List of Abbreviations
CRT cardiac resynchronisation therapy
CSP conduction system pacing
AF atrial fibrillation
BVP bi-ventricular pacing
LBBB left bundle branch block
HBP His bundle pacing
Introduction
Cardiac resynchronisation therapy (CRT) remains an important treatment for patients with heart failure with reduced ejection fraction and inter-ventricular dyssynchrony. It aims to improve cardiac function by delivering a more physiological pattern of ventricular pacing. Biventricular pacing (BVP) is the most established form of CRT and has been shown to significantly improve morbidity and mortality in patients with heart failure [1].
The development of new tools and techniques has improved success rates of LV lead implantation, but this aspect of the procedure is still a limiting step to BVP. Furthermore, approximately 30% of those receiving CRT with BVP do not experience significant improvement and some even worsen [2]. BVP results in only modest reduction in ventricular activation time and is only beneficial in patients with prolonged QRS reduction, especially left bundle branch block (LBBB). Conduction system pacing (CSP) is an emerging technique to deliver a more physiological pattern of ventricular pacing by directly stimulating the conduction system (bundle of His or more recently left bundle) than the tradition method of CRT with BVP.
Development of CRT delivery over the last decade: tailored CRT therapy:
A number of studies have addressed different techniques in a bid to improve patient selection and to tailor CRT, thus resulting in an improvement in responder rates which is currently 60-70%.
Echo-guided LV lead implantation:
Former studies in CRT echocardiography have shown a relationship between LV reverse remodelling in non-ischaemic cardiomyopathy with concordant LV leads (LV lead placement to the latest contracting area) in relation to segments with delayed mechanical contraction. This was described using TDI with the longest duration of the pulsed wave (PW) A wave to the E wave. This concept was reproduced with both speckle-tracking radial strain [3], colour coded TDI and also 3D echocardiographic LV volume curves.
Other studies have shown the adverse effect of myocardial scarring, particularly in the posterolateral segment of the LV which is the usual site of LV lead electrode placement. It is recognised that patients with LV lead placement in scarred regions have increased heart failure hospitalisations and a reduction in survival when compared to patients with LV leads in scar-free areas. This was shown by studies utilising decreased shortening with circumferential strain as an indirect measure of myocardial scarring [4] and was also supported by MRI based studies [5].
Novel approaches are centred on evaluating speckle-tracking echocardiography for preprocedural targeting of optimal sites for the LV lead electrode to guide the LV lead stimulus towards late-contracting segments and to avoid scarring. Two single centre randomised trials have shown promise when compared to conventional non-image guided implantation, but these results need to be replicated in multicentre studies for validation.
The TARGET study randomised 220 patients to either routine LV lead placement or to implantation into a scar-free site, with the most delayed mechanical activation using speckle-tracking echocardiography. The definition of scar was maximal radial strain amplitude <10%. It was shown that patients with speckle-tracking guided LV lead placement had significantly improved LV reverse remodelling. Furthermore, heart failure hospitalisation and a composite endpoint of mortality were notably higher in patients with routine lead placement [6].
The STARTER study randomised 187 patients to routine LV lead implantation or to echocardiography-guided LV lead placement. Speckle-tracking echocardiography was utilised to target the LV lead to the segment with the most delayed peak in radial strain amplitude. The results demonstrated that echo-guided lead implantation showed significantly improved morbidity and LV remodelling in the echo-guided treatment arm [7].
As both studies incorporated guiding the LV lead towards the latest mechanical activation region and avoided sites of myocardial fibrosis, it is not easy to differentiate which mechanism played a bigger role in the clinical and echocardiographic improvement.
There have been advancements in various echocardiography techniques for individual tailoring of atrioventricular and interventricular delay settings dependant on LV filling patterns, dyssynchrony and stroke volume. Despite showing encouraging single centre study results, data from multicentre trials is inconclusive or lacking.
