Introduction
Aortic regurgitation (AR, also termed aortic insufficiency) results in diastolic blood flow from the aorta back into the left ventricle (LV) and may be caused by a variety of acquired or congenital aetiologies preventing complete coaptation of the aortic valve (AV) leaflets (also called cusps) (Table 1). Primary (organic) AR is an intrinsic disease of the AV leaflets, whereas secondary (functional) AR results from distortion or dilation of the aortic root and/or ascending aorta leading to incomplete aortic leaflet coaptation. In analogy to mitral regurgitation (MR), the adapted Carpentier classification is used for AR according to the leaflet motion independently of the aetiology. Type 1: aortic root dilatation with normal leaflet motion and incomplete coaptation or leaflet perforation; type II: excessive leaflet mobility (e.g., prolapse or bicuspid AV) and type III: restricted leaflet motion as the consequence of rheumatic disease or leaflet tenting due to dilatation of the sinotubular junction. Such assessment offers direct clues as to the possibility of valve repair.
Table 1. Aetiologies of chronic aortic regurgitation.
| Primary (organic) | Secondary (functional) |
|---|---|
|
Congenital malformations of aortic valve Leaflet prolapse (degenerative disease) Leaflet perforation (e.g., bacterial endocarditis) Rheumatic heart valve disease Connective tissue disorders (e.g., Marfan’s syndrome) Subaortic stenosis (e.g., membrane, ridge, tunnel) |
Arterial hypertension Idiopathic Syphilis Other connective tissue disorders |
Some disease entities may cause AR by both mechanisms. For example, Marfan’s syndrome and bicuspid aortic valve (BAV) pathologies do affect the structure of the AV and may also cause dilatation of the aortic root and ascending aorta leading to a functionally regurgitant valve. Endocarditis may cause valve destruction by erosion of the leaflet edges or leaflet perforation. In addition, a periannular abscess, at worst, can create a fistula between the aorta and LV.
In fixed subaortic stenosis, which is usually classified as a congenital lesion, the incessant high-pressure jet may damage the aortic leaflets. Therefore, some degree of AR is frequently incurred in a progressive manner dependent on the severity of obstruction.
In the Euro Heart Survey, AR with a grade ≥2/4 was detected in 10.4% of the study cohort [1], while the Framingham Heart Study revealed that the overall prevalence of AR detected by colour Doppler echocardiography of any grade was 13.0% in men and 8.5% in women [2]. Prevalence and severity increase with age and AR is frequently underdiagnosed [3]. The male predominance could possibly reflect a sex bias in referral patterns for diagnosis and treatment [4]. Chronic AR generally evolves slowly and imposes a combined volume (manifested by LV enlargement) and pressure overload (indicated by increased end-systolic pressure) on the LV. This hybrid haemodynamic overload results in remodelling of the LV to normalise wall stress and maintain systolic function and is characterised by eccentric hypertrophy and LV dilatation to accommodate for the regurgitant volume. However, many patients will progress from the compensated into the decompensated phase and develop left ventricular systolic dysfunction with heart failure symptoms. The onset of symptoms (spontaneous or on exercise testing) represents a key development and denotes the strongest indication for intervention.
Bicuspid aortic valve disease
A BAV is the most common congenital cardiac malformation in adults, affecting 1–2% of the general population with a male predominance of ~3:1 [5]. This anatomic abnormality is caused by fusion of the aortic cusps during valvulogenesis creating a so-called “raphe” at the fusion line. The diagnosis is based on the demonstration of two cusps and two commissures during short-axis visualisation in systole. In diastole, a prominent raphe can give the appearance of a third coaptation line and the valve may appear trileaflet, resulting in a false negative result. Supportive features include doming of the cusps and eccentric valve closure line in the long-axis view. There is a wide spectrum of BAV phenotypes with different morphologic characteristics, including partial or complete leaflet fusion and absence or presence of one or two raphes [6]. Rarely, only a single linear coaptation line between two cusps of nearly equal size during diastole (“pure” bicuspid valve) is present. The disease entity is not limited to the valve itself (“valvulopathy”) but may also affect the thoracic aorta (“aortopathy”) by dilatation/aneurysmatic formation of the ascending aorta and/or aortic root as well as coarctation of the aorta. Whereas some of this vascular pathology may be secondary to flow dynamics (so-called post-stenotic dilation), aortic enlargement can also be found in the absence of significant AS or AR, reflecting medial degeneration of the aortic wall. These structural abnormalities at the cellular level are independent of the haemodynamic lesion severity [7]. While both factors are probably operative, the relative contribution of histology and haemodynamics remains debated.
