Overview
Accurate evaluation of aortic valve disease severity and aetiology for both aortic stenosis (AS) and aortic regurgitation (AR) is critical for appropriate management [1]. A multiparametric assessment involves analysing aortic valve anatomy, gradients, velocities, areas, regurgitant volumes and fractions. In addition, cardiac chamber size and function must be quantified, the severity of concomitant valve disease analysed, and aortic pathology interrogated [1]. Standard transthoracic echocardiography (TTE), including two-dimensional (2D) and Doppler imaging, is the first-line imaging modality to provide these essential data. However, patient-related factors and the inherent limitations of the modality may impair the evaluation [2, 3]. Accordingly, this review discusses the utility of advanced echocardiography techniques for aortic valve assessment (Table 1).
Table 1. Applications and limitations of advanced echocardiography techniques and other imaging modalities to evaluate aortic valve disease.
| TOE | 3D Echocardiography | CT | MRI | |
|---|---|---|---|---|
|
Aortic Stenosis |
Morphology and aetiology | Morphology and aetiology | Morphology | Morphology |
| Aortic valve planimetry | Aortic valve planimetry | Aortic valve calcium score | Aortic valve velocity | |
| Aortic valve gradients/velocities | Left ventricular quantification | Aortic annulus sizing | Aortic dimensions | |
| Aortic, coronary, and peripheral vascular evaluation | Thoracic anatomy | |||
| Thoracic anatomy | Left ventricular quantification | |||
| Tissue characterisation | ||||
|
Aortic Regurgitation |
Morphology and aetiology | Morphology and aetiology | Aortic dimensions | Regurgitant volume/fraction |
| Vena contracta | Vena contracta | Thoracic anatomy | Aortic dimensions | |
| Proximal isovelocity surface area | Proximal isovelocity surface area | Thoracic anatomy | ||
| Regurgitant volume and fraction | Regurgitant volume and fraction | Left ventricular quantification | ||
| Diastolic flow reversal (aorta) | Left ventricular quantification | Tissue characterisation | ||
|
Strengths |
Resolution for valve anatomy/aetiology | Resolution for valve anatomy/aetiology | Spatial resolution for aortic and coronary anatomy | Spatial resolution for function |
| Aortic valve planimetry | Aortic valve planimetry | Multiplanar reconstruction | Multiplanar reconstruction | |
| Intraprocedural guidance/assessment | Left ventricular quantification | Pre-TAVI workup | Left ventricular quantification (gold standard) | |
| Aortic valve gradients/velocities | Multiplanar reconstruction | Aortic, coronary, thoracic and peripheral evaluation | Regurgitant volume/fraction | |
| Aortic evaluation | ||||
| Tissue characterisation | ||||
|
Limitations/ Contraindications |
Invasive procedure | Suboptimal 2D images | Contrast administration | Availability, cost and length |
| Requires sedation/anaesthesia (affects loading | Radiation exposure | Contrast administration | ||
| Gastroesophageal pathologies/procedures | Arrhythmias (getting) | MRI-incompatible devices | ||
| Resolution for valve anatomy/aetiology | Claustrophobia | |||
| Resolution for valve anatomy/aetiology |
Two-dimensional transoesophageal echocardiography
If needed to further clarify the diagnosis, guidelines specifically recommend transoesophageal echocardiography (TOE) in two scenarios: to evaluate severe low-flow low-gradient AS and to assess AR of indeterminate severity [4]. In general, TOE is performed in moderate or severe aortic valve disease when TTE has suboptimal image quality, often with inconsistent or indeterminate estimates of valve disease severity and/or aetiology [5]. The superior spatial resolution of TOE assists in visualising aortic valve morphology, degree of stenosis, and abnormal pathologies such as calcification, prolapse, thrombus, endocarditis (where TOE is routinely performed to assess for vegetations and abscess), prosthetic valve dysfunction, as well as annular and aortic ectasia. The left ventricular outflow tract (LVOT) diameter can be measured from multiple mid-oesophageal views with greater precision, and colour Doppler identifies AR and flow acceleration. Furthermore, transgastric views are mandatory for measuring the peak velocity and velocity time integral (VTI) of the aortic valve and LVOT to calculate peak and mean aortic valve gradients, dimensionless index and aortic valve area [2]. With TOE, multiple levels of the aorta are also measured: from the sinus, sinotubular junction, ascending aorta, aortic arch and descending thoracic aorta. Moreover, TOE can identify aortic dissection and can also assess for diastolic aortic flow reversal, a marker of significant AR.
