I - Background
Diseases of the peripheral arteries have often not been of major interest to cardiologists. One of the reasons is that intermittent claudication – the most typical symptom of atherosclerotic disease in the limb arteries- is considered a less disturbing symptom than cardiac dyspnoea or angina pectoris. Still, intermittent claudication can severely affect the quality of life of the patients.
However, more importantly, is the fact that PAD also is a marker of atherosclerosis in arteries elsewhere in the body (ref. 1). Therefore, prognosis of PAD patients is often compromised by the presence of associated coronary and/or cerebral artery disease. Surveys of several medical and surgical series have shown that 40% of the claudicants also have angina pectoris; coronary angiography performed before vascular surgery, reveals significant coronary lesions in almost 90% of the cases (ref. 2). Also, other locations of atherosclerosis such as at the renal arteries can severely increase morbidity and mortality (ref.3).
Diagnosis of PAD can be made quite easily at the consultation by palpation and auscultation of the arteries in the limbs. However, this needs time and expertise. Measuring the ankle brachial index(ABI) is an easy and cheap alternative, totally non invasive, that can elegantly be performed during the consultation. It is expressed as the systolic blood pressure measured at the ankle divided by systolic pressure at the arm. A portable Doppler device is sufficient to perform the measurements.
II - Results of the ankle brachial index (ABI)
a) Result records
The technique has excellent records: for values below the limit of 0.9, specificity is almost 100% and sensitivity 95% (ref. 4). The only confounding condition is that of severe hardening of the vascular wall such as what can be seen in diabetic patients. If this should occur, ABI should be replaced by more complex techniques such as pressure measurement or oscillation recordings at the toe.
b) How to interpret the results
Information obtained by ABI recordings is not like an all or none; the increasing severity of the vascular obstruction is accompanied by a progressive decrease in the index (table 1). In case of the presence of an arterial ulcer, the chances for spontaneous healing or by medical means, can be estimated by measuring the systolic pressure at the ankle; below 50 mm Hg., there rarely is a spontaneous healing, even with careful medical therapy; in diabetic patients, even higher figures (like 80 mm Hg.) are needed to allow for a successful healing of the ulcers.
Table 1. ABI and Severity of Arterial Stenosis.
Mild stenosis: 0.9 to 0.7
Moderate stenosis : 0.6 to 0.5
Severe stenosis : lower than 0.5
c) How the results help in the diagnosis
Besides helping in the diagnosis of PAD and estimating the degree of the obliterative process, measuring ABI can very much help in estimating the long term prognosis of PAD patients. There is a graded inverse correlation between the decrease in ABI and the risk of developing a cardiovascular event (figure 1) (ref. 5-6). Statisticians have sometimes criticised the robustness of this correlation; still, recently, its value was again clearly confirmed in a recent study on a large, well controlled group of patients, followed over a long period of time (ref 7).
Fig.1. Inverse relationship between ABI and the risk of developing CV events.
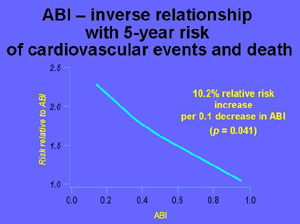
( taken from references 5 and 6.).
Such information is extremely useful in general and especially during the outpatient consultation at the cardiologist’s office. This indication fits very well with the present views as the actual approach and management of patients is largely guided by the patient’s total cardiovascular risk. PAD carries a surprisingly elevated risk ; at middle age, yearly mortality is around 5%!; such impressive figure are further increased by the presence of other risk factors such as hypertension and lipid disorders. Total cardiovascular risk is further increased by associated clinical conditions such as coronary artery disease, whatever the presentation, whether it be stable angina pectoris or previous myocardial infarction. Finally, physicians should keep in mind that patients with no or atypical symptoms also carry this elevated risk; this is another reason why the risk of PAD is often underestimated.
Measuring an ABI has also been used in large scale studies to detect PAD in the population or at the consultation of the general practitioner. In the PARTNERS study (ref 8), the diagnosis of PAD was missed in 86 % of the cases, as long as only clinical history and symptoms were used; ABI was an excellent tool to improve the detection of PAD in this very large group of patients; in many cases, detected by measuring ABI, the presence of PAD was totally unknown to the physician.
The content of this article reflects the personal opinion of the author/s and is not necessarily the official position of the European Society of Cardiology.
Conclusion:
As for many other cardiovascular disorders (see EuroAspire studies), PAD is an underdiagnosed, underestimated and thus, undertreated disorder; symptoms of PAD often are absent or atypical; therefore, physicians and cardiologists in particular, are encouraged to increase their awareness to the disease. Measuring ABI is a very useful tool in reaching this goal.


 Our mission: To reduce the burden of cardiovascular disease.
Our mission: To reduce the burden of cardiovascular disease.