Introduction
As a result of the ageing population in developed countries, the prevalence of degenerative aortic valve disease and its treatment options are increasing [1]. Consequently, it is of vital importance to obtain an accurate assessment of the true severity of aortic valve disease. Severe aortic stenosis (AS) or aortic regurgitation (AR) causing symptoms or left ventricular (LV) dysfunction usually warrants intervention; however, management may be unclear in those with severe disease without symptoms, non-severe disease with symptoms and in symptomatic low-flow AS. Under these circumstances, stress echocardiography may provide additional useful diagnostic and prognostic information [2,3].
Stress echocardiography with physiological exercise is uniquely placed, beyond other imaging modalities, in enabling an objective assessment of functional capacity and correlation of symptoms with the severity of valve disease and LV contractile response. As valve disease tends to progress slowly, symptoms may develop gradually, and many patients may either be unaware of subtle changes in effort tolerance or regard any changes as being non-specific. Moreover, the evaluation of valve disease under haemodynamic stress permits the detection of changes in transvalvular gradients, severity of regurgitation, LV contractility, pulmonary artery (PA) pressure and concomitant myocardial ischaemia. Consequently, the comprehensive information gained from advanced echocardiography may play a pivotal role in the management of aortic valve disease, in particular AS.
Aortic Stenosis
Grading of Aortic Stenosis Severity
The definition of severe AS is based on the knowledge that prognosis is affected once the peak aortic valve (AV) velocity is >4 m/s, corresponding to a mean AV gradient >40 mmHg and a valve area of 0.8 cm2 at a normal flow rate. It has also been shown that a valve area <1 cm2 confers an adverse prognosis. Therefore, severe AS is defined as a valve area <1 cm2 and mean AV gradient >40 mmHg. However, approximately 30% of patients have discrepant findings usually with a valve area <1 cm2 and a lower than expected gradient [4]. In the absence of measurement error, these findings should be considered in the context of the flow across the valve. Historically, a stroke volume index (SVI) <35 ml/m2 has been used to define low-flow, and current guidelines incorporate the use of this parameter to categorise patients into AS stenosis subgroups based on the flow-gradient pattern [5,6]. However, stroke volume refers to the ejection volume rather than flow because it is not indexed to time.
The actual flow rate (FR) across the aortic valve can be derived by dividing the stroke volume by the ejection time. Recent studies have shown that FR may be a more reliable marker of AS severity than SVI [7], and a better predictor of clinical outcome [8,9]. A normal resting FR of >200 ml/sec tends to indicate the true severity of AS as defined by the AV area. However, a reduced FR, which leads to diminished pushing forces through the stenosed AV, may reduce both the valve area and the pressure gradient. Consequently, three main categories of AS can be described as follows:
- Severe AS, defined as a valve area <1.0 cm2 and mean gradient >40 mmHg, irrespective of EF, SVI and FR.
- Low-flow, low-gradient AS, with reduced EF defined as valve area <1 cm2, mean gradient <40 mmHg, EF <50%, SVI <35 ml/m2 and FR ≤200 ml/sec.
- Low-flow, low-gradient AS, with preserved EF defined as valve area <1 cm2, mean gradient <40 mmHg, EF ≥50%, SVI <35 ml/m2 and FR ≤200 ml/sec.
In approximately 25% of patients there is discordance between the SVI and FR thresholds for defining low-flow, and, under these circumstances, FR may be the preferred parameter to guide interpretation of AS severity and subsequent clinical decision making.
Indications for Stress Echocardiography in Aortic Stenosis
In unequivocal severe AS, valve intervention is indicated in the presence of symptoms or LV dysfunction and therefore further diagnostic evaluation is not needed [5,6]. However, stress testing in conjunction with echocardiography may provide useful additional information in patients with (i) asymptomatic severe AS, (ii) symptomatic moderate AS, (iii) low-flow, low-gradient AS with reduced EF, and (iv) low-flow, low-gradient AS with preserved EF [2,3].