There is a heritable component to BAV disease involving multiple causative and modifier genes and current guidelines [8,9] recommend screening all first-degree relatives of patients with BAV. However, there are no data addressing the possible impact of screening on outcomes or the cost-effectiveness.
The natural history of unoperated patients with BAV has not yet been fully defined. According to the Second Natural History Study, freedom from AV intervention was estimated to be 67% at 20-year follow-up for patients presenting after two years of age [10]. In the community, asymptomatic patients with BAV and no or minimal haemodynamic abnormality enjoy an excellent long-term survival [11]. However, given its high prevalence, BAV disease nevertheless imposes a potentially large burden on cardiovascular medicine.
Aortic stenosis, AR, endocarditis, and aortic dissection are all potential complications of this disease entity [5,11]. In the Olmsted county series of asymptomatic patients with BAV [11], 47% had some degree of AR at baseline. However, the main valvular lesion requiring surgical intervention was aortic stenosis followed by AR and mixed AV disease. AR in BAV occurs independently of aortic stenosis and the mechanism includes prolapse of the larger of unequal cusps, myxoid degeneration of the valve, or may be the result of a former infective endocarditis [5,12]. In addition, the BAV-associated aortopathy can lead to a functionally regurgitant AV, further aggravating the degree of AR.
Rheumatic heart disease (RHD)
RHD is the only long-term consequence of acute rheumatic fever (RF) from single or repetitive episodes leading to a chronic valvular disease. The time interval between the initial episode of RF and clinical evidence of valve disease varies, ranging from a few years to over 20 years [13,14], reflecting the cumulative effect of recurrent episodes of RF and the gradual immune-mediated cellular response. While RF is predominantly a disease in children, a large proportion of patients with RHD present after the age of 40 years and it can even be diagnosed in elderly patients [13,14].
RHD is more common in females and the AV is the second most common valve after the mitral valve to be involved in RHD, almost always with associated mitral valve involvement [14]. Isolated involvement of the AV is exceptionally rare and RHD of the AV almost exclusively leads to AR, while aortic stenosis is a very rare consequence. The most common rheumatic multivalvular disease (anatomic or haemodynamic combination of lesions) consists of mitral stenosis and AR. The histomorphological features of rheumatic AR are characterised by neovascularisation and fibrosis with thickening at the leaflet edges, resulting in leaflet retraction and restricted motion.
The World Heart Federation (WHF) has developed new criteria for the echocardiographic diagnosis of RHD [15]. Three categories, namely definite RHD, borderline RHD (minor abnormalities revealed by echocardiography that could also represent normal variation in the valve structure), and normal, were defined based on 2D-derived valve morphology, continuous-wave Doppler, and colour Doppler. However, there is no diagnostic gold standard, and some of the proposed echocardiographic features are not unique to RHD and should always be interpreted in conjunction with the patient’s clinical findings and with consideration of the individual’s pre-test probability in order to exclude congenital, acquired, and degenerative heart disease as the aetiology of valvular abnormalities before presuming rheumatic origin.
So far, only a few prospective studies with a limited number of patients have evaluated the natural history of milder (latent) forms of asymptomatic RHD incidentally detected through echocardiographic screening. It appears that RHD is a dynamic phenotype, which may regress to normal, remain unchanged, or progress to more severe and even symptomatic disease [16].
Diagnostic modalities
Transthoracic echocardiography (TTE) is the first-line imaging modality for the diagnosis of AV disease including the assessment of the aortic root and proximal ascending aorta. In case of suboptimal acoustic windows, transoesophageal echocardiography (TEE) should also be considered, allowing for better visualisation of the mid-distal ascending aorta and the aortic arch.
Cardiac magnetic resonance (CMR) is an accurate and reproducible imaging modality with increasing availability that serves as an additional diagnostic tool for assessment of AR severity and quantification of LV remodelling. In turn, radionuclide methods no longer play a role in evaluating AR and left ventricular volume and function. Aortic dilatation can be a cause or consequence of AR. CMR and computed tomography (CT) imaging provide excellent visualisation beyond the proximal ascending aorta. Contrast or non-contrast CMR angiography is preferred over CT to minimise iatrogenic radiation exposure.
