I - Reduction in Atherothrombotic for Continued Health (REACH) Registry
The results from the Reduction in Atherothrombotic for Continued Health (REACH) Registry have characterised the atherosclerosis risk factor profile or treatment intensity of individuals with atherothrombosis in many countries around the world. This large, international, contemporary database shows that classical cardiovascular risk factors are consistent and common but are largely undertreated and undercontrolled in many regions around the world.
- Patients with atherothrombosis throughout the world had similar risk factor profiles with a high proportion with hypertension (81.8%), hypercholesterolemia (72.4%) and diabetes (44.3%).
- The prevalence of obesity ( 26.6%) and being overweight (39.8%) were similar in different geographic regions, but was highest in North America (overweight 37.1% and obesity 36.5%; p<0.001 vs other regions).
- Statin use prevalence varied from 56.4% in patients with cerebrovascular disease to 76.2% for coronary artery disease patients;
- Antiplatelet agents varied from 53.9% in patients with > 3 cardiovascular risk factors to 85.6% in patients with coronary artery disease.
- Undertreated arterial hypertension was extremely frequent with 50.0% of the patients with elevated blood pressure at baseline (> 140/90 mmHg),
- Impaired fasting glucose was present in 36.5% in those not known to be diabetic.
- Among those with symptomatic atherothrombosis (coronary artery disease, cerebrovascular disease and peripheral arterial disease) 15.9% had symptomatic poly-vascular disease.
- There were relevant differences in cardiovascular outcomes between geographic areas (Figure 1), with the highest rates of major adverse cardiovascular events in Western Europe and Middle East vs other regions; 1-year composite of cardiovascular death, myocardial infarction, stroke, and hospitalisation for atherothrombotic events affected to 21.6% and 18.0% of the patients in both regions, respectively.
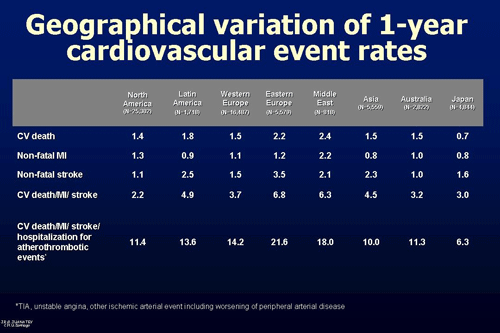
Differences in patients' clinical profile, uncontrolled risk factors and use of proven treatment strategy may explain the differences in outcomes and suggest that additional efforts aimed to improve patients prevention strategy must be carried out, both from an epidemiological and a clinical point of view.
II - HOPE-2 Trial
The results from the HOPE-2 Trial have shown a neutral effect on the incidence of cardiovascular complications of vitamin supplementation (folic acid and B6 and B12) in high risk patients (patients with atherothrombotic disease and type 2 diabetics with at least an additional risk factor included in the HOPE Trial).
This and other clinical trials published during this year have clearly demonstratedthat vitamin supplementation does not help to improve cardiovascular outcomes both in general and high risk populations.
III - The SPARCL Study
The results from the Stroke Prevention by Aggressive Reduction in Cholesterol Levels (SPARCL) Study were one of the cornerstones in the Congress. This was the first trial on secondary prevention with statins in patients with cerebrovascular disease.
Patients (4731) with a stroke or a transient ischemic attack (TIA) in the last 6 months without clinical evident ischemic heart disease and a baseline LDL-cholesterol between 100-190 mg/dl were randomised to atorvastatin (80 mg/day) or placebo.
At the end of follow-up (6 years) the mean LDL was 129 mg/dl in the placebo group and 73 mg/dl in the group of patients receiving atorvastatin with a 16% reduction in the components of the primary endpoint (fatal and non-fatal stroke, 0.84 relative risk, p=0.03) (Figure 2).
