Introduction
Sudden cardiac death (SCD) in an apparently healthy athlete is undoubtedly a tragic event. Young athletes appear to be at an excessive risk of SCD compared with age-matched non-athletes, although the true incidence of SCD in an athletic population remains controversial [1].
Several studies have noted a higher than expected incidence of “morphologically normal hearts” in young athletes with SCD than was previously suspected [1]. Autopsy-negative sudden unexplained death, also referred to as sudden arrhythmic death syndrome, has been reported in up to 44% of presumed SCD cases, depending on the study population. This group may comprise a significant number of primary electrical disorders or channelopathies that may be identified best by molecular autopsy. In addition to these disorders, failure to detect structural heart disease at post mortem can be explained by the concealed or unknown nature of the pathologic substrate, as well as an underestimation of the potential role of an underlying cardiac pathology in triggering a lethal ventricular arrhythmia.
Sleep health is of growing concern in young athletes, given the detrimental impact of poor sleep on physical and mental health, and in several domains of sports performance, such as aerobic exercise capacity. Sleep disorders may be one of the unrecognised risk factors for SCD in athletes. Sleep-disordered breathing/obstructive sleep apnoea (OSA) can be arrhythmogenic and lead to SCD [2, 3]. However, both the incidence and significance of OSA in young athletes and highly active young people are scarcely known.
Therefore, this article attempts to describe the impact of sleep disorders, especially OSA, on the cardiovascular health of athletes.
Sleep disorders and cardiovascular outcomes
As a resting physiological state, sleep takes up approximately one third of our lifetime; thus, sleep health is an important public health issue. Sleep disorders such as insomnia, inadequate sleep duration, poor sleep quality, and sleep-disordered breathing have a negative impact on physical and social performance and the quality of life and have been associated with cardiovascular outcomes [4-7].
Insomnia is characterised by symptoms such as difficulty initiating sleep, difficulty maintaining sleep, early morning awakening, and/or non-restorative sleep. A meta-analysis of prospective cohort studies demonstrated that insomnia symptoms were associated with an increased risk of future cardiovascular and cerebrovascular events [5]. Both sleep duration and quality are important factors for cardiovascular health. Moreover, abnormal sleep duration, either shorter or longer than optimum, has been shown to be a predictor of cardiovascular morbidity and mortality [4]. However, there is currently a lack of evidence on the link between SCD and abnormal sleep conditions in both the general and athletic populations.
OSA is the most common type of sleep-disordered breathing in the general population [6]. OSA is prevalent in obese, inactive, and aged subjects. The pathogenesis of OSA in obese individuals is through the mechanical effects of enlarged soft tissue structures within and surrounding the airways, thereby contributing significantly to pharyngeal airway narrowing. Thus, OSA is a clinical condition characterised by recurrent episodes of apnoea or hypopnoea of the upper airway, leading to increased negative intrathoracic pressure, sleep fragmentation, and intermittent hypoxia during sleep. The threshold for the diagnosis of OSA is set at an apnoea-hypopnoea index (AHI) of ≥5 over a total sleep duration of more than 3 hours. AHI is evaluated by laboratory polysomnography (PSG) or alternatively home-based portable monitoring devices such as the WatchPAT-200 device (Itamar Medical Ltd, Caesarea, Israel). The AHI is calculated as the sum of the average number of obstructive apnoea and hypopnoea episodes per hour of sleep. The severity of OSA is usually defined as follows: mild: 5 ≤ AHI < 15; moderate: 15 ≤ AHI < 30; and severe: 30 ≤ AHI.
There is accumulating evidence of a relationship between OSA and all major cardiovascular events, including coronary artery disease, atrial fibrillation, and heart failure [2, 6-8]. OSA was found to be a significant predictor of coronary artery disease after adjustment for multiple risk factors in men aged <70 years old [7]. The Wisconsin Cohort Study [8] found a significantly increased risk of combined coronary heart disease and heart failure in patients with mild OSA compared to their non-OSA counterparts. OSA has also been linked to an increased susceptibility to arrhythmogenesis [2]. Furthermore, there is a clear risk of SCD associated with OSA [3].