The measurement of pressures and cardiac output by cardiac catheterisation or the assessment of valvular regurgitation by aortography is restricted to situations where echocardiographic evaluation is inconclusive or discordant with clinical findings and other non-invasive imaging modalities such as CMR are contraindicated or not available [8,9]. In addition, coronary angiography is indicated before valve intervention in patients with symptoms of angina, objective evidence of ischaemia, decreased LV systolic function, history of coronary artery disease, or coronary risk factors (including men aged >40 years and post-menopausal women) [8,9].
Grading of AR severity
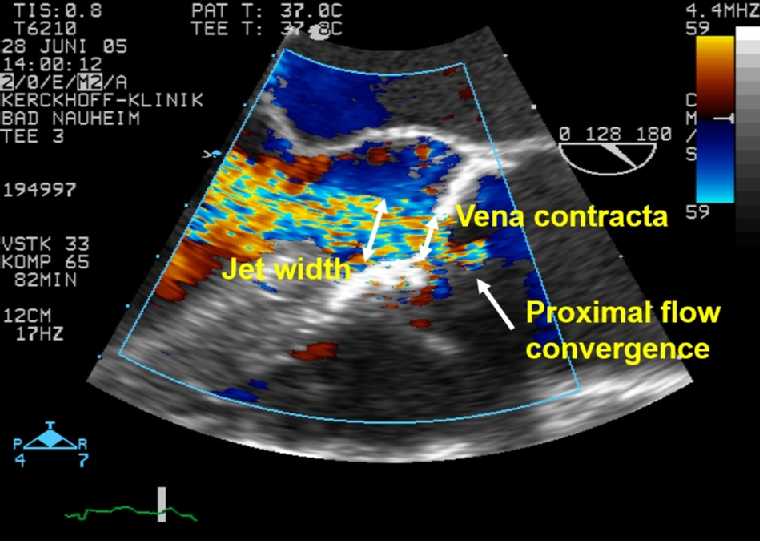
Trivial valvular regurgitation is considered physiological except for the AV. Similar to MR, AR is quantified on a three-grade scale. Colour Doppler echocardiography is the primary method for assessment of AR. While the apical approach is most sensitive for detection, the zoomed parasternal long- and short-axis views are essential for semiquantitative characterisation with visualisation of the three components of the colour jet - proximal flow convergence, vena contracta (VC) and jet width (Figure 1). The jet width and area relative to the left ventricular outflow tract (LVOT) dimension measured within 1 cm apical to the aortic valve leaflets (Figure 2) are an elementary measure of AR severity, but not reliable in eccentric or multiple jets. The VC is the narrowest portion of the regurgitant jet that occurs at or immediately downstream to the regurgitant orifice and reflective of the effective regurgitant orifice area (EROA). The VC width is a simple, easy to obtain semiquantitative parameter that represents a surrogate measurement of the regurgitant orifice area. Since the width of the VC is small, even little measurement variability may have a decisive impact on AR grading. In contrast to MR (VC 7 mm), the cut-off value for severe AR is smaller (6 mm), taking into account the longer duration of diastolic AR compared to systolic MR at rest resulting in a comparable regurgitant volume (RV). Importantly, the length and area of the AR jet into the LV is not a reliable parameter of AR severity. Quantitative methods include pulsed wave Doppler, volumetric and proximal isovelocity surface area (PISA) measurements allowing calculation of the EROA, absolute RV and regurgitant fraction (ratio of RV to forward stroke volume) [17,18]. EROA and RV provide the strongest imaging predictors of outcome in AR [19].
However, no single echocardiographic parameter can accurately grade the severity of AR because of geometric assumptions, technical limitations, and variable haemodynamic conditions. Although it is recommended that an integrated, comprehensive approach using multiple parameters should be used to assess AR severity [17,18], there are no data or validated guidelines on how to weigh each parameter hierarchically. When the evidence from the different parameters is congruent, it is easy to grade AR severity (Table 2). Individual outlying data should be discounted. When different echocardiographic parameters are contradictory, inconclusive or discordant with the clinical assessment, CMR may play a useful role in grading AR severity and assessing LV remodelling [20]. An additional reasonable indication for CMR is detection of replacement or reactive fibrosis. However, quantitative data obtained by CMR and echocardiography are not necessarily interchangeable, in part because of different CMR protocols, and there are no uniform thresholds for grading AR severity coupled with a paucity of outcome data [21]. Further studies are necessary to assess the clinical relevance of CMR-derived data.
