Take-home message
- QTc prolongation is a common finding in diabetes mellitus
- This group is at significant increased risk of sudden cardiac death
- QTc prolongation is multifactorial, due to acute and chronic diabetes-related changes in physiology
- Of the modifiable factors, glucose control, particularly avoiding hyperglycaemia, is key
- The effect of QTc-prolonging medication may be heightened in diabetes and should be prescribed with caution
Introduction
Diabetes mellitus (DM) affects approximately 9% [1] of the global adult population. It is caused by a lack of pancreatic beta-cell insulin production in type 1, and peripheral tissue insulin resistance in type 2. Patients with diabetes mellitus have a two-to-tenfold higher risk of sudden cardiac death (SCD) [2], but the underlying pathophysiology is unclear. The main mode of SCD in this group is likely fatal cardiac arrhythmia, and prolongation of the QT interval is a quantifiable measure of risk. Despite this, general clinician awareness of the specific arrhythmia risk in this cohort is low and improving this is key to addressing modifiable risk factors.
The QTc interval
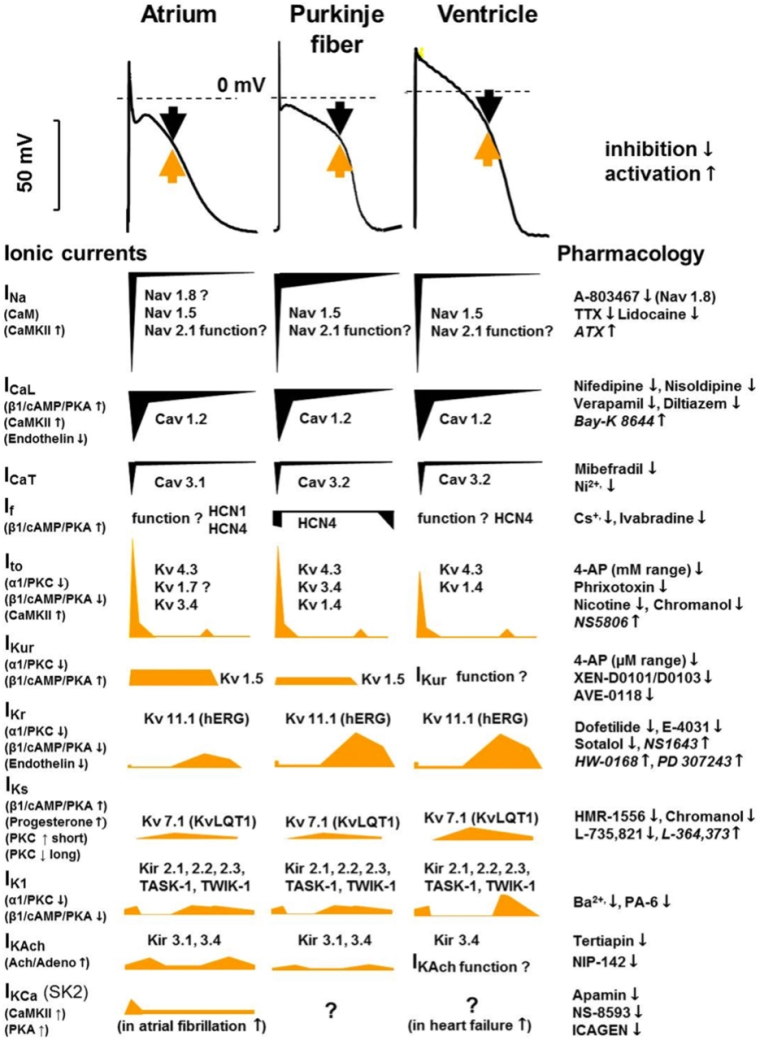
Cardiac myocytes depolarise and repolarise through the exchange of ions (Na+, K+, Ca+) via voltage-gated ion channel openings in the cell membrane (Figure 1). There are multiple ion channels that contribute to the phases of de- and repolarisation, particularly the potassium channels IKr, IKs, and ITo. The QT interval represents the cumulative surface depolarisation and repolarisation of the myocardium and varies with heartrate. This has been further characterised into subintervals JTpeak and TpeakTend, reflecting the differing ion channel activity in each phase of repolarisation.
Figure 1. An overview of the myocardial action potentials and the underlying ionic currents, with corresponding pharmacology and modulation. (Reproduced with permission, Varró et al) [3]
QTc prolongation in diabetes mellitus
QTc prolongation (>440ms) occurs in 18%-44% [4, 5] of type I and II diabetes mellitus patients.
