I – Cardiometabolic Risk : DEFINITION, RELATION TO METABOLIC SYNDROME, HYPERTENSION, AND DIABETES MELLITUS
1 – CARDIOMETABOLIC RISK
Definition
Cardiometabolic risk represents a situation where the possibilities of developing atherosclerotic cardiovascular disease and diabetes mellitus are significantly enhanced as a consequence of the presence of insulin resistance and atherogenic dyslipidemia, this latter characterised by the presence of low HDL-cholesterol and high triglyceride levels (1,2).
Diagnosis
The clinical diagnosis of this situation is made through the finding of an enhanced waist circumference (above 102 cm in males and 88 cm in females) accompanied by the above quoted alterations in lipid profile (HDL-cholesterol below 40 mg/dl in males and 50 mg/dl in females and serum triglycerides above 150 mg/dl).
2 – METABOLIC SYNDROME
Cardiometabolic risk is particularly prevalent in patients diagnosed as having metabolic syndrome
Elements to make the required correct diagnosis of a metabolic syndrome, besides an increased waist circumference, are, a low HDL-cholesterol and elevated triglycerides, the potential presence of blood pressure (BP) values above 130/85 mmHg and a fasting serum glucose above 100 mg/dl according to a recently revisited ATP-III definition (3,4).
The International Diabetes Federation has also defined the metabolic syndrome in Europids by a waist circumference above 94 cm in males and 80 in females accompanied by the finding of two out of the other four criteria that remain unchanged as compared to the ATP-III definition (5).
Nearly half of the hypertensive population can be diagnosed with the Metabolic Syndrome
Figure 1 contains the prevalence of metabolic syndrome according to ATP-III (3) and IDF (5) definitions in a population of more than 1800 hypertensive patients attended at the Hypertension Unit located at the 12 de Octubre Hospital in Madrid, Spain. As can be seen, around 50% to 70% of patients (all hypertensive) were diagnosed as having metabolic syndrome according to the two aforementioned definitions. This figure is not very different when hypertensive population attended in primary care settings is considered (6).
Cardiometabolic risk is then prevalent in the hypertensive population
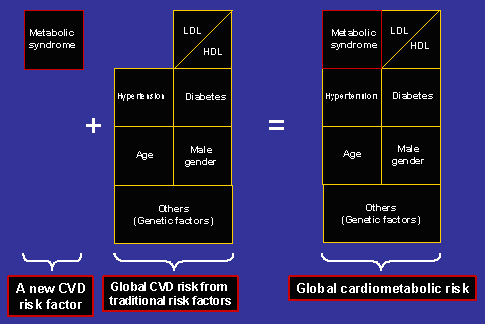
As depicted in figure 2, cardiometabolic risk needs to be incorporated to a correct stratification of risk that has to be done in every hypertensive patient. In fact, the new Guidelines of the European Society of Hypertension- European Society of Cardiology (7) consider the concomitant finding of arterial hypertension and metabolic syndrome as a situation of high-added cardiovascular risk. The reason for this conclusion is based on two facts. Firstly, metabolic syndrome and the accompanying cardiometabolic risk result in a significant increment in cardiovascular morbidity and mortality in several population-based studies, as recently reviewed (9), as well as in hypertensive patients (10). Secondly, the presence of metabolic syndrome is accompanied by a 3-6 times increase in risk of developing type 2 diabetes (11).
As well, the presence of cardiometabolic risk is accompanied by a significant enhancement in the risk of developing chronic kidney disease (albuminuria and/or a diminished estimated glomerular filtration rate). The higher the number of components used for the definition of metabolic syndrome the higher will be the prevalence of either microalbuminuria and/or an estimated GFR value < 60 ml/min/1.73 m2 (12).
3 – DIABETES MELLITUS
The development of new onset diabetes and its relevance in hypertensives has been widely considered recently (13,14). The type of antihypertensive therapy used, alone or in combination, is relevant to accelerate the appearance of diabetes. A recent network meta-analysis (15) has shown that the best protection is obtained when angiotensin receptor blockers and converting enzyme inhibitors are used, while diuretics and betablockers are occupying the last position, in particular when used in combination.