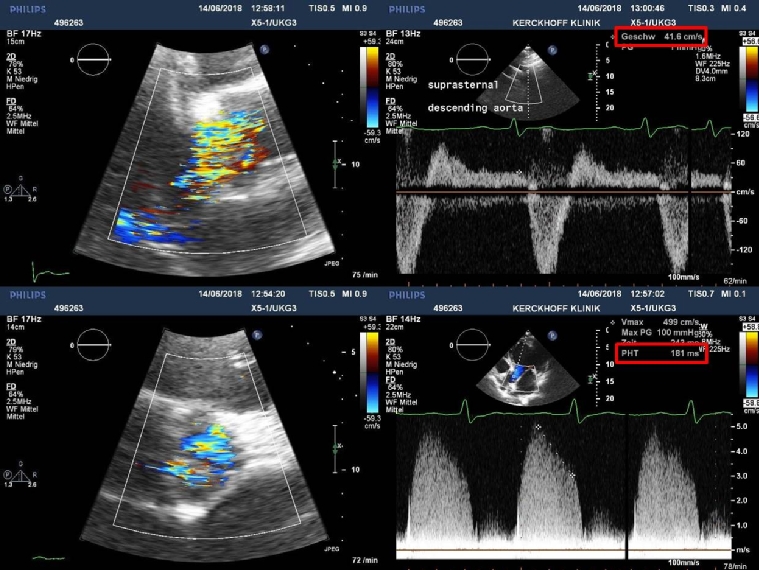
The regurgitant jet width obtained from the parasternal long-axis view (upper left) and area obtained from the parasternal short-axis view (lower left) fill most of the LVOT in diastole (zoom views). Pulsed-wave Doppler spectrum with the sample volume in the descending aorta (upper right) obtained from the suprasternal window depicts holodiastolic flow reversal with an end-diastolic flow velocity >20 cm/sec indicative of severe AR. The continuous-wave Doppler spectrum from the apical long-axis view (lower right) shows a pressure half-time of 180 msec.
Pressure-half-time (PHT)
The PHT derived from the continuous-wave Doppler signal of the AR jet is a function of the pressure equilibration rate between the aorta and LV. The assessment of the PHT requires a good Doppler beam alignment. The early diastolic flow velocity should be at least 4 m/s corresponding to a pressure difference of 64 mmHg according to the modified Bernoulli equation. With increasing severity of AR, the driving pressure significantly decreases throughout diastole with a steeper deceleration slope. However, stiffness of the aorta and LV as encountered in diastolic dysfunction has a confounding influence on the PHT and, depending on the grade of diastolic dysfunction, may cause overestimation or underestimation of the AR severity for a given regurgitant fraction [22].
Table 2. Grading of chronic AR severity with echocardiography.
| Mild | Moderate | Severe | |
|---|---|---|---|
| Vena contracta (mm) | <3 | 3 – 6 | >6 |
| Jet width/LVOT width (%) | <25 | 25 – 64 | ≥65 |
| Jet area/LVOT area (%) | <5 | 5 – 59 | ≥60 |
| Pressure half-time (msec) | >500 | 200 – 500 | <200 |
| Regurgitant volume (mL/beat) | <30 | 30 – 59 | ≥60 |
| Regurgitant fraction (%) | <30 | 30 – 49 | ≥50 |
| Effective regurgitant orifice area (cm²) | <0.1 | 0.1 – 0.29 | ≥0.3 |
LV size and function
The severity and chronicity of AR invariably lead to LV remodelling, which represents a supportive but not specific indicator for AR severity. The European and US joint guidelines on cardiac chamber quantification [23] recommend measuring LV internal diameters in the parasternal long-axis view at or immediately below the level of the mitral valve leaflet tips. However, LV linear measurements are prone to some intraobserver and interobserver variability depending on image quality, timing, and method used. Moreover, a non-uniform LV remodelling pattern in AR towards a more spherical shape with enlarged midventricular or distal portions may not be adequately described by linear dimensions at the LV base. Therefore, LV remodelling may be better appreciated by volume measurements, also because the diameter-volume relationship is non-linear [24]. Indeed, LV volumetric measures were shown to be superior to cavity diameters in predicting clinical outcome in asymptomatic patients with severe AR [19]. Three-dimensional echocardiography reveals more accurate assessments of volumes and ejection fraction (EF), as it avoids foreshortening of the LV [25]. Nevertheless, the current guidelines [8,9] still refer to LV diameters for clinical decision making because of limited outcome data for LV volumes.