TOE also plays important roles in the intraoperative evaluation and guidance of aortic valve procedures [5, 6]. Immediately before and after cardiac surgery, the velocities and gradients across native or prosthetic aortic valves can be interrogated. In addition, aortic valve morphology is visualised, and the degree of AR, whether valvular or paravalvular (described in terms of origin on a clockface, severity and direction), is evaluated with colour Doppler [6]. Beyond the aortic valve, left and right ventricle size and function, other valves, and pericardial or aortic complications are all assessed by TOE. TOE had also traditionally offered the opportunity for intraprocedural guidance of transcatheter aortic valve implantation (TAVI) or closure of paravalvular leaks. However, with increasing experience, TTE has often supplanted TOE in this setting. In our institution, TOE is only used for TAVI when a non-transfemoral alternative access route is sought, whereas TTE is used for the majority of routine transfemoral cases.
The contraindications of TOE must be noted. Absolute contraindications include perforated viscus, oesophageal tumour, stricture, perforation or diverticulum, and active gastrointestinal bleeding [5]. Relative contraindications include dysphagia, Barrett’s oesophagus, oesophagitis, peptic ulcer disease, varices, a history of gastrointestinal surgery, as well as bleeding or radiation to the neck and mediastinum. As TOE requires a topical anaesthetic such as lidocaine and sedative drugs such as midazolam and fentanyl, medication and allergy history need to be obtained. If there are contraindications to TOE, and TTE does not provide a definitive answer, other imaging modalities should be pursued [4]. Notably, TOE is an invasive test with low but finite risks of major adverse events such as bleeding and perforation. The advantages and disadvantages of TOE and other imaging techniques for aortic valve disease are listed in Table 1.
Three-dimensional echocardiography
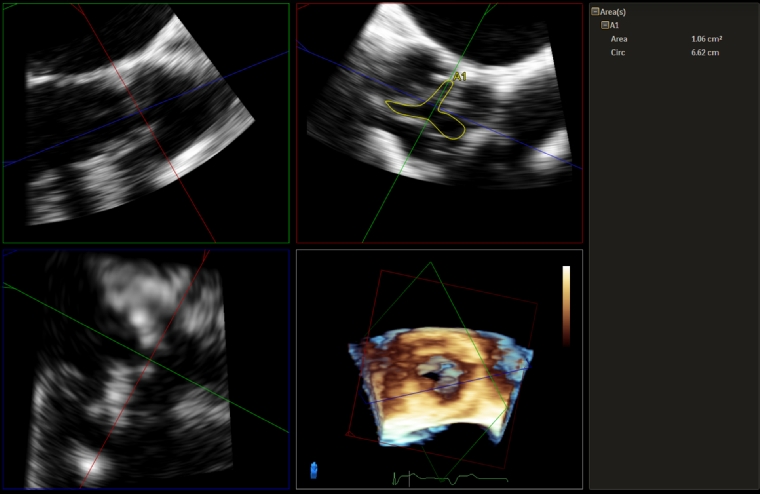
Three-dimensional (3D) echocardiography can be performed with both TTE and TOE, and the assessment of valvular heart disease is a key clinical application. The important views to acquire 3D images of the aortic valve are from the parasternal long and short axis on TTE, and mid-oesophageal long axis (120-140 degrees) and aortic valve short axis on TOE, both with and without colour [7]. Multiplanar reconstruction techniques are also available in 3D echocardiography to help with accuracy of measurements. For AS, 3D allows confirmation of AS aetiology such as calcific, degenerative or rheumatic, and clarifies both the location and extent of these pathologies. Direct planimetry of the aortic valve on 3D using multiplanar reconstruction is particularly useful (Figure 1), especially when 2D echocardiography does not provide the optimal aortic valve short-axis view. 3D TTE has high reproducibility and agreement with TOE, although this correlation is in part dependent upon the quality of 2D TOE views [8]. In addition, 3D is especially helpful in measuring the dimensions of the LVOT, which is a major potential source of error in the continuity equation calculation for aortic valve area. The suboptimal accuracy of 2D LVOT and aortic valve area measurements is a result of calculating the cross-sectional area from a single diameter and erroneously assuming that this area is circular [9]. In fact, in a recent meta-analysis, there was good correlation between the LVOT area, aortic annular area, perimeter and diameter measured on 3D TOE compared to computed tomography (CT), which is critical in assessing AS severity and preprocedural planning for TAVI [10]. Although CT has largely replaced TOE for TAVI prosthesis sizing, TOE remains an alternative strategy, especially when CT is contraindicated.