Experimental evidence in animal models and clinical studies of both type 1 and type 2 diabetes has demonstrated multiple cellular, hormonal, inflammatory, neuropathic and physiological processes that influence the QTc interval in diabetes. The catch-all term ‘diabetic myocardium’ [6] has been used to describe the chronic physiological changes in the myocardial tissue induced by long-term diabetes, including increased deposition of advanced glycated end-products, myofibroblast activity, and concurrent coronary heart disease and heart failure. These changes appeared to be mediated by a systemic inflammatory response, with inflammatory factors IL-1b and TNF-alpha [7] demonstrated in animal models to mediate QTc changes. Other hormonal factors have also been observed, summarised in Figure 2.
Figure 2. A summary of factors influencing QTc prolongation in diabetes mellitus.
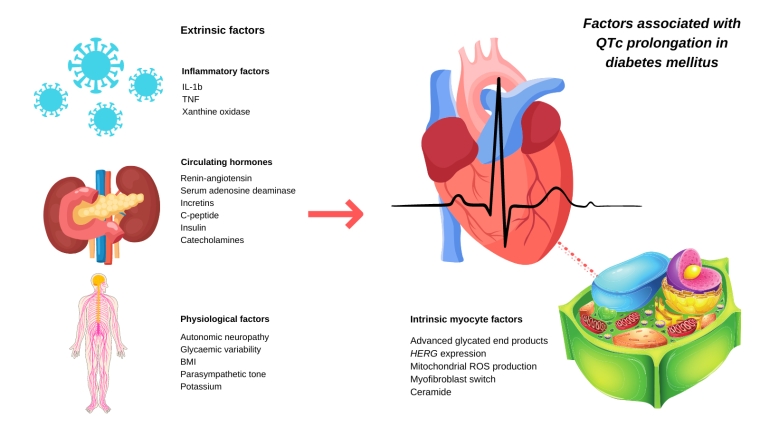
IL: interleukin; TNF: tumour necrosis factor; BMI: body mass index; hERG: human ether a-go-go; ROS: reactive oxygen species
Acutely, blood glucose plays a direct role in modulating QTc, with both hypo- and hyperglycaemia prolonging QTc in healthy volunteers as well as in patients with diabetes. Zhang [8] examined this effect in rat models, hypothesising that hypoglycaemia-induced QTc prolongation was predominantly due to an acute lack of ATP, reducing human ether a-go-go (hERG) ion channel (IKr) function, while hyperglycaemia induced increased reactive oxygen species (ROS), reducing hERG function with the same result. In controlled clinical studies, hyperglycaemia in healthy volunteers considerably prolongs QTc [9], and in large population studies, chronic hyperglycaemia and a raised HBA1C is linked to QTc prolongation [10]. In the critically ill, hyperglycaemia has been observed to correlate closely with QTc prolongation and is a predictor of mortality [11], although this study failed to take into account the severity of illness, an important confounder.
Ion channel basis
Elucidating the underlying cellular pathophysiology is vital to understanding and potentially mitigating the risk of hyperglycaemia-induced QTc prolongation. In animal models, diabetes-induced downregulation of hERG/IKr expression prolongs QTc [12], while this effect is partially corrected by the administration of insulin. Lengyel et al [13] demonstrated a downregulation of Ito and IKs in canine diabetic models, again ameliorated by the administration of insulin. Insulin appears to be an effective counter to the effects of QTc prolongation, and even in healthy volunteers a rise in insulin following a meal shortens the QTc [14].
In-human clinical studies
Gordin et al [6] studied healthy volunteers and patients with type 1 diabetes, demonstrating hyperglycaemia-induced QTc prolongation in both groups. In real-world studies, the use of ambulatory Holter monitoring and continuous glucose monitoring have demonstrated a similar effect [15]. Overall glucose control is a significant contributor to QTc risk. This is important to consider as one potentially modifiable risk factor in this cohort.
Drug-induced QTc prolongation
Drug-induced QTc prolongation is mediated by modulation of several myocardial ion channels, specifically potassium channels (IKs, IKr, ITo). Some drug effects exhibit a QTc-prolonging class effect, such as fluroquinolone antibiotics [16], which led to the withdrawal of several from the market due to FDA guidance. Moxifloxacin for example exhibits an equipotent reduction in function of IKs and IKr, two ion channels specifically implicated in the mediation of hyperglycaemia and diabetes-induced QTc prolongation [16].
‘Repolarisation reserve’
Roden et al [17] described the phenomenon of multiple redundancies in repolarisation mediated by multiple mechanisms as an overall ‘reserve’, which once breached, leads to QTc prolongation. The threshold at which this occurs will vary from person to person depending on their underlying comorbidity, genetic disposition, and physiology. In diabetes mellitus there are both non-modifiable risk factors contributing to this ‘reserve’ and modifiable ones, of which glycaemic control and prescription medications are the most important.
