Introduction
In patients with severe valvular heart disease, guideline-based surgical valve replacement or transcatheter implantation of a prosthetic heart valve is associated with improved survival and relief of symptoms. Prosthetic heart valves are designed to replicate the function of native valves by maintaining unidirectional blood flow and can be separated into two broad categories, mechanical and bioprosthetic (also called tissue) valves, each with different advantages and disadvantages.
This mini-series is divided into 4 parts:
Part 1 – Prosthetic valves: selection
Part 2 – Prosthetic valves: antithrombotic therapy
Part 3 – Prosthetic valves: imaging
Part 4 – Prosthetic valves: complications and dysfunction, pregnancy
Part 1 – Prosthetic valves: selection
History
In 1954 Hufnagel and colleagues [1] reported their successful treatment of aortic regurgitation by heterotopic implantation of a ball-valve prosthesis into the descending thoracic aorta. This novel concept in the therapy of heart valve disease set the stage for the development of a new disruptive technology that has caused a sea change. Since then, major advances in technology, surgery, and anaesthesia have made valve replacement a common procedure with relatively low mortality.
Mechanical heart valves
Mechanical heart valves (MHV) are made entirely from non-biological (artificial) materials including metals (titanium, cobalt), pyrolytic carbon, and polymers in order to provide mechanical stability and durability. Different types of MHV include caged ball valves such as the Starr-Edwards valve, tilting disc valves, e.g., the Björk-Shiley and Medtronic valves, and bileaflet valves such as the St. Jude Medical valve with various modifications on these designs. Caged ball valves are no longer implanted; bileaflet valves are the most common type used today. The primary components in this type are usually a fabric-covered sewing ring, stent, hinge, and 2 occluders (also called discs or leaflets) in order to be effective as a one-way valve.
The contemporary disc surfaces are characterised by improved blood biocompatibility and are somewhat resistant to thrombus formation. However, the lack of endothelial lining and blood stasis with the discs in closed position left mechanical valves at risk of thromboembolic complications. The major advantage of mechanical valves is their longevity; however, this benefit must be weighed against necessary lifelong anticoagulation with the combined risk of haemorrhage secondary to anticoagulation and thromboembolic complications in case of subtherapeutic anticoagulation.
The orientation of a bileaflet MHV influences intraventricular haemodynamics. An anti-anatomic orientation (the MHV hinge line oriented towards the left ventricular [LV] outflow tract and not parallel to the commissure line of the native valve) is associated with a more favourable LV flow pattern [2]. This MHV positioning redirects the LV inflow towards the LV outflow tract and may avoid unfavourable effects on LV energy kinetics and decrease the risk of thrombus formation (Figure 1).
Bioprosthetic valves
Bioprosthetic valves (BPV) are, at least in part, made from animal (e.g., porcine, bovine or equine) (heterograft or xenograft) or human (homograft or allograft) tissue mounted onto a metal or polymer supporting structure with three pillars (also called struts or posts) and have a trileaflet configuration that resembles the geometry of a native aortic valve. While porcine aortic valves can be harvested whole with preservation of the natural leaflet attachments (valvar hinges), bovine or equine pericardial tissue has to be trimmed and fitted to a stent to mimic the functional architecture of valve leaflets.
Stentless valves have been introduced with the objective of improving haemodynamics, are essentially metal free and produce few, if any, ultrasound artefacts such as acoustic shadowing. Hence, their appearance on two-dimensional echocardiography and their flow dynamics are close to native valves. In order to decrease the immunogenicity and avoid a humoral or cellular immune system rejection when implanted in the human body, animal-derived valvular or non-valvular tissue valves are fixed in glutaraldehyde, a process that cross-links and masks the antigens and makes the tissue valve biocompatible [3]. Although BPV offer the advantage of avoiding lifetime anticoagulation, they are subject to structural valve degeneration, resulting in limited durability and exposing the patient to the risk of reintervention.