MRI guided LV lead implantation
Cardiac MRI (CMR) can give detailed circumferential strain data to highlight areas of mechanical dyssynchrony. Coupled with late gadolinium enhancement (LGE) to provide scar assessment, it shows potential such as by improving clinical response rates for targeted LV lead placement.
A large retrospective study of 559 patients with heart failure were enrolled for CRT implantation either guided or not by CMR. The LV lead tip and myocardial scar were localised by fluoroscopy and LGE-CMR. Patients were divided into 3 groups; A) lead placement guided by CMR deployed in scarred region, B) LV lead placement guided by CMR deployed in unscarred region, C) LV lead placement not guided by CMR. After 9.1 years follow-up, group A had highest rates of cardiovascular death or heart failure hospitalisation compared to group B (HR 5.57, p<0.0001). Group C had intermediate risk of both endpoints [8].
Endocardial CRT
LV endocardial placement may overcome limitations of coronary venous anatomy. Endocardial pacing provides multiple benefits over epicardial pacing. In allowing access to shorter path lengths for impulse conduction, fast endocardial conduction mimics a more physiological conduction pattern from the endocardium to the epicardium. It also provides a lower pacing capture threshold and a lower probability of phrenic nerve stimulation.
The paramount benefit of endocardial pacing is the operator’s freedom to select and pace at the optimal region inside the left ventricle, unrestricted by coronary venous anatomy. It is particularly beneficial in patients with ischaemic cardiomyopathy and transmural scarring and is thus less arrhythmogenic.
The ALSYNC study has been the largest prospective multicentre registry assessing the viability and safety of LV endocardial pacing. It enrolled 138 patients with a failed conventional LV lead, suboptimal coronary sinus anatomy or CRT non-response. Procedural success rate was demonstrated in 89% of patients, however it was found that 3.8% of patients had non-disabling strokes. At 6 months, the NYHA class improved in 59% of patients, and 55% had a reduction in LV end-systolic volume (LVESV) ≥15%. Although the response rate in this difficult patient group was promising, the main limitations were the need for lifelong anticoagulation, the high incidence of strokes and the lower rate of optimal lead placement (optimal lead placement was achieved in 81% of implants) [9].
His bundle pacing
His bundle pacing (HBP) involves direct stimulation of the cardiac conduction system, at the level of the native His-Purkinje system to maintain a physiological pattern of ventricular activation. The concept of a pacing modality capable of preserving and potentially restoring physiological conduction is very attractive, especially in heart failure patients. His pacing has been shown to be technically feasible [10] and implant success has increased as a result of the development of dedicated tools for delivering the lead and increased operator experience [11].
Preventing the deleterious effects of right ventricular pacing, delivering more effective cardiac resynchronisation and avoiding complications associated with coronary sinus instrumentation are a few of the other potential advantages.
Abdelrahman et al conducted the largest prospective study comparing HBP with right ventricular pacing (RVP) with 304 successful HBP implants compared with 433 RVP controls. They concluded that HBP implants were correlated with a reduction in all-cause mortality (29% vs 35%, HR 0.73, p=0.58), heart failure hospitalisations (12.4% vs 17.6%, HR 0.63, p=0.02) and importantly, a reduction in the need to upgrade patients to biventricular pacing at a mean follow-up of 4.3 + 3.9 years [12]. These differences were primarily observed in patients with ventricular pacing >20%. Long-term follow-up has shown rising threshold of HBP with a resultant increased frequency of lead revisions in HBP compared to RVP implants, however the long-term benefits of reduction in heart failure hospitalisations has been consistent amongst other studies. Huang et al demonstrated in an observational study of 74 patients with LBBB and heart failure, that patients with HBP had demonstrated an improvement in the LV function and functional class at a mean follow-up of 37.1 months [13]
HBP has significant potential as a therapy for patients with heart failure who are not eligible for CRT, but as discussed above, it is also proving to be a strong alternative for CRT in traditionally eligible patients. HOPE-HF is a UK trial currently investigating the role of HBP in patients with heart failure and first-degree atrioventricular block who are not eligible for CRT due to a narrow QRS or RBBB. Change in exercise capacity is the primary outcome measure, and 160 patients will be randomised to 6 months of AV optimised HBP or no pacing.