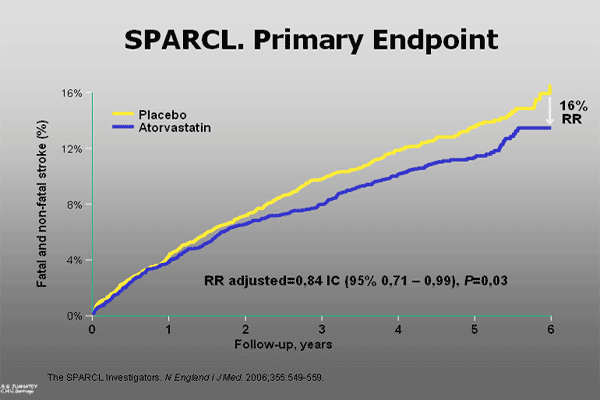
The incidence of stroke and/or TIA (secondary endpoint) was also reduced by 23%.
The prevention of coronary events in this high risk population without previous coronary disease was of special relevance, with a 35% reduction in the incidence of major coronary events and a 42% reduction in the total coronary events (Figure 3).
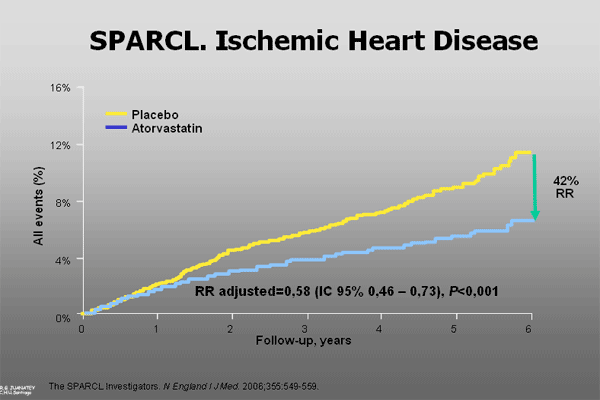
Long-term high dose atorvastatin treatment was well tolerated with a very few cases of severe adverse events, 2 and 3 patients in the atorvastatin and placebo groups suffer from rabdomiolisis.
The SPARCL Trial is the first clinical trial that demonstrates the efficacy of statin treatment in patients with previous cerebrovascular disease; the ischemic heart disease prevention was of special relevance.
Based on the results of a recent meta-analysis of statin trials in coronary artery disease patients, for the LDL reduction obtained with atorvastatin in SPARCL the coronary artery disease risk reduction observed was very similar than that seen in patients with ischemic heart disease. The Trial results indicate that a patient with cerebrovascular disease has a high coronary risk and the benefit observed with statin treatment is similar to what was observed in trials with these drugs in patients with ischemic heart disease. Several factors may influence the limited impact of statin treatment on stroke prevention in patients with previous cerebrovascular disease; a number of patients with ischemic stroke form an embolic origin may dilute the expected beneficial effect of atorvastatin in patients with atherothombotic strokes, and on the other hand may be possible a different impact of statin treatment on coronary and cerebrovascular atherothrombosis.
From a clinical point of view the results of the SPARCL suggest that treatment with a high statin dose may be included in the secondary prevention strategy in patients with cerebrovascular disease.
IV - ASCOT
.
In this clinical trial antihypertensive treatment based on modern drugs :
- a combination of a calcium channel blocker and an angiotensin converting enzyme inhibitor –amlodipine plus perindopril- were more protective than
- therapy based on classic antihypertensives - a betablocker and a diuretic –atenolol plus hydroclorotiazide-.
The differences observed in cardiovascular complications cannot be explained by the slight brachial blood pressure differences during follow-up. The reduction in the incidence of new-onset diabetes and protective vascular actions (on atherothrombotic and vascular remodelling) exerted by the amlodipine plus perindopril combination may contribute to explain the outcome differences.
Nevertheless the results obtained in the ASCOT-CAFÉ sub-study offer the best reasons to explain these differences; in this sub-study, and for the same brachial blood pressure control, the patients treated with the amlodipine plus perindopril combination showed a lower central aortic pressure than in the group treated with atenolol plus hydroclorotiazide.
Based on these results the recent published NICE Guidelines for hypertension treatment have excluded betablockers from first line therapy in uncomplicated patients with hypertension (Figure 4).

The content of this article reflects the personal opinion of the author/s and is not necessarily the official position of the European Society of Cardiology.


 Our mission: To reduce the burden of cardiovascular disease.
Our mission: To reduce the burden of cardiovascular disease.