Transcatheter aortic valve implantation
The traditional treatment for severe symptomatic valve disease is surgical valve replacement or repair, utilising an open chest approach or minimally invasive surgery and cardiopulmonary bypass. With the advent of transcatheter aortic valve implantation (TAVI), there has been a paradigm shift in the management of patients with aortic valve stenosis in the last decade, with a rapid development of new devices and expansion to high, intermediate and even low surgical risk populations. As these patients are generally younger with a greater life expectancy, structural valve integrity becomes an important issue. Because TAVI is still a young technology compared to conventional surgical aortic valve replacement (SAVR), it will require several more years, possibly a decade, to demonstrate the same favorable long-term durability.
Compared to SAVR, TAVI shows a higher incidence of paravalvular regurgitation but a lower incidence of severe prosthesis-patient mismatch and lower transvalvular pressure gradients. The number of TAVI procedures has outnumbered aortic valve replacement by open heart surgery for treatment of symptomatic severe aortic stenosis in some countries. This percutaneous technique is now available across the entire surgical risk spectrum. In many patients, life expectancy may exceed transcatheter valve durability, and these patients may require a reintervention. A second TAVI within the first TAVI (so called TAV-in-TAV), which failed because of structural valve failure, is feasible and associated with a similar favourable outcome compared to TAVI for surgical aortic valve dysfunction [4].
Choice of prosthetic valves
The choice between a mechanical and a bioprosthetic valve is a complex decision based on prosthetic valve characteristics, patient-related factors (e.g., comorbidities) and patient preferences such as wishes and expectations. In general, all bioprosthetic valves are at risk of structural valve degeneration leading to a higher rate of reoperation; all mechanical valves require life-long anticoagulation to prevent thromboembolism, which is associated with a higher risk of bleeding. While structural valve degeneration increases with time and decreases with advanced age at the time of implantation, the risk of bleeding grows with increasing age [5]. Life expectancy for patients at the time of valve implantation is greater following aortic versus mitral valve replacement. Hence, patients after aortic valve replacement are more likely to experience clinically relevant BPV degeneration necessitating reintervention taking into account that not all BPV are alike in terms of durability.
A key attraction of BPV is the non-existing requirement for chronic anticoagulation, but the risks of bleeding and thromboembolism with mechanical prostheses are nowadays acceptable, especially in compliant patients with appropriate internationalised normalised ratio (INR) monitoring.
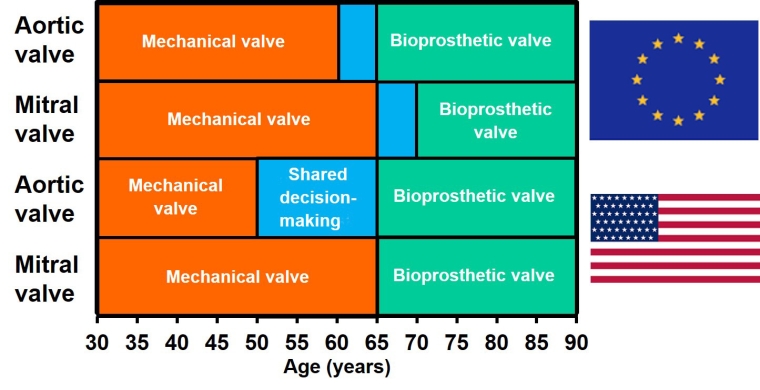
Therefore, the choice of valve depends heavily on the patients’ age, reflecting the time-dependent trade-off between risk of bleeding and need for reoperation. The recent institution of valve-specific U.S. guidelines, in accordance with the European guidelines but with different age limits regarding the selection of MHV versus BPV, takes into account the differences in outcome depending on the valve position [6]. Thus, the balance between valve durability versus risk of bleeding and thromboembolic events favours the choice of a mechanical valve in the aortic position in patients <50 years of age [7] and <60 years of age [8] who require an aortic valve replacement, unless anticoagulation is contraindicated, is not desired, or cannot be monitored (Figure 2). Both guidelines recommend a BPV in patients >65 years of age for aortic valve replacement. For middle-aged patients, both valve types are acceptable and, rather than setting arbitrary age limits, the choice should be individualised through a shared decision-making process with full discussion of the advantages and disadvantages. Anticoagulation with vitamin K antagonists (VKA) requires regular visits for monitoring of INR, point-of-care testing or self-testing (home monitoring) in connection with dietary restrictions, and may be hazardous in case of physical or occupational activities which may lead to accidents.