In AR, 3D echocardiography provides clear depiction of aortic valve morphology and defines the pathology, including prolapse, endocarditis and also prosthetic valvular and paravalvular AR [6, 7]. These may have important implications for planning subsequent intervention strategies, for example valve-in-valve transcatheter aortic valve replacement (TAVR) for valvular AR and AS versus percutaneous closure for paravalvular AR. In addition, TOE provides a quantitative assessment of AR severity. Both vena contracta (VC) and proximal isovelocity surface area (PISA) can be assessed with 3D using orthogonal planes for accurate planimetry. The PISA radius is subsequently used to calculate effective regurgitant orifice area, regurgitant volume and regurgitant fraction [3, 7]. This assessment of AR is more accurate compared to 2D methods, especially in the setting of eccentric or multiple AR jets [11]. In clinical practice, however, both 3D and 2D quantification of AR can be technically challenging and data are therefore complementary.
When evaluating aortic valve disease, assessment of the left ventricle by 3D echocardiography is also mandatory. Both left ventricular dimensions and left ventricular ejection fraction (LVEF) reflect the severity of aortic valve disease and inform indications for valvular intervention [1]. Typically, AS results in pressure overload of the left ventricle with subsequent concentric hypertrophy, whereas AR and resultant volume overload leads to dilation, and progression of both lesions eventually leads to impaired systolic function. The 4-chamber view is the standard view to acquire 3D volumetric assessment of the left ventricle in both TTE (apical) and TOE (mid-oesophageal) [7]. 3D measurement of left ventricular volumes and systolic function has several advantages, including lack of geometric assumption, making it more accurate when chamber shape is irregular and distorted, not being affected by foreshortening, and greater reproducibility. However, 3D volumes are reliant on image quality, and there are less data available for normative ranges, prognostic value, and optimal thresholds for intervention [12]. In one meta-analysis, 3D volumes by echocardiography have good correlation with the gold standard, magnetic resonance imaging (MRI), although there is systematic underestimation by about 10 ml for end-diastolic volume and 5 ml for end-systolic volume but very similar LVEF (<0.2% discrepancy) [13].
Strain echocardiography
Strain with echocardiography measures myocardial deformation using speckle tracking and, although it can be obtained for all the cardiac chambers and in different directions and segments, left ventricular global longitudinal strain (LVGLS) is the most widely studied [14]. LVGLS is a reproducible and sensitive measure of subclinical and early left ventricular systolic dysfunction with many applications, including in aortic valve disease. For AS, a meta-analysis of 10 studies and 1,067 patients with preserved LVEF found an association of LVGLS and mortality with a hazard ratio of 2.62. The best cut-point for discrimination was -14.7% [15]. In AR, our institution has shown that LVGLS predicts higher mortality with a hazard ratio of 1.62 and a best cut-point of -19%. Worsening LVGLS or persistently abnormal LVGLS after surgery is also associated with higher mortality [16]. Collectively, these findings suggest an additional prognostic value of LVGLS beyond LVEF and potentially identifies aortic valve disease patients who would benefit from earlier intervention. Accordingly, routine LVGLS assessment is encouraged in these patients.
