Introduction
Any young individual who intends to participate in competitive sports needs to be screened in detail for risk factors and fitness to prevent the sad and disheartening situation of sudden cardiac death (SCD). SCD is defined as death due to a cardiovascular cause within one hour of onset of symptoms. The prevalence of cardiovascular disease leading to SCD in young athletes is around 0.2%-0.7% [1]. Maron et al reported 0.6 SCD per 100,000 person-years in young competitive athletes in the USA [2]. Corrado et al studied SCD incidence in young Italian players and reported 3.6/100,000 person- years before routine pre-participation screening (PPS) and an incidence of 0.4/100,000 person-years after starting routine PPS [3]. An Italian study comparing the fatality among non-athletic young individuals and young competitive athletes showed 0.9/100,000/year and 2.3/100,000 athletes per year, respectively, suggesting that physical activity in this age group increases the risk of sudden death by 2.5-fold [4].
The various aetiologies of SCD in young athletes are listed in Table 1.
Table 1.
| Common cardiovascular diseases causing sudden cardiac death in athletes | |
|---|---|
| Hypertrophic cardiomyopathy (HCM) | Trauma (commotio cordis) |
| Congenital heart disease | Rupture of ascending aorta |
| Arrhythmogenic right ventricular cardiomyopathy (ARVC) | Mitral valve prolapse (MVP), congenital aortic stenosis |
| Myocarditis | Anomalous origin of coronary arteries (AOCA) |
| Wolff-Parkinson-White (WPW) syndrome | Hypothermia or hyperthermia |
| Long QT syndrome | Drugs (cocaine, amphetamine) |
| Early repolarisation syndrome | Electrolyte imbalance |
| Premature coronary artery disease | Unexplained |
History and physical examination
It is important to obtain a detailed history and conduct a physical examination of young athletes planning to take part in competitive sports. A retrospective study found that 29% of athletes who had SCD had symptoms suggesting cardiac disease before cardiac arrest [5]. A history of present illness, past history and family history will give an insight into the probability of SCD during pre-participation screening. The relevant questions need to be asked after taking the anthropometric data such as height and weight of the young athlete. The physical examination should include comparison of blood pressure in both arms, auscultation for presence of murmurs and examination for features of Marfan syndrome. The routinely asked questions include [6]:
- History of dizziness, chest pain or shortness of breath during or post exercise?
- Any first-degree relatives who have died suddenly or had premature significant cardiac disease under the age of 60 years?
Investigations
12-lead ECG
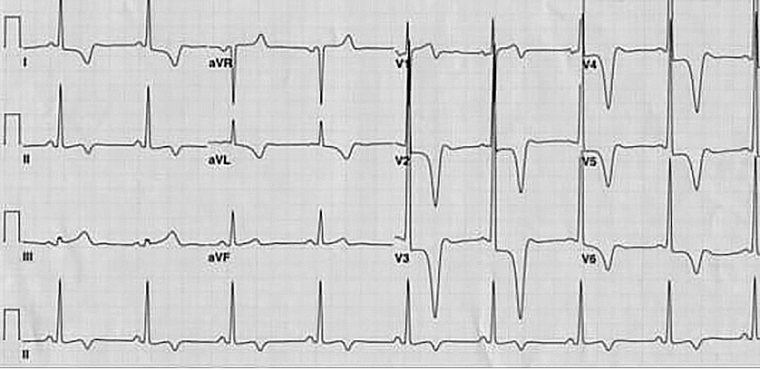
Routine use of ECG for screening athletes has a low specificity and therefore needs strict interpretation criteria. An ECG has been shown to detect hypertrophic cardiomyopathy (Figure 1) in 50%-75% of asymptomatic young athletes and 25% of adolescents with arrhythmogenic right ventricular cardiomyopathies [7]. Premature coronary artery disease may go undetected by ECG alone and warrants further evaluation. Primary electrical diseases are inherited genetic alterations in cell ionic and electric behaviour which lead to arrhythmias and SCD and include Wolff-Parkinson-White syndrome, long QT syndrome, Brugada syndrome, catecholaminergic polymorphic ventricular tachycardia (CPVT) and idiopathic ventricular fibrillation.