Asymptomatic Severe Aortic Stenosis
Exercise testing is a safe and well-established investigation in the asymptomatic patient with normal EF, and when used in conjunction with electrocardiography may be performed to assess symptom status, exercise workload, blood pressure response and ST changes objectively [10]. The procedure may be performed using treadmill exercise or semi-supine bicycle exercise depending on patient and physician preference. A symptom-limited exercise protocol is used with the goal of achieving 85% of the age-predicted target heart rate. When performed in conjunction with echocardiography, the haemodynamic and valve-related data provide useful additional information.
Methodology of Stress Echocardiography in Asymptomatic Severe Aortic Stenosis
Stress echocardiography is the only imaging technique able to provide a comprehensive assessment of valve function, global and regional LV contractility and haemodynamic responses with normal physiological exercise [2,3]. With treadmill exercise, images can only be acquired immediately post-exercise, whereas bicycle exercise allows interrogation of the heart throughout exercise with image acquisitions at lower workloads and at peak stress. Bicycle exercise is also the preferred modality if assessing multiple stress parameters at peak exercise.
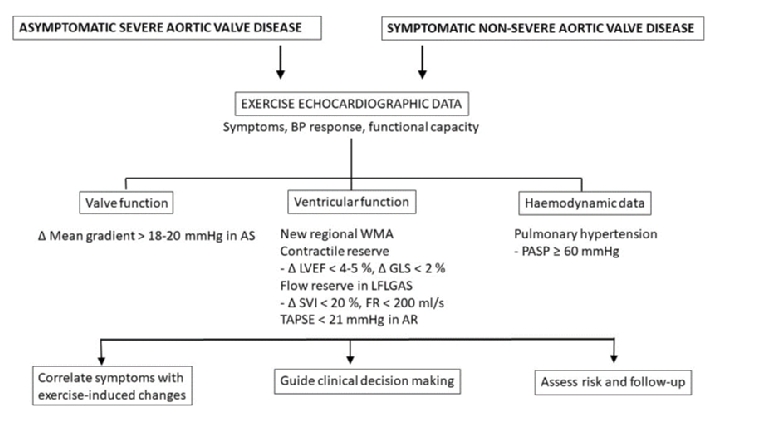
Table 1 provides a summary of the stress echocardiographic protocols in aortic valve disease. In aortic stenosis, the aortic valve measurements at rest should include peak and mean AV gradients and AV area. Apart from interrogating the valve itself, a number of parameters reflecting the exercise-induced haemodynamic consequences of AS should also be measured (Figure 1).
These include a comprehensive assessment of LV function using a quad screen display of conventional apical and parasternal views. Biplane EF can be measured at each stage of image acquisition, global longitudinal strain (GLS) derived at a low exercise workload and regional wall motion analysed at peak stress. This allows an assessment of global LV systolic function, contractile reserve and reversible ischaemia. Contractile reserve is defined as an absolute EF increase >5% and GLS increase >2% at a low exercise workload. As 3D echocardiography is superior for estimation of LV volumes and EF, acquisition of 3D images for assessment of LV contractile reserve may be preferred, but 3D imaging has limited feasibility and, like strain measurements, is constrained by inadequate frame rates at higher heart rates. Resting and peak exercise pulmonary artery systolic pressure (PASP) should also be measured as derived by continuous wave Doppler of the tricuspid regurgitation jet. It is important to note that, in patients with severe AS who are unable to exercise, the use of dobutamine for stress testing should be used with caution and preferably avoided.
Table 1. Summary of Stress Echocardiography Protocols in Aortic Valve Disease Including Type of Stress, Sequence of Image Acquisition and Adverse Endpoints.