Finally, the personal perception of many patients regarding valve-related events such as bleeding or reoperation is emotionally charged. When a mitral valve repair is not possible, the age cut-off for implantation of an MHV is set at 65 years [7,8]. In addition to the patient and family, discussion by a Heart Team (consisting of a cardiologist, imaging specialist, cardiac surgeon, anaesthesiologist and others) may be helpful in some cases to take into account all available data and make a recommendation for the best choice and management of prosthetic valve complications after careful, comprehensive evaluation.
Depending on the institutional surgical expertise, the so-called Ross procedure (native pulmonary valve harvested and moved as an autograft to the aortic position combined with a homograft placed in the pulmonary position) represents a relevant alternative in younger patients in whom anticoagulation is either contraindicated or undesired (Class IIb, level of evidence [LoE] B) [7]. The Ross procedure provides excellent valve haemodynamics without the need for anticoagulation. However, both the pulmonic homograft and the neoaortic autograft are at risk of structural degeneration and failure, requiring reintervention at a later stage.
Reoperation for prosthetic valve dysfunction is usually associated with increased risk of morbidity and mortality when compared with the initial surgery due to advanced age and a higher comorbidity burden, particularly in patients undergoing urgent or emergency procedures. Although the in-hospital mortality rate has declined from more than 10% before the 1980s, it remains approximately 5% in contemporary surgery practice [9].
Several other factors should be considered. A mechanical prosthesis may be preferred for patients with another indication for permanent anticoagulation such as atrial fibrillation, high risk of repeat open heart surgery (e.g., porcelain aorta or those with prior radiation therapy) or a small aortic root size which precludes a future valve-in-valve procedure for a structurally failed aortic bioprosthesis. On the other hand, it may be prudent to choose a bioprosthetic valve even in patients who require anticoagulation for another indication with a simultaneous high bleeding risk requiring frequent interruptions for an extended period of time, which would place an MHV at serious risk of thromboembolism. Moreover, certain subgroups of patients, such as continuing drug abusers, in whom life expectancy is lower than the presumed durability of the bioprosthesis, may be candidates for a bioprosthetic valve irrespective of age. A bioprosthesis should also be considered in young women contemplating pregnancy, in whom a bioprosthetic valve is preferred, given the risks of anticoagulation and thromboembolism during pregnancy (Class IIa, LoE C) [10].
The mortality rate among dialysis patients remains high, especially after surgical valve replacement. The current societal guidelines have ceased to have explicit criteria for valve selection; this should be individualised to the patient [7,8]. Young patients without diabetes or New York Heart Association (NYHA) Class III or IV symptoms might survive long enough to justify placement of a mechanical valve, while a biological valve is suitable for most patients as the default option despite accelerated calcification and degeneration [11].
Following the TAVI trend, the ratio of surgically implanted BPV to MHV has been steadily increasing over recent decades [12], owing to a favourable haemodynamic profile, low thrombogenicity, and improvements in tissue processing intended to increase performance and durability. From a lifelong management perspective, a lower age threshold for implantation of BPV includes the consequence that not only will more patients need a reoperation in the future but also that that need will occur at a higher age with a negative impact on survival. The increasing use of BPV in younger patients may also be attributable to the availability of transcatheter valve-in-valve (VIV) implantation for failed aortic and mitral BPV as an acceptable alternative to redo surgery. Since new percutaneous therapies employed for native valve disease also have the potential to replace classic surgical reoperations in patients with deteriorated BPV, this option already has a profound impact on the dynamic discussion regarding the trade-offs between mechanical and bioprosthetic valves. However, transcatheter VIV implantation could also result in several adverse events including suboptimal haemodynamics with higher post-implantation gradients, coronary obstruction, and limited later access to coronary arteries [13].