Drug-induced QTc prolongation in diabetes mellitus
Given the heightened risk of sudden cardiac death in patients with type 1 diabetes, this remains a relatively unexplored subject in the literature. In animal studies of extirpated guinea pig hearts [18], the effect of QTc prolonging medication such as dofelitide (selective IKr blockade) was amplified by hyperglycaemia AND hypoglycaemia as compared to euglycaemia, with a greater effect seen with high blood glucose as opposed to low. In anecdotal reports of sevoflurane-induced [19] QTc prolongation, chronic hyperglycaemia (raised HBA1C) was a key risk factor for QTc prolongation. In another study of fluoroquinolone use in a hospital ward setting [20], the effect of QTc prolongation was markedly different in diabetic patients versus non-diabetic. Further research into the specific underlying mechanisms and further awareness of this for the general clinician are vital to better address this high-risk area.
Managing QTc-risk in diabetes mellitus
The first step in managing QTc risk in diabetes is quantifying the QTc appropriately. While many 12-lead ECGs will record QTc according to Bazett formulae, other more accurate methodologies exist, including Framingham and Hodges, with Fridericia shown to be most closely associated with 30-day mortality [21]. Consequently, the authors use QTc corrected by Fridericia (QTcF) in all QTc research and recommend clinical application of this as preference.
Secondly, quantifying QTc over a longer period of time, to account for diurnal variation and effects of everyday activities such as meals, is important to accurately quantify risk. As such, the QTcF should be measured and reviewed over at least 3 ECG recordings separated in time, if possible. A full work-up for family history, long-QT and congenital heart disease should be performed in all women with a QTc >470ms and in all men >450ms. It is important to review all ECGs for consistency of this diagnosis, as opposed to a single spurious result.
Once a patient is defined as at-risk of QTc prolongation, categorising patients into relatively low- moderate- and high-risk groups helps to delineate management. Various cut-offs in the literature have been used to define QTc prolongation, and the authors recommend a simple structure for low risk (<450ms) and high risk (>500ms) patients, with an intermediate group in between.
Addressing the main modifiable risk factors is the mainstay of management, namely glycaemic control and prescription medicines.
Recommendations for practice
Glucose control
As detailed, both hypo- and hyperglycaemia are risk factors for QTc prolongation, with a possible predominance of hyperglycaemia being of greater importance. ‘Glycaemic variability’ [22] has also been associated with increased QTc prolongation, meaning swings from high to low blood glucose levels or vice versa can also increase risk. Glucose control therefore should be targeted to reduce HBA1C but also to maintain diurnal consistency of blood glucose as far as possible, which may require both dietary AND prescription changes. When considering blood glucose control in diabetes patients, consider the degree of left ventricular dysfunction. Prescribing a SGLT2 inhibitor has been shown to reduce the risk of ventricular arrhythmia in heart failure with reduced ejection fraction (HFrEF), for example [23]. Whether this has a direct effect on blood glucose control, the myocardium, fluid overload or all of the above is unclear, but clinical evidence increasingly suggests a better outcome for patients with HFrEF. The addition of insulin to an existing antidiabetic medicine regime in type II diabetes mellitus may also improve glucose control and reduce arrhythmia risk but needs to be carefully weighed against the risk of hypoglycaemia.
Prescribing
A baseline assessment of the patient’s current medications against a recognised database of QTc prolongation properties (such as crediblemeds.org) should be performed, particularly paying attention to anti-arrhythmic medications, antidepressants and antipsychotics, non-penicillin antibiotics, antimalarials, antiemetics and dementia medications (donepezil). Where possible, regular medications should be exchanged for equivalent non-QTc prolonging medications.
Chemotherapeutic agents and anaesthetic agents also carry a particularly high risk of Torsades de Pointes (tDP) and in particular situations (i.e., upcoming surgery or contemplating cancer treatment) additional scrutiny should be given. The high-risk patient (QTc >500) with diabetes should be advised to speak to their healthcare provider in future situations where new medication may be prescribed, particularly in the context of infection, where the combination of dysglycaemia due to sepsis and a new medication causing QTc prolongation could heighten arrhythmia risk significantly.
Lifestyle measures
Raised BMI is a recognised risk factor for QTc prolongation [5] in diabetes and for diabetes mellitus in general, so weight loss is always in the patient’s interest. Similarly, reducing large swings of glucose control by avoiding large meals or high glycaemic index foods may also improve risk.
Conclusions
Sudden cardiac death in patients with diabetes mellitus is a highly significant and under-recognised problem. QTc prolongation is a common finding in this cohort and may serve as a proxy risk of ventricular arrhythmia. The underlying mechanism of this is unclear but is likely multifactorial, involving both acute and chronic changes in the myocardium itself and extrinsic factors. Importantly hyperglycaemia is a key risk factor in the acute setting. Clinicians should assess QTc risk in diabetes and attempt to address modifiable risk factors, predominantly better glucose control, and carefully assess current and potential future medications for additive risk. Patients should be coached on these risks and any potential lifestyle measures that may help reduce it.