| VALVE LESION | ASSOCIATED FEATURES | TYPE OF STRESS | STRESS STAGES | IMAGING SEQUENCE | PARAMETERS MEASURED | ADVERSE ENDPOINTS |
|---|---|---|---|---|---|---|
| Moderate or severe aortic stenosis |
Asymptomatic
EF >50% |
Exercise
|
Rest, low workload, peak stress
|
AV CW Doppler
LVOT PW Doppler LVOT diameter (baseline) LV views TR CW Doppler Colour flow MR |
Peak AV gradient
Mean AV gradient AV area DVI LVEF, wall motion PASP Inducible MR |
Symptoms
∆ systolic BP <20 mmHg ∆ MG 20 mmHg ∆ LVEF < 5% Ex PASP >60 mmHg New regional WMA |
| Low-flow, low-gradient aortic stenosis |
Symptomatic
|
Dobutamine
|
Rest, low workload
|
AV CW Doppler
LVOT PW Doppler LVOT diameter (baseline) LV views |
Peak AV gradient
Mean AV gradient AV area SVI Flow rate LVEF, WMSI |
True severe AS: increase in MG >40 mmHg and AV area <1.0 cm2
Contractile reserve for risk assessment |
| Severe aortic regurgitation |
Asymptomatic |
Exercise
|
Rest, low workload, peak exercise
|
LV views
Colour flow for MR TR CW Doppler |
LVEF
GLS Wall motion Inducible MR PASP |
Symptoms
∆ LVEF <5% New regional WMA |
AS: aortic stenosis; AV: aortic valve; BP: blood pressure; CW: continuous wave; DVI: Doppler velocity index; EF: ejection fraction; GLS: global longitudinal strain; LV: left ventricular; LVEF: left ventricular ejection fraction; LVESD: left ventricular end-systolic diameter; LVOT: left ventricular outflow tract; MG: mean gradient; MR: mitral regurgitation; MV: mitral valve; PASP: pulmonary artery systolic pressure; PW: pulsed wave; SVI: stroke volume index; TR: tricuspid regurgitation; WMA: wall motion abnormality; WMSI: wall motion score index
Diagnostic and Prognostic Value of Stress Testing
An abnormal exercise electrocardiogram (ECG) in asymptomatic severe AS includes symptoms attributable to AS, insufficient rise in blood pressure (BP) (<20 mmHg) or ventricular arrhythmias [10]. Exercise-induced ST changes in severe AS tend to be non-specific and not regarded in isolation as fulfilling the criteria for a positive test. An abnormal exercise ECG has been associated with an increased risk of an adverse outcome, and the development of symptoms may be the most prognostically important finding.
When echocardiography is used in conjunction with an exercise ECG, the echocardiographic data at peak exercise have been shown to provide additional prognostic information. An increase in the mean AV gradient ≥18 mmHg from baseline, an increase in PASP to >60 mmHg and lack of an increase in EF are indicators of a poor prognosis [11-13]. The increase in mean AV gradient may reflect a more rigid, non-compliant valve. Lack of contractile reserve may reflect more advanced myocardial dysfunction and/or exhaustion of coronary flow reserve. Ejection fraction may not be sensitive enough to detect earlier subclinical dysfunction; GLS may be a better predictor of outcome in this group of patients [14]. However, cut-off values for exercise-induced changes in EF and GLS have not been reliably established to aid clinical decision making.
Guidelines recognise a role for stress testing in asymptomatic severe AS and preserved LV systolic function [5,6]. The development of symptoms, a lack of BP increase or ventricular arrhythmias during exercise testing are a class IIa indication for surgery. AV intervention may also be considered when exercise echocardiography demonstrates an increase in mean AV gradient >20 mmHg, but this is not currently incorporated in guidelines.
Moderate Aortic Stenosis
In patients considered to have moderate AS, multiple acoustic windows including suprasternal and right parasternal views should be used to detect the highest AV gradients to avoid underestimation of stenosis severity. Stress echocardiography may be performed in symptomatic patients with moderate AS to assess valve haemodynamics, global LV contractile response and to exclude myocardial ischaemia. Patients with resting mean AV gradient >35 mmHg and an exercise-induced increase in mean gradient by more than 20 mmHg have been shown to be at a higher risk for surgery or death during a mean follow-up of 20 months [15]. These patients should be followed up more closely for monitoring the progression of AS.
Low-flow, Low-gradient Aortic Stenosis with Reduced Ejection Fraction
Low-flow, low-gradient AS refers to a lower than expected AV gradient relative to an AV area <1 cm2. This mismatch occurs due to a reduction in transvalvular flow leading to a reduction in the pushing forces across the AV. The gradient across the AV is proportional to flow squared and therefore a small reduction in flow leads to an exponential fall in gradient. Under these circumstances, the question arises as to whether the AS is truly severe or whether the valve area could increase to a more moderate level with normalisation of the flow rate, so-called pseudo-severe AS. Approximately one third of these patients have true severe AS, one third have pseudo-severe AS and the other third indeterminate AS due to failure to increase FR sufficiently because of reduced contractile reserve, myocardial ischaemia, arrhythmias or induction of left bundle branch block. Flow rate can increase even in the absence of contractile reserve due to shortening of the ejection time during stress.