Table 1. Factors to be considered for the choice of prosthetic heart valve.
|
In favour of a bioprosthesis |
In favour of a mechanical valve |
|---|---|
|
For AVR: age >65 years |
For AVR: age <60/50 years [7,8] |
|
For MVR: age >70/65 years [7,8] |
For MVR: age <65 years |
|
Anticoagulation contraindicated, not desired, or cannot be monitored
|
Other indication for anticoagulation (e.g., atrial fibrillation) |
|
Desired pregnancy Limited life expectancy |
Small aortic root size (preclusion of future transcatheter valve-in-valve [VIV] implantation for failed aortic bioprosthesis) |
|
Patient preference (avoidance of risk or inconvenience of anticoagulation) |
Patient preference (avoidance of risk of reintervention) |
Selection of valve prosthesis size
The term prosthesis-patient mismatch (PPM) was coined in 1978 by Rahimtoola [14] and describes the situation where the effective orifice area (EOA) in a normally functioning prosthetic valve is too small in relation to the patient’s body size to meet cardiac output requirements with incomplete relief of symptoms. The concept of relating the EOA to the body surface area in order to define PPM (severe <0.65 cm²/m², moderate 0.65–0.85 cm²/m² for aortic valves, and severe <0.9 cm²/m², moderate 0.9–1.2 cm²/m² for mitral valves) has become widely accepted [15,16]. However, indexation to body surface area may not be valid in obese patients; other indices based on the lean body mass or height may be more meaningful in this patient population. To overcome this limitation, it is thus recommended to use lower cut-off values of indexed EOA in patients with a body mass index ≥30 kg/m² (severe <0.55 cm²/m², moderate 0.55–0.70 cm²/m² for aortic valves, and severe <0.75 cm²/m², moderate 0.75–1.0 cm²/m² for mitral valves) [15]. PPM is a frequent cause of a high transprosthetic gradient immediately after valve implantation and should be suspected when the measured EOA is in accordance with the normal reference values in a valve with normal leaflet structure and mobility. In contrast to structural valve degeneration, abnormal valve haemodynamics are already present at the time of prosthesis implantation and there is no deterioration in haemodynamics over time.
Of note, prosthetic valve dysfunction (stenosis and/or regurgitation) and PPM may coexist. After aortic valve replacement, PPM is associated with worse haemodynamic performance and diminished cardiac reverse remodelling, leading to impaired functional capacity, accelerated structural valve degeneration (in bioprosthetic valves), and increased perioperative and overall mortality proportional to its severity [17]. Likewise, severe PPM after TAVI is associated with a higher mortality and heart failure hospitalisation rate at 1 year [18].
PPM following VIV implantation for failed aortic bioprosthetic valves could also negatively affect clinical and functional short- and long-term outcomes, specifically in patients with small (internal diameter ≤20 mm) compared to large failed bioprostheses and those with predominant valve stenosis as opposed to regurgitation [19,20]. A potential solution to avoid PPM with the VIV procedure would be to fracture the degenerated BPV preceding the new implantation, although not all BPV stents are breakable. In this context, it would be advisable to surgically implant a valve as large as possible and to avoid a small BPV with resultant PPM in order to preserve the option for a future VIV implantation.
Finally, no randomised controlled trials comparing this technique with redo surgery are available as yet. Nevertheless, this technology has recently been incorporated into the U.S. guidelines for patients with a high or prohibitive risk for redo surgery (Class IIa, LoE B) [7]. PPM after surgical mitral valve replacement has been less widely investigated. Despite some conflicting results, it appears that severe PPM is also an independent predictor of mortality after mitral valve replacement [21].
A prospective strategy to avoid or reduce the degree of PPM includes implantation of a prosthesis with a size as large as possible or a newer-generation prosthesis with superior haemodynamics. Occasionally, concomitant enlargement of the aortic annulus or root may be necessary, permitting implantation of a larger-sized prosthesis. This, however, adds complexity to the surgical procedure and possibly increases the operative risk. Most patients tolerate a mild degree of PPM. For symptomatic patients with severe PPM unresponsive to medical therapy and after exclusion of other causes of increased transprosthetic gradients, the only effective intervention is redo surgery with implantation of a larger prosthetic valve and/or annulus/root enlargement. This approach requires careful consideration of the potential benefits versus the risk of reoperation.