The cost-effectiveness of an ECG has been reported from studies in the USA which showed that the cost per athlete life saved ranges from US $44,000 to $204,000 [8]. ECG interpretation with international recommended criteria was associated with significant reductions in secondary investigations and has been shown to reduce the cost of ECG screening by nearly 25% without compromising the ability to identify serious cardiac disease [9]. In 2017, international recommendations were devised by a group of American and European experts with the aim of unifying the recommendations for interpretation of the athlete’s ECG [10, 11]. These recommendations take into account adolescent athletes, black ethnicity and some non-specific electrical anomalies, notably axis deviation and voltage criteria for atrial enlargement, as shown in Table 2.
Table 2. International consensus standards for electrocardiographic interpretation in athletes (with permission from Dhutia et al [11]).
|
Normal
|
Borderline*
|
Abnormal
|
|---|---|---|
| QRS voltage consistent for LVH or RVH | Left axis deviation | T-wave inversion |
| Incomplete RBBB | Left atrial enlargement | ST-segment depression |
| Early repolarisation/ST-segment elevation | Right axis deviation | Pathologic Q-waves |
| ST-segment elevation and TWI V1–V4 in black athletes | Right atrial enlargement | Complete LBBB |
| TWI V1–V3 age <16 years old | Complete RBBB | QRS ≥140 ms duration |
| Sinus bradycardia or arrhythmia | Epsilon wave | |
| Ectopic atrial or junctional rhythm | Prolonged QT interval | |
| 1st degree AV block | Ventricular pre-excitation | |
| Mobitz type I 2nd degree AV block | Brugada type 1 pattern | |
| Profound sinus bradycardia <30 bpm | ||
| PR interval ≥400 ms | ||
| Mobitz type II 2nd degree AV block | ||
| 3rd degree AV block | ||
| ≥2 PVCs | ||
| Atrial tachyarrhythmias | ||
| Ventricular tachyarrhythmias |
* Further evaluation if two or more ‘borderline’ ECG findings identified. Further evaluation required in the presence of any ‘abnormal’ ECG finding.
No further evaluation required if ‘normal’ ECG findings are found in asymptomatic athletes who report no family history of inherited cardiac disease or SCD.
AV: atrioventricular block; ECG: electrocardiogram; LBBB: left bundle branch block; LVH: left ventricular hypertrophy; PVC: premature ventricular contraction; RBBB: right bundle branch block; RVH: right ventricular hypertrophy; SCD: sudden cardiac death; TWI: T-wave inversion
The above-mentioned methods of cardiovascular screening by history, physical examination and ECG have limitations, although the addition of ECG interpretation standards enhances the ability to diagnose underlying pathologies. However, many disease processes may remain undiagnosed and warrant further evaluation. While echocardiography may identify additional structural disorders, there is insufficient evidence to recommend an echocardiogram for routine screening [12]. The diagnosis on an echocardiogram may at times need to be confirmed and substantiated by use of more diagnostic tools such as a computed tomography (CT) scan or cardiac magnetic resonance imaging (CMR) for confirmation of anatomy, and stress testing and Holter monitoring to study exercise tolerance and to unearth underlying arrhythmias in young athletes.
Echocardiogram and other imaging modalities
Echocardiography is a very useful and non-invasive technique which can help the clinician to find the structural heart disease leading to SCD with high sensitivity and specificity. The various presentations of an echocardiogram and further diagnostic tests when indicated in some specific structural abnormalities are discussed as follows.