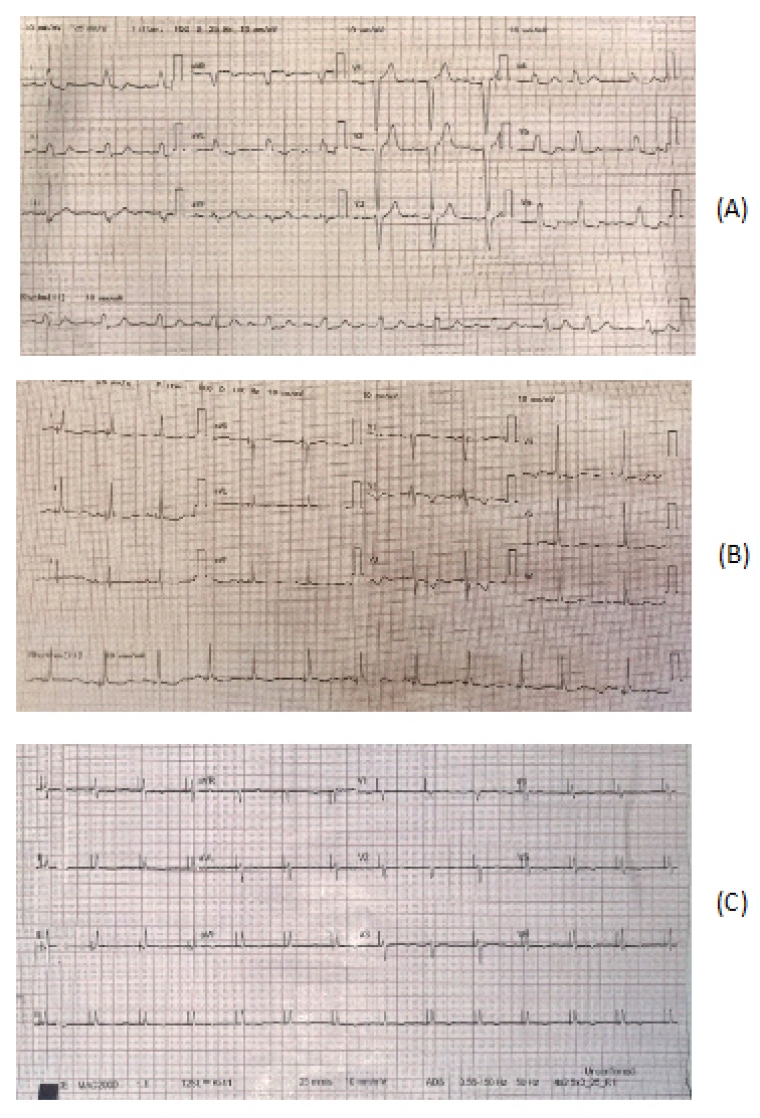
Wang et al assessed the impact of HBP or LBBB pacing combined with AVN ablation in patients with persistent AF with heart failure in a prospective study of 86 patients. 55 patients underwent His pacing with ICD implant and AVN ablation (44 patients underwent HBP, 8 patients underwent LBBB), and 31 patients underwent conventional ICD only with optimal drug therapy. The group with HBP-ICD implantation combined with AVN ablation had a lower incidence of inappropriate shock therapy (15.6% vs 0%, p<0.01) and adverse events (p=0.011)). There was also a significant improvement in LV function noted in the subgroup with HBP (15% vs 3%, p<0.001), therefore validating the benefits of HBP in a range of patients [14]. ECG examples of His bundle pacing in a patient with failed CRT implant and in a patient with AF and AVN node ablation are included in Figure 1.