Even though certain authors have denied that the development of new onset diabetes contributes to worsening the short-term (3-5 years) prognosis of hypertensive patients according to the data of studies like SHEP (16) and ALLHAT (17), it seems clear that becoming a diabetic must be relevant for the long-term patient’s prognosis. In fact, it has been shown that with 2 and a half years of follow-up above that in ALLHAT the risk of new diabetics was equal to that of patients entering the study as declared diabetics (18).
II – Cardiometabolic risk : PATIENT MANAGEMENT
1 - MANAGEMENT OF PATIENTS WITH ESSENTIAL HYPERTENSION AND CARDIOMETABOLIC RISK
The aim of intervention in patients with cardiometabolic risk is to achieve an optimal reduction of risk.
Lifestyle modifications counteract the effect of the underlying risk factors (abdominal obesity, physical inactivity and atherogenic diet).
Moreover, hypertensives also require a tight BP control, a choice of antihypertensive treatment not producing other metabolic disturbances, and quite often, parallel drug treatment for associated metabolic risk factors (dyslipidemia, insulin resistance and prothrombotic and proinflammatory states).
Lifestyle interventions
Lifestyle modifications are obviously the first step in achieving the goal of cardiometabolic risk reduction. The key lifestyle interventions are the promotion of exercise and energy expenditure and the reduction of overweight by caloric restriction (19).
The minimal requirements for long-term effectiveness include caloric restriction in the range of 500-1000 Kcal with a 7%-10%, weight loss in 12 months and regular aerobic exercise of 30-45 minutes daily. Whereas extreme caloric-restricted or element dissociated diets have no long-term advantages, more intensive exercise programs have additional cardiovascular benefits and help to maintain weight loss. Lifestyle interventions have clearly beneficial effects on BP and the lipid profile and reduce the incidence of new-onset diabetes (20). Moreover, recent data suggest a long-term effect on the reduction in cardiovascular morbidity (21).
Other lifestyle changes also have a beneficial effect on specific CV risk factors and must be encouraged in specific patients. Lowering salt intake and alcohol consumption have moderate BP lowering effects, which are enhanced in conjunction with weight loss and increased exercise (7). In addition, a diet rich in fruits, vegetables and low-fat dairy products (DASH diet) substantially lowers BP in comparison to the standard American diet (22). Finally, the Mediterranean diet, which is also rich in fruits, vegetables, fish and olive oil, has a favourable impact on atherogenic dyslipidemia in metabolic syndrome patients (23).
Maintenance of lifestyle changes requires counselling and may prove difficult in the long run. For this reason, pharmacological treatment of BP, dyslipidemia, insulin resistance and obesity will be required for most patients to reduce cardiometabolic risk.
BP control
As mentioned above, the new European Society of Hypertension/European Society of Cardiology Guidelines (7) emphasise the importance of metabolic syndrome as an indicator of high added cardiovascular risk in hypertensives, thus indicating early antihypertensive treatment if lifestyle measures are not enough to reach BP targets.
No comparative studies of the different antihypertensive drug classes in hypertensives with metabolic syndrome are available. However, considering the increased risk of developing new-onset diabetes in these subjects, as one of the components of cardiometabolic risk, the choice of antihypertensive treatment must take this additional risk into account.
Some years ago, Lithell showed the influence of drugs on insulin resistance, initiating the idea of categorising drugs as favourable, neutral or negative, in respect to their impact on insulin resistance (24). A few international guidelines recommend diuretics as the first-step therapy for hypertensive patients, without a compelling indication for other antihypertensive drug classes. However, it has been clearly established that diuretics increase the risk of new-onset diabetes compared to placebo (23% increase for diuretics) (15). Conversely, calcium channel blockers and, especially, renin-angiotensin system blockers (angiotensin receptor blockers and angiotensin-converting-enzyme inhibitors) decrease this risk (33% decrease with ACE inhibitors and 43% decrease with angiotensin receptor blockers). These differences are probably even more pronounced in the specific subset of patients with metabolic syndrome and high cardiometabolic risk. Thus, it seems reasonable that the first consideration in antihypertensive treatment in hypertensives with high cardiometabolic risk should focus on the inhibition of the renin-angiotensin system with either ACE inhibition or angiotensin blockade (25).
There is no evidence to support a preference for one or the other of these two forms of renin-angiotensin system blockade. Some of the angiotensin receptor blockers seem to have specific metabolic actions, such as partial agonism of the nuclear receptor PPAR-gamma, although this has not yet been clearly translated into a proven clinical benefit.