The second consideration is whether the low-flow state due to LV dysfunction is because of afterload mismatch due to the AS or an alternative concomitant cause such as an underlying ischaemic or non-ischaemic cardiomyopathy. Aortic valve intervention predominantly benefits those with LV dysfunction attributable to severe AS. The presence of regional wall motion abnormalities with scarring and adverse remodelling may point to previous myocardial infarction as the underlying cause. Alternatively, a hypertrophied LV with global hypokinesia may point to advanced severe AS or other contributors of myocardial dysfunction such as longstanding hypertension, diabetes, chronic kidney disease and excessive alcohol consumption. It is important to assess LV contractile reserve as its absence increases the risk of AV intervention.
Low-dose Dobutamine Echocardiography
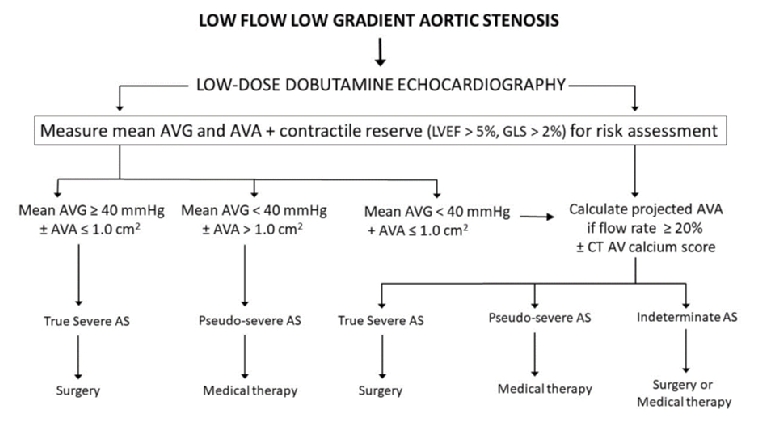
The most commonly used method to evaluate both the severity of AS and contractile reserve is low-dose dobutamine echocardiography. The dobutamine infusion should be started at 5 ug/kg/min and slowly titrated upwards to a maximum dose of 20 mcg/kg/min. The AV gradients, AV area and flow should be measured at each stage of the dobutamine infusion; LV views should also be taken to assess global myocardial contractility and regional wall motion (Table 1). The test should be terminated if the mean gradient exceeds 40 mmHg with an AV area <1 cm2, or with onset of significant ischaemia or haemodynamic compromise. In the absence of the above and an AV area >1.2 cm2, 85% of target heart rate may be aimed for to exclude ischaemia as the cause of LV dysfunction.
Diagnostic Algorithm for Assessing Aortic Stenosis Severity
The diagnostic algorithm for the use of low-dose dobutamine echocardiography in determining the true severity of AS in low-flow, low-gradient AS is shown in Figure 2. The diagnosis of true severe AS is established when the mean pressure gradient is ≥40 mmHg with an AV area <1 cm2. Conversely, pseudo-severe AS is indicated by a mean gradient <40 mmHg and AV area >1.0 cm2. It has been suggested that an AV area of ≤1.2 cm2 at a normalised FR should be considered haemodynamically significant as moderate to severe AS may not be so well tolerated in patients with impaired LV systolic function [16].
When discordance between the AV area and pressure gradient remains (peak stress AV area <1.0 cm2 and peak stress gradient <40 mmHg), it is helpful to calculate the projected AV area at a flow rate of 250 ml/s. The projected AV area is derived from the following formula:
Projected AVA = AVArest + (∆AVA/∆FR) x (250-FRrest) where ∆AVA and ∆FR refer to the absolute changes in AV area and flow rate during low-dose dobutamine. Flow rates and valve areas at each increment of the low-dose dobutamine infusion are used to derive the compliance of the valve from regression of the created line. A projected AV area <1.0 cm2 suggests true severe stenosis. This methodology has been validated to define severity of stenosis and predict adverse events in those with lower projected AV areas [17].
