Keywords
Renal denervation, hypertension, blood pressure, sympathetic nervous system, kidney
Pathophysiological aspects
First, we begin with a few words about the pathophysiological mechanisms associated with renal denervation (RDN). RDN affects both afferent sensory and efferent sympathetic nerves: it inhibits the efferent pathway that effects tubular reabsorption of water and sodium and increases vascular resistance and renin release [1,2]. This reduction of the efferent sympathetic nerve activity to the kidney, indicated by a decrease of renal noradrenaline release in experimental studies, represents one part of the blood pressure (BP)-lowering mechanisms of RDN [3]. On the other hand, afferent sensor signalling to the central sympathetic nervous system is reduced after RDN, causing a decrease in sympathetic nerve activity of the central nervous system and thereby also to various organs such as peripheral resistance vessels, the heart and the kidneys [4,5].
The history of RDN - initial rise and fall
The history of RDN dates back to the year 1953, when the method of splanchnicectomy – surgical removal of renal nerves – was reported as a treatment option in patients with severe primary hypertension [6]. In the 1990s, interventional techniques with radiofrequency were developed and proof-of-concept studies found significant reductions in BP after RDN [7-9].
The RDN procedure itself was performed in the catheterisation lab by means of delivering either radiofrequency energy, ultrasound energy or neurotoxic substances (alcohol), without any clear evidence at the moment that any one method is superior to another [10-12].
The first sham-controlled trial, the SYMPLICITY HTN-3 study, was initiated in 2014. The results were disappointing since no significant difference in BP reduction was observed between RDN and sham procedure [13]. A thorough workup of the study results revealed several aspects which certainly contributed to the negative outcome of the study. First, the results of the SYMPLICITY HTN-3 study have to be interpreted in the context of a rather heterogeneous study group, e.g., one fourth of patients in the treatment arm and nearly one third of patients in the control group were of African-American ethnicity, in whom a different pathophysiological mechanism of primary arterial hypertension is presumed compared to Caucasians. When analysing only the group of Caucasian subjects, a significant difference between RDN and sham procedure in favour of the former could be detected. Other factors that were not accounted for were poorly and not thoroughly controlled adherence to medication during the study, early and uncontrolled changes in antihypertensive medication after RDN treatment as well as the wide range of antihypertensive medication at the time of study inclusion. Besides, a large number of study centres participated with a low level of experience in performing the RDN procedure. For example, only in 19 out of 340 procedures were all four quadrants of the renal artery at both sides successfully treated as generally recommended [14]. All these aspects contributed to the negative results of the study in terms of efficacy but provided evidence of the safety of the RDN procedure. Overall, a sharp decrease in euphoria developed concerning the role of RDN as a valid and effective interventional treatment option in patients with hypertension.
The revival of RDN – learning from mistakes
Since then, based on the experience gained from the SYMPLICITY HTN-3 study, several new trials (generation 2.0) have been initiated to re-evaluate the method with improved study methodology and with more homogeneous study populations (Table 1) [15].
Table 1. Characteristics of the SPYRAL HTN-ON MED, HTN-OFF MED and RADIANCE-HTN SOLO RDN studies.
| Characteristics | SPYRAL HTN-ON MED | SPYRAL HTN-OFF MED | RADIANCE-HTN SOLO |
|---|---|---|---|
| Number of patients | 80 | 80 | 146 |
| Design | sham-controlled | ||
| Antihypertensive medication | 1-3 | - | - |
| Method | radiofrequency | radiofrequency | radiofrequency |
| Catheter | Symplicity SpyralTM | Symplicity SpyralTM | Paradise® |
| Primary endpoint | Change in 24-hr ABPM after 6 months | Change in 24-hr ABPM after 3 months | Change in systolic daytime 24-hr ABPM after 2 months |
24-hr ABPM: 24-hour ambulatory blood pressure monitoring
In this context, the results of three randomised, sham-controlled studies have been published in 2018. The results were encouraging, since they consistently delivered a clear biological signal that RDN decreases BP in patients with primary hypertension (Table 2).