Hypertrophic cardiomyopathy
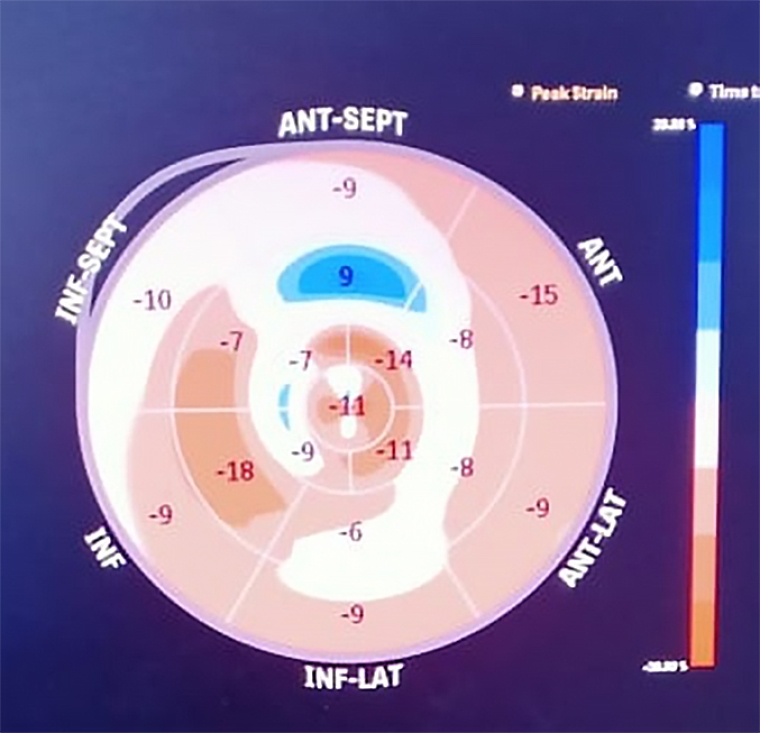
Measurements of left ventricular (LV) wall thickness in end diastole are carried out, preferably in short axis at the mitral, mid LV and apical levels. An LV wall thickness of ≥15 mm in one or more myocardial segments in adults is essential for diagnosis [13]. Screening of first-degree relatives in such cases is also important. Systolic anterior motion (SAM) of mitral leaflets producing LV outflow tract obstruction (LVOTO) in hypertrophic cardiomyopathy (HCM) is defined as an instantaneous peak Doppler LVOTO pressure gradient ≥30 mmHg at rest or during physiological provocation (Valsalva manoeuvre). Strain and strain rate are reduced in spite of a normal ejection fraction (EF), more at the site of hypertrophy. This also helps us to pick up a subclinical disease well before increased wall thickness is evident, as shown in Figure 2.
Idiopathic dilated cardiomyopathy
Mild LV cavity enlargement may be encountered in athletes; however, the indexed LV cavity dimension does not fall within pathological limits. The biplane modified Simpson’s rule to measure EF is operator dependent; therefore, three‑dimensional echocardiography helps to improve the LV volume calculation and EF reproducibility. Abnormal sphericity index is noted in idiopathic dilated cardiomyopathy (IDC), which is calculated by the ratio of length (mitral annulus to apex in the apical view) and diameter (mid‑cavity level in the short‑axis view). The adaptive cardiac remodelling shows normal or even supranormal values of strain and strain rate, while in IDC patients these values are reduced. Stress echocardiography is of assistance in differentiating physiological LV remodelling which shows normal or supranormal contractile reserve, while a reduced EF recovery is suggestive of IDC [14].
Non-compaction cardiomyopathy
Left ventricular non-compaction cardiomyopathy (LVNC) is a rare form of cardiomyopathy caused by arrest of normal myocardial development characterised by multiple deep ventricular trabeculations and is associated with exercise-related SCD in athletes who may be otherwise asymptomatic and undiagnosed. Studies have reported trabeculations in 20% of athletes, and about 8% had conventional criteria for the diagnosis of LVNC; this prevalence increased to 13% when only black athletes were considered. This high prevalence could be due to expression of cardiac adaptation to increased preload and afterload, influenced by genetic factors. Echocardiographic characteristics are useful to distinguish between normal and pathological pattern: the site of trabeculations (apical region in LVNC cardiomyopathy vs mid‑cavity region in athletes), systolic and diastolic function (reduced in LVNC vs normal in athletes), and the preservation of contractile reserve at stress echocardiography in athletes with hypertrabeculation [15].
Anomalous origin of coronary arteries
Anomalous origin of coronary arteries (AOCA) is a frequent cause of SCD during sports and is a rare congenital disease. This subset of individuals is mostly asymptomatic: they have a normal ECG or may rarely present with syncope or chest pain on exertion. Echocardiography is utilised to visualise the origin of both proximal left and right coronary arteries from the coronary sinuses. The main AOCAs associated with SCD in athletes include the origin of the left main coronary artery from the right aortic sinus and the origin of the right coronary artery from the left sinus [16]. Transthoracic echocardiography (TTE) in the parasternal short‑axis view in the plane of the aortic root may distinguish the two coronary ostia and even identify the initial course of the coronary arteries. Colour Doppler echo is useful to identify the proximal course of the coronary arteries. Definitive diagnosis relies on imaging tests such as CMR or CT coronary angiography, which allow an accurate demonstration of the anomalous coronary artery origin and course.