Recently, it has been shown that SVI may not be an optimal marker of transvalvular flow as it represents ejection volume rather than ejection flow. The latter is more appropriately represented by FR and a threshold of 200 ml/s may predict outcome over and above SVI [7-9]. Although prospective studies have established FR as a good marker of flow in AS, it has not yet been incorporated into guidelines.
Guidelines recommend low-dose dobutamine echocardiography to aid decision making in low-flow, low-gradient AS and EF <50% [2,5,6]. Patients with true severe AS should be considered for AV intervention in accordance with guidelines. Those with pseudo-severe AS do not require intervention but should have optimisation of heart failure therapy with close echocardiographic monitoring. In those with indeterminate AS severity, AV calcium scoring by computed tomography (CT) imaging may be used to assess stenosis severity with gender-specific cut-offs for very likely severe AS of approximately 1,600 Agatston units for women and 3,000 for men [6]. AV intervention is associated with higher operative risk, but the outcomes are potentially worse with a conservative approach. Consequently, guidelines provide a weaker recommendation for surgery in these patients; however, if intervention is considered desirable, a less invasive transcatheter approach should be considered.
Low-flow, Low-gradient Aortic Stenosis with Preserved Ejection Fraction
Patients with low-flow low-gradient AS and EF >50% are usually elderly with small LV cavity size and concentric hypertrophy leading to a reduced stroke volume. In these patients, it is of utmost importance to exclude measurement error, particularly of the left ventricular outflow tract (LVOT) diameter. Although low-dose dobutamine echocardiography may be performed, physiological exercise may be used as an alternative method. However, with either modality the test is often not feasible or is inconclusive due to heart failure symptoms related to a restrictive LV physiology and intolerance of sinus tachycardia, or the potential for inducing intracavity gradients and arrhythmias, particularly with dobutamine. Under these circumstances, AV calcium scoring by CT imaging may be used to assess stenosis severity. Although there are no clear guidelines for stress echocardiography in these patients, surgery is indicated in patients with symptoms considered to be due to significant AV obstruction.
Aortic Regurgitation
Surgical AVR is indicated when chronic severe AR is associated with the onset of symptoms, or when EF is <50% or left ventricular end-systolic diameter (LVESD) is >5 cm [5,6]. Exercise testing is recommended in asymptomatic patients with chronic severe AR and normal LV size and function [5]. The purpose of exercise is to assess functional capacity and symptoms objectively, and to assess contractile reserve. There is usually no significant change in AR severity during stress. The shorter duration of diastole with an increasing exercise-induced tachycardia leads to a lower regurgitant volume with each beat, although regurgitant volume over time may not be affected. Resting and stress LVEF, PA systolic pressure and TAPSE should be measured along with LV images using a quad screen display to assess regional wall motion (Table 1). The lack of contractile reserve defined as a <5% increase in EF with exercise has been found to be a predictor of future LV systolic dysfunction [18,19]. Elevation in PA systolic pressure >60 mmHg, presence of dynamic mitral regurgitation (MR) and reversible ischaemia may be potential contributors to symptoms. Assessment of RV function by TAPSE can be performed with exercise and, if <21 mm, has been associated with the need for earlier AVR [20]. The use of GLS and tissue Doppler-derived mitral annular velocities has also been proposed.
Although the information obtained from stress echocardiography in chronic severe AR can be clinically useful on an individual basis, the evidence base for stress echocardiography for this particular indication is limited and to date not included in guidelines.
Conclusions
Exercise testing combined with echocardiography provides useful clinical and prognostic information in those with moderate or severe AS without a clear indication for valve intervention. Low-dose dobutamine echocardiography is an important diagnostic tool for the assessment of true AS severity in low-flow, low-gradient states. The role of exercise echocardiography in AR is less well established, but exercise testing provides an objective assessment of symptoms and exercise capacity which may influence the need for surgery. Larger studies are needed to define better the impact of stress echocardiographic parameters in decision making for intervention and clinical outcomes in aortic valve disease. Further technological advances are also needed to incorporate 3D and myocardial strain imaging with stress echocardiography at higher heart rates.