Table 2. Study outcomes in systolic 24-hour ABPM and systolic office BP.
| Parameter | SPYRAL HTN-ON MED | SPYRAL HTN-OFF MED | RADIANCE-HTN SOLO |
| Mean difference in systolic 24-hr ABPM after 6/3/2 months* |
-7.4 mmHg (-12.5 to -2.3) p=0.0051 |
-5.0 (-9.9 to -0.2) p=0.0414 |
-6.3 mmHg (-9.4 to -3.1) p=0.0001 (daytime) |
| Mean difference in systolic office BP after 6/3/2 months* |
-6.8 mmHg (-12.5 to -1.1) p=0.0205 |
-7.7 mmHg (-14.0 to -1.5) p=0.0155 |
6.5 mmHg (-11.3 to -1.8) p=0.007 |
* Mean baseline-adjusted difference between RDN and sham procedure.
24-hr ABPM: 24-hour ambulatory blood pressure monitoring; BP: blood pressure
The SPYRAL HTN-ON MED study, including subjects on one, two or three antihypertensive drugs who received either RDN by means of radiofrequency ablation or sham procedure in a 1:1 ratio, showed a significant difference of -7.4 mmHg in 24-hour ambulatory and -6.8 mmHg in office systolic BP in favour of RDN six months after treatment [10].
In addition, there were two studies including drug-naïve subjects evaluating the pure effect of RDN independent of antihypertensive drug treatment - the SPYRAL HTN-OFF MED and the RADIANCE-HTN SOLO study. In the former, subjects were randomised 1:1 to either RDN or sham procedure and – similar to the HTN-ON MED study – RDN was performed applying radiofrequency energy. After three months of follow-up, the investigators observed a difference of -5 mmHg in 24-hour ambulatory BP and a difference of -7.7 mmHg in systolic office BP, also in favour of RDN [12].
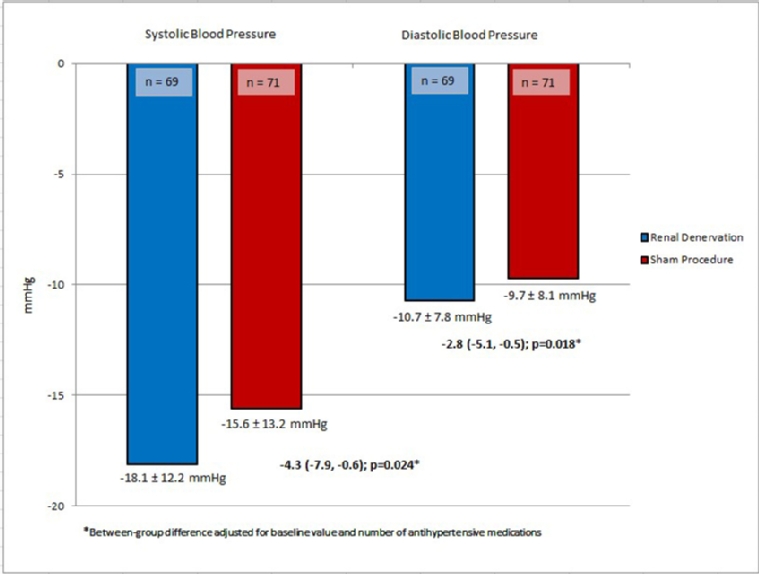
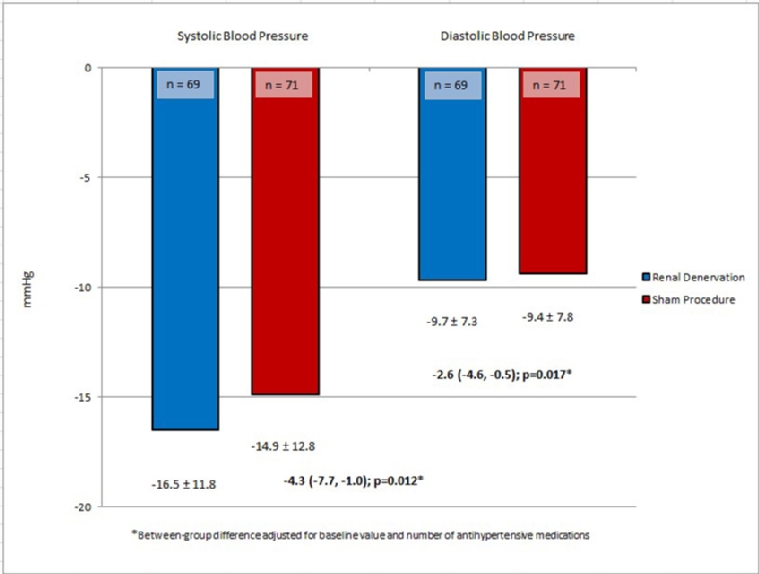
In the above-mentioned RADIANCE-HTN SOLO study, subjects were also randomised in a 1:1 ratio to either RDN or sham procedure, but the RDN procedure was performed by means of a catheter system based on ultrasound energy. After two months, there was a difference of -6.3 mmHg in daytime ambulatory BP and of -6.5 mmHg in office systolic BP in favour of RDN [1]. Recently, the six-month results of this study were published. In addition to a now confirmed sustained BP-lowering effect, subjects in the RDN group were prescribed significantly fewer antihypertensive drugs than subjects in the sham group (Figure 1) [16].
Figure 1A. Change in daytime ambulatory blood pressure from baseline to 6 months.
Figure 1B. Change in 24-hour ambulatory blood pressure from baseline to 6 months.
Taken together, RDN led to a decrease of about 10 mmHg in office BP from baseline and 6 mmHg in 24-hour ambulatory BP compared to the sham group. Thus, although the above-mentioned studies finally provided evidence that RDN is an effective treatment option for hypertension, without any safety signal, several unanswered questions remain. Which RDN procedure is most effective? Which hypertensive patients are the ones who profit most from RDN [16]? The clinical relevance of these results can be estimated from previous meta-analyses [17,18]. A decrease of 10 mmHg in office BP is related to a risk reduction for cardiovascular diseases of -17%, heart failure of -28%, cardiovascular event rate of -20% and total mortality of -13% [19]. This was confirmed by another extensive meta-analysis of trials with hypertensive patients only [20].