Myocardial deformation
In severe AR, multiple haemodynamic factors can alter the EF and could limit its use as the sole measure of left ventricular performance. Myocardial deformation (also called strain) is a newer echocardiographic measure of systolic function and readily assessable by 2D speckle tracking, a relatively angle-independent technique that tracks unique intramyocardial features in conventional echocardiographic images, referred to as speckles. Global longitudinal strain (GLS) seems to be a more sensitive parameter of incipient LV dysfunction compared to EF. Preliminary, hypothesis-generating studies [26] have suggested that GLS adds incremental information to standard measures of LV volume and function in asymptomatic patients. The current guidelines acknowledge the potential role of LV GLS in detecting subclinical myocardial dysfunction in AR [8,9]. However, more validation and data on cut-offs and outcome are necessary for GLS to become part and parcel of the clinical decision-making process.
Biomarkers
In patients with AR, biomarkers may be useful for assessment of the LV response to the haemodynamic overload. For example, B-type natriuretic peptide (BNP) is released as a prohormone by ventricular myocytes in response to stretch. NT-proBNP (biologically inactive N-terminal fragment of its prohormone) levels have a positive correlation with AR severity [27], and BNP levels provide an incremental prognostic value in patients with severe asymptomatic AR [28]. However, many variables affect natriuretic peptide levels and the precise cut-off values have not yet been well defined. Little information is available on the use of other biomarkers in this setting. In summary, biomarkers may be currently used in a complementary fashion as an additional clue in determining the optimal timing of intervention. However, further experience pertaining to patient outcomes is necessary before firm recommendations can be proposed [8,9].
Exercise
In the compensated stage, patients with severe AR remain asymptomatic for years to decades. The onset of symptoms represents a key development since symptom presence is a well-known predictor of adverse outcome and a class I indication for surgery [8,9], yet symptom status is highly subjective. Many patients with severe AR may not recognise their slowly and insidiously progressing symptoms, may deny them, or may adopt a sedentary lifestyle to remain asymptomatic. Therefore, in allegedly asymptomatic patients or those with equivocal symptoms, a standard exercise stress test is helpful to confirm or disprove the absence of symptoms with daily life activities, can be used to objectively assess functional capacity, and provide the baseline for future symptom assessment. Kinetic changes in LV contractility as assessed by EF have only a limited role because the exercise EF response reflects the total stroke volume and does not distinguish between regurgitant flow and forward flow.
Serial examinations
Once a diagnosis of severe regurgitation is established, clinical surveillance and periodic echocardiography with TTE is currently the method of choice to assess the progression of the haemodynamic consequences on LV size and function [8], because significant LV dysfunction may occur before the patient becomes symptomatic. Careful attention to consistency of measurements to mitigate technical effects and individualised interpretation of results are critical to the assessment of cardiac remodelling as a sign of regurgitation severity. Serial echocardiograms should be performed every 12 months for asymptomatic patients with severe AR and LV end-diastolic size of 60–65 mm and normal LV systolic function. Echocardiograms should be performed more often (every six months) for patients with LV end-diastolic diameter close to 70 mm, or end-systolic diameter close to 50 mm (25 mm/m²). Additional echocardiograms are also required when symptoms occur (which represent a class I indication for surgery), worsening AR or increasing LV size is clinically suggested, or in patients with aortic root dilatation [17]. Alternatively, CMR with less variability than echocardiography is an option for longitudinal follow-up in individual patients.
Medical therapy (pharmacotherapy) in AR
Vasodilating drugs improve haemodynamic abnormalities in patients with AR and increase forward cardiac output. However, two small randomised controlled trials yielding discordant results did not conclusively show that these drugs alter the natural history (onset of symptoms) in asymptomatic patients with chronic severe AR and normal LV systolic function. Thus, vasodilator therapy is not recommended routinely in patients with chronic asymptomatic AR and normal LV systolic function.