Stress echocardiography
The two main stress echocardiography modalities, exercise (ESE) and dobutamine (DSE), both have important roles in the evaluation of aortic valve disease. During ESE, the presence of symptoms or a fall in blood pressure below baseline is an indication for surgery in patients with severe AS, and ESE may also unmask symptoms in AR [1]. Abnormal ESE including impaired exercise capacity, decline in LVEF, lack of contractile reserve, and pulmonary hypertension is associated with cardiovascular events, whereas a normal test supports delaying surgery [17]. Conversely, DSE plays a critical role in the assessment of low-flow (stroke volume index <35 ml/m²) AS and impaired LVEF <50%. DSE differentiates severe AS, characterised by a mean gradient exceeding 40 mmHg with an aortic valve area remaining <1.0 cm² after reaching target, from pseudosevere AS, characterised by an aortic valve area >1.0 cm² [2]. With DSE, the lack of contractile reserve makes the test inconclusive, but it is a poor prognostic factor. Importantly, severe symptomatic aortic stenosis, acute coronary syndrome, uncontrolled hypertension, heart failure and tachyarrhythmias are contraindications for stress echocardiography.
When it doesn’t add up? Role of multimodality non-invasive imaging
Cardiac computed tomography
Cardiac CT can provide important information in aortic valve disease, especially in AS. Aortic valve morphology and calcification can be routinely detected on CT. The calcium score, derived from CT, contributes to the assessment of severity (Agatston score severe AS likely: men >2,000 and women >1,200; unlikely: men <1,600, women <800). This assessment is particularly useful when echocardiographic findings are conflicting or equivocal, and is part of the AS diagnosis algorithm in guidelines [2]. Furthermore, CT is now considered mandatory in the preprocedural evaluation of TAVR, the preferred modality for evaluation of aortic annulus size and shape, number of cusps, degree of calcification, coronary ostia height from annulus, atherosclerotic burden and aortic dimensions, peripheral vessels for arterial access, and occasionally coronary artery disease as an alternative to invasive coronary angiography [4, 18]. Multiplanar reconstruction is central for accurate measurements of these dimensions. The main limitations include need for iodinated contrast, radiation exposure, cost and challenges with image acquisition in unstable patients.
Cardiac magnetic resonance imaging
Cardiac MRI provides further complementary information to echocardiography in aortic valve disease, particularly in AR. Occasionally, chamber quantification informs the need for intervention [1]. Flow can be measured on phase contrast velocity mapping sequences in a plane perpendicular to the structure of interest such as the aortic valve, so that direct measurements of regurgitant volumes and fractions in AR can be calculated [3]. This assessment is particularly useful when there are suboptimal echocardiography windows or discordance between echocardiographic parameters and clinical assessment. Aortic dimensions and thoracic anatomy can also be evaluated similarly to CT [18]. Limitations include availability, cost, length of study, cardiac devices particularly if MRI is incompatible, claustrophobia and challenges with image acquisition in unstable patients.
Conclusions
TOE and 3D echocardiography can provide additional valuable information in the assessment of aortic valve disease, both in anatomic definition and quantitative assessment of valvular lesions. In comparison to 2D TTE, correlation is good, and accuracy is often improved. Stress echocardiography is useful in identifying exercise response and symptoms, as well as evaluating low-flow AS using dobutamine. Left ventricular global longitudinal strain provides additional prognostic value to consider aortic valve intervention beyond LVEF. In situations where findings are equivocal or conflicting, other imaging modalities such as CT for AS and MRI for AR are helpful to reconcile and determine valve severity.