Arrhythmogenesis and sudden cardiac death in obstructive sleep apnoea
OSA is frequently accompanied by the modification of sleep stage-dependent interactions between the sympathetic and parasympathetic tone. Acute apnoeic episodes can cause hypoxia and hypercapnia, both of which induce a global shift in autonomic nerve balance towards a sympathetic predominance via the chemoreflex [6]. The OSA-related imbalance in autonomic nerve activity may be the primary trigger for cardiac arrhythmogenesis. Intermediate pathways by which OSA may trigger arrhythmias include increased systemic inflammation, oxidative stress, and cardiac structural remodelling [2].
The Sleep Heart Health Study [9] reported a higher rate of cardiac arrhythmias in patients with OSA than in the general population. Patients with OSA exhibit various cardiac rhythm abnormalities including sinus bradycardia, atrioventricular blocks, and atrial and ventricular arrhythmias [2, 10]. OSA is well known to be a risk factor for the initiation and progression of atrial fibrillation. Findings from the Sleep Heart Health Study showed that subjects with severe OSA have a prevalence of 5% for non-sustained ventricular tachycardia and 25% for complex ventricular ectopy such as bigeminy, trigeminy, or quadrigeminy, higher than the general population [9].
In a longitudinal study, OSA predicted incident SCD, and the magnitude of risk was predicted using multiple parameters for OSA severity in a population of 10,701 adults who underwent polysomnography [3]. This study showed that nocturnal hypoxemia strongly predicted SCD, independent of well-established risk factors.
The interaction between OSA and SCD appears to be highly complex and may involve enhanced coagulation activity, myocardial ischaemia, altered ion channel expression, and increased arrhythmogenicity [2, 3]. A number of studies have examined the role of OSA in malignant arrhythmias by characterising its association with high-risk electrocardiographic features [2]. These studies implicate OSA as a concealed risk factor for SCD, although there remains a paucity of evidence for a direct link between the two conditions. The European Society of Cardiology Guidelines recommend that OSA should be taken into consideration in the prevention of SCD [10] (Table 1).
Table 1. Management of ventricular arrhythmias and bradyarrhythmias in sleep apnoea from [10] Priori SG, et al. 2015 ESC Guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: The Task Force for the Management of Patients with Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death of the European Society of Cardiology (ESC). Endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC). Eur Heart J. 2015;36:2793-2867 by permission of Oxford University Press on behalf of the European Society of Cardiology.
Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death Guidelines
| Recommendations | Class | Level |
|---|---|---|
| Sleep apnoea syndrome should be considered in the differential diagnosis of bradyarrhythmias. | II a | B |
| The presence of sleep apnoea and reduced oxygen saturation may be considered as a risk factor for SCD in subjects with sleep disordered breathing. | II b | C |
SCD: sudden cardiac death
Obstructive sleep apnoea in young athletes
Reggie White was one of the best defensive players in the history of the National Football League (NFL) in the USA. He retired in 2000 and died at the age of 43 in 2004. His premature demise was reportedly due, in part, to a fatal arrhythmia associated with OSA. One year before this tragedy, a research letter reported a higher estimated prevalence (14%) of OSA with AHI ≥10 in NFL players than in a cross-section of age- and sex-matched adults and cautioned about the associated cardiovascular complications in such athletes [11].
The prevalence and significance of OSA remain to be explored in athletes who are young and ostensibly in excellent physical condition after the first report from NFL players [11]. Many surveys on the prevalence of OSA have been performed among professional and collegiate players of American-style football. Despite the limited amount of data on a relatively small number of subjects, OSA appears to be highly prevalent in American-style football players [12]. Notably, Kim et al [13] reported that 55% of collegiate athletes demonstrated OSA with an AHI ≥5. The prevalence was higher in linemen than in non-linemen. The prevalence of OSA among athletes, as in other non-athletic populations, is probably driven to a large extent by body habitus with increased body mass index (BMI).