Figure 1. ECG showing how LBBB (panel A) can be corrected with His Bundle pacing (panel B), another example of HIS bundle pacing and AVN ablation (Panel C)
ECG showing LBBB with normal sinus rhythm B) ECG post HBP in a patient with pre-existing LBBB with a resultant reduction in QRS post implant from 140 ms to 92 ms C) HBP and AV node ablation in patient with atrial fibrillation and heart failure.
LBBB area pacing
With an increase in HBP device implantations, limitations are emerging such as the possibility of unpredictable delayed increases in lead thresholds over time, necessitating further lead repositioning or premature battery depletion. HBP is more technically challenging as it targets a small zone which can be difficult in dilated ventricles, requiring longer operator training and longer procedural and fluoroscopy times. LBBAP is targeted at about 2 cm distal to HBP lead as shown in Figure 2.
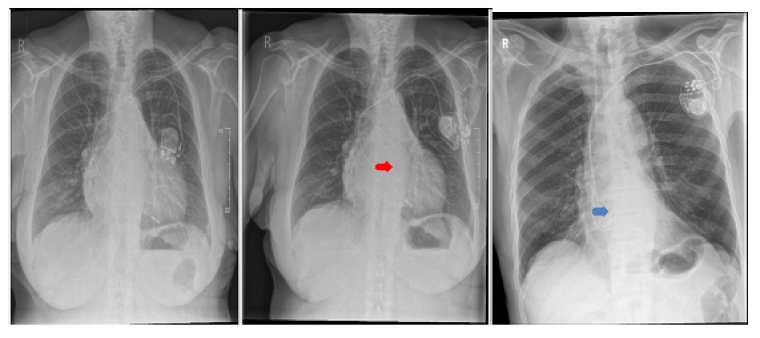
Figure 2. CXR showing the difference in lead placement between (from left to right) RV septal (Panel A), HIS bundle pacing (Panel B) and Left Bundle area pacing (Panel C).
A) VVIR pacemaker implant B) Upgrade from VVIR (previous RV lead in situ) to CRT with left bundle branch pacing lead (red arrow) (Solara CRT-P; Medtronic) C) His bundle CRT-P pacemaker implantation (Note the difference between His lead position [blue] and LBB pacing position [red]).
Regarding these concerns, Huang et al pioneered a novel CSP technique in 2017 whereby the pacing lead was implanted deep in the RV septum to capture the LBBB in patients with heart failure and LBBB [15] resulting in LBBB area pacing (LBBBAP). This has shown promise in overcoming the limitations of HBP whilst maintaining all the benefits of physiological pacing by avoiding the left ventricular dyssynchrony caused by BVP. It is now rapidly emerging as an alternative to failed HBP cases or even as a primary alternative for patients who require brady-pacing.
The early data from a number of prospective studies suggests that capture thresholds are low and stable over time by targeting the more distal conduction system disease.
Hasumi et al attempted to implant LBBAP in 21 patients who had undergone HBP implantation failure in AV block and recorded a success rate of 81%. Notably, the mean procedure time of LBBAP implantation was under 15 minutes and it was observed that the QRS duration decreased from 116±8.3 ms to 108±4.2 ms. Additionally, the group achieved a significant narrowing of the QRS duration in four patients with LBBB (from 151 ± 4.0 to 122 ± 6.7 ms, p=0.01), with a mean capture and LBBB correction thresholds of 0.77±0.07 V/0.4 and 0.89±0.14 V/0.4 ms. Assessment with speckle-tracking echocardiography demonstrated no significant deterioration in the left ventricular total longitudinal strain, relative to intrinsic rhythm, during LBBP. There were also no complications observed during the 6-month follow-up [16].
Vijayaraman et al [17] documented 93% (93/100) and 88% (21/24) LBBP implantation success rates in bradycardia and LBBB patients. They also further validated the finding that LBBBAP could considerably lower QRS duration in patients with LBBB (137±19 ms vs. 162±21 ms, p< 0.001). There was a subset of patients who had procedural complications; three patients had acute lead dislodgments within 24 hours, three had ventricular septal lead perforation and one patient had developed pericardial effusion.