The future of RDN - perspectives
The renaissance of RDN as a valid treatment strategy for treating primary hypertension, complementary and in addition to lifestyle modification and drug therapy, offers many exciting perspectives.
While RDN was initially only considered as a last resort for patients with treatment-resistant severe hypertension, data from the above-mentioned studies indicate that the intervention may also gain importance when treating patients with uncomplicated hypertension. In this regard, patient preference might probably become a more relevant factor in terms of the treatment decision in a shared decision-making process. A cross-sectional survey of 1,000 patients with hypertension in Germany revealed that about one quarter would choose RDN instead of another pill [21].
With renal sympathetic nerves playing a crucial role not only in the pathogenesis of hypertension but also in other cardiovascular processes such as heart failure, cardiac arrhythmias and chronic renal failure, RDN could become even more than solely a treatment option for hypertension [17]. In particular, since increased sympathetic nerve activity contributes to the progression of chronic kidney diseases, RDN might become a new treatment option in this group of patients, as RDN decreases two progressive factors of chronic kidney disease at the same time, namely hypertension and increased sympathetic tone to the diseased kidneys.
However, some questions still remain to be answered. One point that has to be investigated further is which method of RDN - radiofrequency energy, ultrasound energy or alcohol injection - is the safest and most effective one. Furthermore, it would be of importance to identify predictors of patient BP-lowering response. Finally, there remains the question of whether functional re-innervation might occur after RDN treatment, as has already been found after heart transplantation.
Conclusion
RDN has experienced several rises and falls during its development. The results of the latest RDN studies are promising and finally provide evidence that RDN is effective in lowering BP in hypertensive patients. Ultimately, RDN may become another pillar of treating patients with hypertension, aside from drug therapy and lifestyle modification. Nevertheless, there are still aspects that have to be examined in detail in currently ongoing sham-controlled randomised studies prior to recommending the application of RDN in daily clinical practice.