Physiognomy may be a more important risk factor for OSA in athletes than physical conditioning. Athletes who participate in other collision sports also tend to exhibit a large neck circumference and elevated BMI. Twelve of nineteen heavyweight-class judo players with BMI over 30 had OSA evaluated using the respiratory disturbance index [14]. Three studies that emerged in 2019–20 demonstrated a high prevalence of OSA among rugby football players [15-17]. The prevalence of OSA in rugby football athletes in these studies exceeded that previously reported in middle-aged men in the general population.
In a survey of professional rugby teams, OSA with an AHI ≥5 was present in 24% and 45% of athletes screened using in-laboratory PSG [16] and home-based PSG [17], respectively. Increased BMI was associated with a moderate increase in the AHI [17]. We evaluated OSA in freshman athletes on a collegiate rugby football team [15]. Even in athletes in their late teens, 43% of the subjects met the criteria set for OSA, with an AHI of ≥5. However, in a comparison between athletes with and without OSA, there were no significant differences in the BMI and neck circumference. OSA has also been detected in 30% of elite swimmers with a BMI of approximately 20 [ 18]. Thus, the onset of OSA in athletes appears not only to be conferred by body habitus, but may also involve a more complex pathophysiology.
OSA and other sleep disorders negatively affect daytime sleepiness or sports performance in athletes [14, 15, 17, 18]. However, there are few reports on the interaction between OSA and cardiovascular physiology in young and relatively healthy athletic populations. Our exploratory study demonstrated that heart rate during sleep and premature ventricular complex counts on Holter monitoring were significantly higher in teenage rugby players with OSA than in non-OSA athletes, but there were no differences in the cardiopulmonary exercise testing and echocardiographic parameters between the two groups. No microvascular damage or atherosclerotic changes in the retina were detected in young athletes with OSA based on funduscopic observations.
In an analysis of collegiate American-style football athletes, athletes with OSA exhibited a pathologic cardiovascular phenotype associated with higher pulse wave velocity and relative impairment in the left ventricular diastolic function determined by echocardiography, compared with athletes without OSA [13].
OSA, even in the mild stages, is known potentially to have a serious impact on cardiovascular health. However, data defining clinical outcomes in young athletes with OSA are as yet unavailable.
Obstructive sleep apnoea as a potential future cardiovascular risk in athletes
While no serious pathological conditions have yet been detected in collegiate athletes with OSA, several potential warning signs related to a cardiovascular phenotype have appeared [13, 15]. Thus, the primary concern in this population is the worsening of OSA and adverse cardiovascular complications later in life, after conclusion of their athletic careers.
Former NFL athletes were found to have higher cardiovascular mortality than former Major League Baseball players [19], thereby indicating differential sport-specific health outcomes. Retired NFL athletes with obese levels of BMI at playing-time or linemen-position players exhibited increased cardiovascular mortality compared with the other players [12]. Early life cardiometabolic disorders, including OSA, probably contributed to adverse outcomes in NFL athletes.
OSA screening at a younger age in athletes at risk may prevent associated cardiovascular consequences in the future, although further longitudinal studies are necessary.