The largest registry of LBBAP published in 2021 by Huang et al that included 632 patients showed high success of 97.8%. The excellent LBB capture threshold at implant was 0.65±0.27 mV at 0.5 ms and 0.69±0.24 mV at 0.5 ms at 2-year follow-up. An improvement in LV ejection fraction (57.08±16.60% vs 62.36±12.20%, p<0.001) and decrease of LV end-diastolic dimension (52.27±7.51 vs 50.73±6.71 mm, p<0.001) were also observed after 1-year follow-up [18].
The limitations of LBBBAP, as it primarily activates the LBBB system, have not been observed sufficiently to assess any long-term disadvantages of a delayed activation of the RBBB.
Conduction system pacing for CRT in patients with LBBB
Over the past few years, conduction system pacing has gained interest with potential to achieve resynchronisation in patient with LBBB. It represents a potentially viable alternative to BVP when LV lead placement is not possible and could also be a method for delivering more effective resynchronisation.
His-CRT
To date, there have only been two randomised trials which compare His-CRT with BVP. However, there were small numbers of patients, and the studies were not designed to assess superiority.
The first was carried by Lustgarten et al using a crossover design with patients randomised to each pacing arm for 6 months. Cardiac function and symptoms improved in both arms with no significant difference detected. However, only 12 patients completed the study and QRS narrowing with His-bundle pacing was achieved in 72% of cases [19].
More recently, Upadhyay et al. conducted the His-SYNC trial which enrolled 41 patients who were randomised to receive either His-CRT or BVP. The findings were limited by significant crossover between groups, 48% in the His-CRT arm and 26% in the BVP arm. The intention-to-treat analysis found no significant difference in echocardiographic or clinical endpoints between the two treatment groups. Treatment-received analysis found a trend towards a greater echocardiographic response in the His-CRT group (80 vs 57% [TR], p = 0.14; 91% vs 54% [PP], p=0.078) [20].
Therefore, His-CRT shows promise as an alternative to BVP for delivering resynchronisation. Larger endpoint trials are required to determine whether the promising results observed in acute, observational, and pilot randomised studies translate into improvement in long-term outcomes.
The groups most likely to benefit from CRT with CSP/His bundle pacing are:
- Failed conventional CRT system (failed LV lead implant, displaced LV lead, poor/absence of capture, phrenic nerve capture and no appropriate venous anatomy).
- Patients with symptomatic atrial fibrillation undergoing atrioventricular nodal ablation.
- Patients who exhibit mild to moderate LVSD EF 35-50 % with indication of pacing who require greater than 40% RV pacing
- Patients with pacing induced cardiomyopathy
- Heart failure with reduced EF with evidence of electrical dyssynchrony without LBBB.
Left conduction system pacing-CRT
Direct comparison with BVP is not available for left conduction system pacing as randomised trials have yet to be performed.
Conclusion
Conduction system pacing can potentially deliver more effective ventricular resynchronisation than conventional cardiac resynchronisation therapy. This can result in greater improvements in cardiac function and clinical outcome. It may also provide the opportunity to extend pacing therapy for heart failure to patients who do not have LBBB. Further data is required from randomised trials to assess these promising pacing techniques.
Take-home message:
- Despite the overall advance in biventricular CRT implantation tools, it is not always feasible. Additionally, there are a significant proportion of heart failure patients who do not respond to it.
- His bundle pacing is the most physiologic way of ventricular stimulation. It is technically challenging with some concerns regarding long-term pacing parameters and lead performance.
- Left bundle area pacing is a very promising technique with a shorter learning curve and more stable lead parameters compared to His bundle pacing.
- Conduction system pacing currently represents a viable alternate to biventricular pacing and might take over some routine pacing and biventricular CRT in the future.
- More evidence is required from randomised controlled trials before conduction system pacing takes a step forward in the guidelines.