In patients with hypertension (systolic blood pressure >140 mmHg), blood pressure control is warranted, preferably with dihydropyridine calcium channel blockers or angiotensin-converting enzyme inhibitors/angiotensin receptor blockers (ACEi/ARBs; Class I, LOE B) [8,9]. Beta-blockers (BB) may be less effective because the reduction in heart rate may be potentially harmful by prolonging the diastolic period during which AR occurs and are associated with an even higher stroke volume, which contributes to the elevated systolic pressure in patients with chronic severe AR. Conversely, a slower heart rate may improve left atrial emptying and myocardial blood flow.
In symptomatic patients who are candidates for surgery, medical therapy is not a substitute for AV surgery. Medical therapy can provide symptomatic improvement in individuals with chronic severe AR in whom surgery is not feasible. In patients who undergo surgery but continue to suffer from heart failure or hypertension, ACEi, ARBs and BBs are useful [8].
Patients with Marfan’s syndrome suffer from a gene mutation encoding the connective tissue protein fibrillin-1. Abnormal fibrillin results in higher signalling through the transforming growth factor-beta (TGF-ß) pathway, which may contribute to aortic medial degeneration responsible for aortic complications, such as dilation, dissection, and rupture. BBs and modulators of the angiotensin II pathway (ARBs) alone or in combination slow the progression of aortic dilatation to reduce the future possible risk of aortic complications. The pharmacologic effect of ARBs is believed to be based on suppression of TGF-β signalling at the cellular level, while a haemodynamic component with a reduced force of left ventricular ejection (contractility, blood pressure) leading to a diminished aortic wall shear stress is likely operative for BBs.
Given the histopathological similarities with Marfan’s syndrome, some clinicians have extrapolated the pharmacotherapy with antihypertensive agents in analogy to the treatment of aortopathy associated with BAV disease. However, trials of preventative pharmacotherapy for BAV-associated aortopathy are currently lacking.
Since the risk of bacterial endocarditis associated with invasive procedures compared to daily routine activities is felt to be low in patients with native valvular AR no matter what the cause, current European and US guidelines no longer suggest endocarditis prophylaxis, except in patients with a prior history of endocarditis.
Indications for interventions
Aortic regurgitation is a valvulopathy associated with some controversy with regard to the timing of intervention. According to the current ESC and US guidelines [8,9], the presence of symptoms is a class I indication for surgical intervention (Table 3). Asymptomatic patients with an EF less than 50% or those who require other cardiac surgeries also meet this class I indication for surgical replacement. The “50 rule” has been useful in gauging the timing of surgery in severe AR – that is, it ought to be performed before the EF falls below 50% or the end-systolic left ventricular dimension exceeds 50 mm.
Table 3. Indications for AV intervention for chronic severe AR.
|
Indication |
ESC 2017 |
ACC/AHA 2014 |
|---|---|---|
|
Symptomatic severe AR |
Class I, LOE B |
Class I, LOE B |
|
Asymptomatic severe AR and LV ejection fraction <50% |
Class I, LOE B |
Class I, LOE B |
|
Asymptomatic severe AR undergoing other cardiac surgery |
Class I, LOE C |
Class I, LOE C |
|
Asymptomatic severe AR, normal
LV ejection fraction >50% and severe
LV dilatation: ESD >50 mm or 25 mm/m² in patients with small body size
or EDD >70 mm
or EDD >65 mm with low surgical risk
|
Class IIa, LOE B
Class IIa, LOE B
Class IIa, LOE B
|
Class IIa, LOE B
Class IIb, LOE C
|
Class: class of recommendation; LOE: level of evidence
The ESC guidelines also suggest reporting the indexed LV end-systolic dimensions (ESD) to accurately identify chamber enlargement in individuals with small body size, specifically in women and elderly patients because non-indexed dimensions are less likely to meet surgical indications in this cohort. A recent study concluded that the risk of death in asymptomatic patients with severe AR increased gradually when LVESD reached 20 mm/m², a cut-off even lower than endorsed by current ESC guidelines [29].
AV surgery (replacement or repair)
The two surgical treatment options include AV replacement (using biological tissue or mechanical valve prostheses) or AV repair. Depending on the institutional surgical expertise, the so-called Ross procedure (native pulmonary valve harvested and moved as an autograft to the aortic position combined with a homograft placed in the pulmonary position) represents a relevant alternative. Another option for patients with severe AR and isolated involvement of the aortic root is the implantation of a composite valved conduit (modified Bentall procedure). AV repair is the second surgical treatment option. It includes subcommissural annuloplasty [30], valve-sparing aortic root replacement [31], valve-sparing root remodelling [32], external annuloplasty [33], LVOT suture [34] and root restoration [35] (Table 4).