Valvular heart disease
Several studies have reported SCD in young asymptomatic individuals with mitral valve prolapse (MVP) [17]. Echocardiography shows systolic bulging of one or both leaflets towards the left atrium (>2 mm) beyond a line connecting the annular hinge point. Leaflets are generally elongated and thickened with thickness ≥5 mm. A mitral annulus enlarged over 28 mm is associated with mitral annular disjunction (MAD). Mitral regurgitation (MR) evaluation is carried out using colour flow Doppler to evaluate the haemodynamic consequences of MR. MR occurs typically in mid‑to‑late systole and is associated with progressive left cardiac chamber dilatation. The complications of MVP are ventricular and supraventricular arrhythmias, whereas MR has been demonstrated to be an independent risk factor for producing arrhythmias. Young women with a bileaflet MVP or inverted T-waves in the inferior leads and right bundle branch block have been shown to have complex ventricular arrhythmias.
Premature coronary artery disease
Premature coronary artery disease (CAD) is an important substrate for SCD in young people and athletes. SCD is often the first manifestation of the disease in such cases without a past history of angina or myocardial infarction. The proximal left anterior descending coronary artery is the typical culprit vessel. Echocardiography and exercise testing may fail to show myocardial ischaemia or arrhythmias. Rarely, an echo‑inducible myocardial ischaemia may be demonstrated by stress echocardiography.
Myocarditis
Myocarditis accounts for 5%-22% of SCDs in younger populations, more so during exercise. Myocarditis provides a myocardial electrical substrate for life‑threatening ventricular arrhythmias and SCD. An echocardiogram shows global LV dysfunction and may sometimes show regional wall motion abnormalities (RWMA) or pericardial effusion. Myocardial fibrosis is confined to a small myocardial area and involves outer wall layers without decreasing the myocardial thickening; thus, the scar is usually undetectable by echocardiography [18]. A delayed gadolinium enhancement on CMR is useful for scar identification. Avoidance of competitive activity for such athletes is essential, and regular follow-up with the cardiologist is required.
Dissection of the aorta
Dissection of the aorta is a catastrophic complication which may occur in the presence of Marfan syndrome. Measurement of the aorta using an echocardiogram and a CT scan are recommended when needed [19]. Mild aortic enlargement may be noted in athletes due to intense physical training which should not be confused with underlying aortopathy. Therefore, follow-up for aorta dimensions may be needed whenever aortopathy is suspected.
Arrhythmogenic right ventricular cardiomyopathy
Arrhythmogenic right ventricular cardiomyopathy (ARVC) increases the risk of SCD in athletes by 5-fold because of progressive loss of myocytes and replacement by fibro-fatty tissue, creating the substrate for ventricular arrhythmias. ARVC is present in 4%-22% of athletes with SCD. A meta‑analysis showed that subjects with ARVC had a larger RV outflow tract (RVOT), lower tricuspid annular plane systolic excursion (TAPSE) and myocardial strain compared with healthy controls [20]. ARVC patients show a disproportionate enlargement of the RVOT along with significant RV motion abnormalities.
Limitations
An ECG cannot detect all disorders leading to SCD in athletes, and prevention of SCD in young athletes remains the priority of the sports medicine community. There is a compelling argument that resources would be better utilised in improving facilities, training and availability of cardiopulmonary resuscitation and automated external defibrillators (AED). Early identification of individuals at risk through screening is an important strategy to reduce the overall burden of SCD in young athletes. Despite the variety of causes of SCD, only a few conditions are responsible for most deaths. Most athletes who eventually succumb to SCD have no history of cardiac problems (family or personal) and no symptoms before death. Hence, the detection of at-risk individuals poses a significant challenge in sports medicine. As physicians, we recognise that athletic participation carries inherent risks. An enormous amount of research has been done during the past years evaluating the causes and events surrounding SCD and potential screening mechanisms for identifying those at risk.
Conclusion
A long list of tests should be recommended whenever there is any suspicion of developing SCD from family or personal history, clinical examination and ECG. Further evaluation by more tests is necessary in case two or more borderline ECG findings are identified, as per the above-mentioned international ECG criteria and in the presence of any abnormal ECG finding. Asymptomatic athletes with a normal ECG who report no family history of inherited cardiac disease or SCD are not risk-free, as discussed, and one may find a hidden cause if they are given a thorough work-up. The long list of investigations is obviously more expensive but, in some cases, it can be seen to help to decrease SCD effectively. The athletic trainer, in collaboration with the team physician, can implement changes to develop more prudent and effective means of screening for potential cardiac anomalies for prevention of sudden cardiac death.