2 - MANAGEMENT OF NON-HYPERTENSIVE PATIENTS WITH METABOLIC SYNDROME
Non-hypertensive patients with metabolic syndrome usually have high-normal BP (systolic 130-139 mmHg and/or diastolic 85-89 mmHg). Specific dietary interventions, such as sodium restriction or the adoption of the DASH diet, in addition to caloric restriction and increased exercise, could be helpful.
3 - MANAGEMENT OF HYPERTENSIVE PATIENTS WITH DIABETES OR CHRONIC KIDNEY DISEASE
For patients that also have diabetes or chronic kidney disease, antihypertensive therapy is mandatory (7).
4 - MANAGEMENT OF THE REMAINING SUBJECTS
For the remaining subjects, there is no consensus on whether antihypertensive treatment is recommendable when lifestyle measures are not effective.
In patients with high-normal blood pressure
Various evidences can help to guide this decision. Firstly, the Framingham Study (26) found that subjects with high-normal blood pressure were at an increased risk of cardiovascular events compared to subjects with optimal blood pressure (less than 120/80 mmHg). Secondly, the rate of developing hypertension in a short period (3 years) for those with BP higher than 120/80 mmHg has been reported as very high (40% in subjects older than 64 with BP higher than 130/85 mmHg) (27). In the same way, the recent Trial of Preventing Hypertension (TROPHY) found that, over a period of 4 years, stage 1 hypertension developed in nearly two-thirds of patients with untreated prehypertension (values of 120-139 and/or 80-89 mmHg), and that antihypertensive treatment reduced the risk of incident hypertension in these patients (28).
On the basis of these considerations, it can be hypothesised that antihypertensive treatment could be of benefit for subjects with high-normal blood pressure and high cardiometabolic risk. However, a clear indication needs to be based on a prospective interventional study showing decreased mortality and/or morbidity.
In patients with an increased LDL-cholesterol
Although increased LDL-cholesterol is not considered a component of metabolic syndrome, it must be always a treatment priority. Statins at appropriate doses should be used in all patients with diabetes (29) or cardiovascular disease (30), irrespective of total or LDL-cholesterol levels. Although there is no evidence of a clearly beneficial effect with other drugs, combination therapy with bile acid sequestrants or ezetimibe may help to reduce the statin dose and seems a reasonable alternative.
In patients with cardiometabolic risk but without diabetes or cardiovascular disease
For hypertensives with cardiometabolic risk but without diabetes or cardiovascular disease, the evidence is scarce. However, the AngloScandinavian Cardiac Outcomes Trial demonstrated that treatment with 10 mg atorvastatin was effective in reducing cardiovascular events when hypertension was accompanied by 3 or more additional risk factors, including most that are contained in the definition of metabolic syndrome (31).
The typical dyslipidemia in hypertensives with cardiometabolic risk is characterised by low-HDL cholesterol and increased triglycerides. Two classes of drugs reduce triglycerides and increase HDL-cholesterol. These are nicotinic acid and fibrates. Although limited evidence is available, some post-hoc analyses suggest a beneficial effect of these drugs in patients with metabolic syndrome, insulin resistance, or diabetes (32-35). Thus, current evidence recommends the use of fibrates or nicotinic acid in hypertensive patients with metabolic syndrome and hypertriglyceridemia, but these agents should used with caution in those receiving concomitant statin treatment, especially at higher doses, due to the increased risk of myopathy and liver disorders. Finally, other apparently-promising alternatives, such as cholesteryl ester transfer protein (CETP) inhibition have recently been associated with increased mortality (36).
In patients with impaired glucose tolerance
In addition to lifestyle changes, treatment with metformin (20), acarbose (37) and thiazolidindiones (38) decreases the risk of new-onset diabetes in patients with impaired glucose tolerance. However, the long-term benefits of these drugs and the cost-benefit analysis have not been adequately addressed. In fact, a recent meta-analysis suggests a deleterious effect of rosiglitazone on cardiac outcomes (39).
5 – MANAGEMENT OF WEIGHT LOSS
Abdominal obesity is one the main components of cardiometabolic risk. In addition to a restricted caloric diet and increased exercise, three different pharmacological approaches are available for weight loss.