Evaluation and management for obstructive sleep apnoea
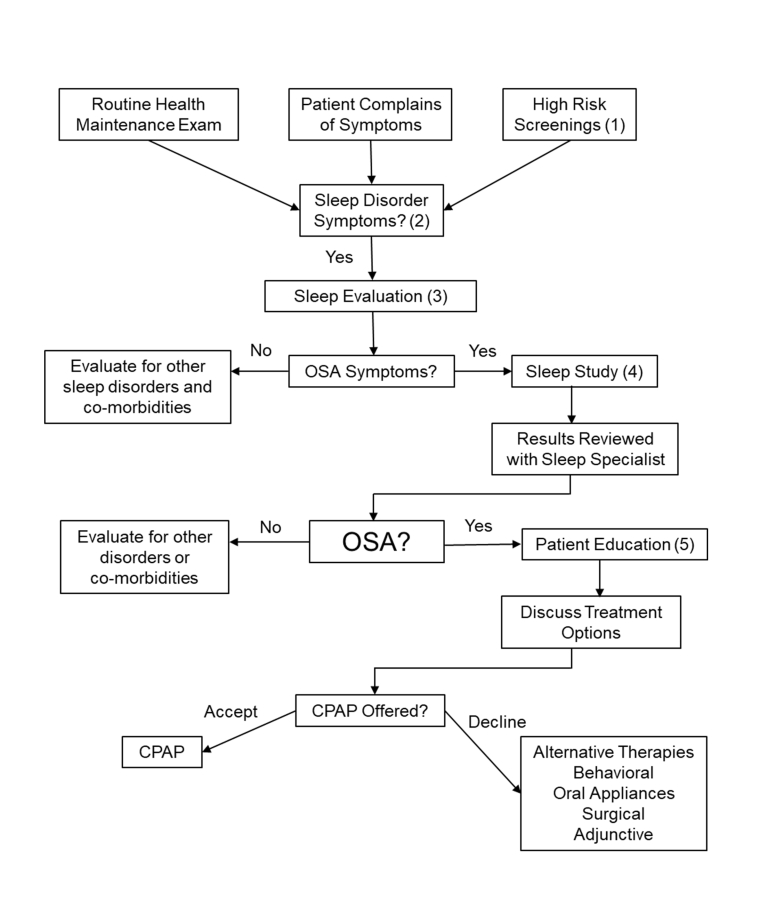
Figure 1 shows the flow chart for evaluation of patients suspected of having OSA from the clinical guideline of the American Academy of Sleep Medicine [20]. Diagnostic criteria for OSA are based on clinical signs and symptoms determined during a comprehensive sleep evaluation, which includes a sleep-oriented history and physical examination, and findings identified by sleep testing. The presence or absence and severity of OSA must be determined before initiating appropriate treatment.
(1) Patient characteristics that indicate they are high risk for OSA include: obesity (BMI >35), congestive heart failure, atrial fibrillation, treatment refractory hypertension, type 2 diabetes, nocturnal dysrhythmias, stroke, pulmonary hypertension, member of a high-risk driving population, and preoperative for bariatric surgery. (2) Along with reviewing the patient’s past medical history and physical evaluations, the following are questions about OSA that should be included in routine health maintenance. Is the patient obese? Is the patient retrognathic? Does the patient complain of daytime sleepiness? Does the patient snore? Does the patient have hypertension? The patient should also be assessed for OSA risk factors. (3) The following sleep disorder symptoms should be evaluated during a comprehensive sleep evaluation: witnessed apnoeas; snoring, gasping, or choking at night; excessive sleepiness not explained by other factors; non-refreshing sleep; total sleep amount; sleep fragmentation/maintenance insomnia; nocturia; morning headaches; decreased concentration; memory loss; decreased libido; and irritability. (4) Sleep studies can include polysomnography (full night or split night), home sleep apnoea testing, and/or other sleep procedures. (5) Components of patient education programmes for sleep medicine include: discussion of the patient’s sleep study and the severity of their disease; pathophysiology of OSA; explanation of the natural course of the disease and associated disorders; risk factor identification, explanation of exacerbating factors, and risk factor modification; genetic counselling when indicated; treatment options; what to expect from treatment; an outline of the patient’s role in treatment, addressing their concerns, and setting goals; consequences of untreated disease; drowsy driving/sleepiness counselling; and patient quality assessment and other feedback regarding evaluation.
CPAP: continuous positive airway pressure therapy; OSA: obstructive sleep apnoea; PCP: primary care physician; SS: sleep specialist
Conclusion
At present, the answer to the question “Are sleep disorders a cause of sudden death during sports activities?” remains uncertain because of a lack of robust evidence.
However, OSA appears to be more prevalent in young athletes than expected, especially in participants of collision sports. The presence of OSA should be inserted in the panel of examinations for risk stratification for SCD [10]. Speculatively, OSA could be attributable, at least in part, to the initiation of pathological processes leading to SCD, even in active athletes. This is a challenging hypothesis that should be further investigated.
Team physicians and cardiologists should be cautious about the possible presence of OSA in active young athletes at risk in order to improve their sports performance and cardiovascular health.