Table 4. Technical characteristics of current aortic root replacement procedures and comparison with aortic root “restoration”.
|
Characteristics |
Subcommissural annuloplasty (Cabrol) [30] |
Reimplantation (David) [31]
|
Remodelling (Yacoub) [32]
|
Remodelling+ external annuloplasty (Lansac) [33] |
Remodelling+ LVOT suture (Schäfers) [34] |
Internal geometric annuloplasty (Rankin) [35] |
|---|---|---|---|---|---|---|
| Annular stabilisation | (+) | + | - | + | + | + |
| Preservation of annulus geometry | (+) | - | - | - | - | + |
| Preservation of aortic sinuses | + | (+) | + | + | + | + |
| Physiologic coronary blood flow | + | - | + | + | + | + |
| Predictable annular dimension | - | + | - | + | + | |
| Avoidance of deep root dissection | + | - | + | - | + | + |
Patients with BAV and moderate aortic dilatation continue to be at risk for future aortic complications, even after valve replacement, favouring concomitant AV surgery and replacement of the ascending aorta or aortic root in this subgroup, if the maximal aortic diameter is greater than or equal to 4.5 cm (Class IIa, LOE C) [8,9].
In patients with AR due to BAV, valve repair is effective and durable with an acceptable long-term survival [36]. However, leaflet calcification and tissue deficiency pose specific problems since no ideal material exists for use of cusp extension despite the availability of pericardial patch augmentation. Tissue-engineered patches or heart valves which attract livings cells may represent a potential solution to this problem [37]. In summary, long-term experience is confined to relatively few centres worldwide; and additional prospective studies will be necessary to promote BAV repair procedures. The so-called Ozaki-procedure (“aortic valve neocuspidisation” using autologous pericardium) represents a very promising novel technique for the surgical treatment of AR [38].
During the past decade, transcatheter aortic valve implantation (TAVI) has revolutionised the interventional treatment of aortic stenosis. For pure severe AR, there are no randomised clinical trials to date; and all the evidence we have comes from retrospective studies with heterogeneous populations. Currently, the availability of TAVI devices approved for the treatment of isolated AR is very limited. Therefore, TAVI is not yet a standard procedure, but currently an option for inoperable severe AR patients because it offers a better prognosis than optimal medical treatment. However, with technical advances, the number of patients suitable for this procedure as an alternative to surgical treatment will likely increase in the near future [39].
Non-cardiac surgery
Severe AR conveys an increased cardiac risk during non-cardiac surgery but is better tolerated than aortic stenosis. In asymptomatic patients with normal LV function, non-cardiac surgery can be performed with a low additional risk. Symptomatic patients and asymptomatic patients with severely impaired LV function are at a higher risk of cardiovascular complications and may benefit from optimisation of pharmacological therapy to produce maximal haemodynamic stabilisation prior to high-risk surgery. During anaesthesia and surgery, preload should be maintained because the LV has increased size and compliance; and excessive systemic afterload should be avoided to optimise forward cardiac output and reduce the regurgitation volume. In the absence of randomised controlled trials addressing perioperative management, patients with severe AR could be monitored postoperatively with invasive haemodynamics and echocardiography in an intensive care setting [8,9].
Future directions
Automated image interpretation is a branch of machine learning with the potential to extract and analyse a sheer amount of multidimensional imaging data that often goes unused [40]. In the future echocardiographic laboratory, computing technology will have a profound impact with the opportunity to standardise and accelerate analysis and assist in clinical decision making.
Conclusion
AR can result from an intrinsic abnormality of the AV leaflets (primary/organic) or dilation of the aortic root with incomplete leaflet coaptation (secondary/functional). Echocardiography to assess AR severity and its impact on the LV is the first-line diagnostic modality, complemented by CMR and CT in selected cases. In a digital world, a deep learning-based approach is able to recognise findings objectively in various imaging modalities in order to make or confirm a diagnosis. In the past, AV replacement has been the standard surgical procedure. With better understanding of the mechanisms of AR, valve-sparing surgical repair is emerging as a feasible and attractive alternative. With the development of new transcatheter devices, TAVI will no longer be restricted to inoperable patients but, in time, will become part of the therapeutic spectrum.