paraesternal view calculating the time from the septum peak contraction to the posterior wall peak contraction. A delay > 130 ms is diagnostic of LV intraventricular asynchrony.
Figure 4
Septum to lateral wall delay measured as the time difference between the time interval from the QRS beginning to the peak of the systolic contraction of the basal septal and lateral segments in the 4 chambers apical view as Bax et al described. A maximum delay to peak systolic tissue velocity > 60 ms is considered as a criterion of LV intraventricular asynchrony.
The content of this article reflects the personal opinion of the author/s and is not necessarily the official position of the European Society of Cardiology.



 Our mission: To reduce the burden of cardiovascular disease.
Our mission: To reduce the burden of cardiovascular disease.