Sibutramine is the only drug affecting monoaminergic systems currently approved for the long-term control of obesity. Several clinical trials have demonstrated the superiority of sibutramine with respect to placebo in reducing weight and waist circumference (median at one year: 4.5 Kg). In hypertensives, sibutramine slightly increases blood pressure and heart rate and should be used with caution (40). However, a recent trial suggests that the effects of sibutramine in hypertensives under antihypertensive treatment largely depend on the type of the antihypertensive drug used (41). Thus, when sibutramine is coadministered with a combination of RAS blockers and calcium channel blockers, it does not interfere with the antihypertensive effect of such combination.
Orlistat is an inhibitor of gastrointestinal lipases, especially pancreatic lipase. Its efficacy in producing a superior weight loss with respect to placebo has been also proven, although the weight loss is usually lesser than that obtained with sibutramine. Orlistat has a favourable influence on lipids and glycemic control, especially in diabetics, although gastrointestinal tolerance is poor (42).
Rimonabant is the first antagonist of the endocannabinoid receptor CB1. The Rimonabant In Obesity (RIO) program has demonstrated a clear effect of this drug on long-term weight reduction in overweight patients with additional cardiovascular risk factors at one year (43), maintained at 2 years (44), and in hyperlipidemic overweight subjects (45) and type 2 diabetics (46). In all these trials, the beneficial effects on HDL-cholesterol and triglycerides, and on glycosilated haemoglobin in diabetics, exceeded those expected due to weight loss. These facts have led to the consideration that Rimonabant is a drug primarily directed to cardiovascular protection through a direct reduction of the components of cardiometabolic risk. Studies of the effect of this CB1 receptor antagonist on cardiovascular outcomes in patients at risk are currently ongoing.
6 – MANAGEMENT OF THE RISK OF THROMBOSIS
A key feature of metabolic syndrome that explains the increased cardiometabolic risk is an enhanced prothrombotic state, especially in the presence of insulin resistance. Postprandial hyperglycemia, increased free fatty acids and elevated triglyceride levels may all have adverse effects on platelets, coagulation and fibrinolysis. Pharmacological interventions targeting these abnormalities have the potential to reduce thrombosis. Antiplatelet drugs such as low-dose aspirin or clopidogrel represent an option in the management of hypertensives with cardiometabolic risk. The benefit is probably higher in type 2 diabetics (47) and conclusive in those with previous CV disease (48). Efforts to control BP should be reinforced before the introduction of aspirin.
The content of this article reflects the personal opinion of the author/s and is not necessarily the official position of the European Society of Cardiology.
Conclusion:
Simple clinical tools exist which identify subjects at a higher risk of developing both type 2 diabetes and cardiovascular disease, and thus having a high cardiometabolic risk. The management of these subjects is based principally on lifestyle measures, but various antihypertensive, lipid-lowering, insulin sensitising, antiobesity and antiplatelet drugs could be helpful in reducing cardiometabolic risk.
Population-based strategies are clearly necessary to reduce the impact of underlying risk factors for cardiometabolic risk (obesity, physical inactivity and atherogenic diet). However, there is general agreement that more aggressive therapy is required to further reduce the risk of new diabetes and cardiovascular disease, even though evidence is scarce. Prospective, randomised trials addressing the effect of potentially beneficial treatments on cardiometabolic outcomes should be strongly encouraged.
Figure 1. Prevalence of metabolic syndrome in the hypertensive population followed in our Hypertension Unit distributed within the algorithm of risk stratification of European Society of Hypertension/European Society of Cardiology guidelines.
(Figures are percentages: ATP (figures in cursive) and IDF (figures in bold))
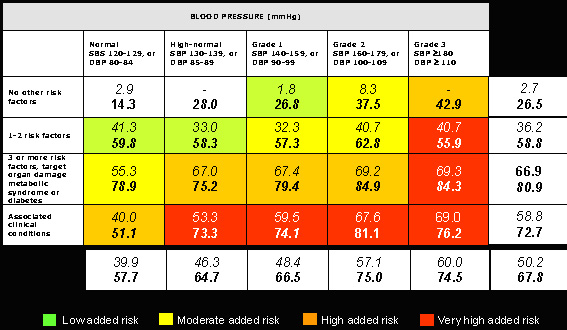
Figure 2. Global cardiometabolic risk includes the presence of metabolic syndrome and traditional cardiovascular risk factors.


 Our mission: To reduce the burden of cardiovascular disease.
Our mission: To reduce the burden of cardiovascular disease.