Introduction
Anne is an 83-year-old female with a long history of hypertension, heart failure (HF), dyslipidaemia, chronic kidney disease (CKD) stage 4 (last estimated creatinine clearance 20 ml/min) and vascular dementia. Fifteen years ago, in 2004, she had numerous episodes of paroxysmal atrial fibrillation (AF) and was therefore started on warfarin. Anticoagulation was interrupted two years later for “bleeding diathesis”, gastrointestinal bleeding most likely attributable to diverticulosis, and she was switched to acetylsalicylic acid (ASA). In 2018, she was admitted to the Cardiology Unit of our hospital due to ventricular tachycardia, that resolved with electrical cardioversion. During this admission, coronary angiography revealed multivessel coronary artery disease and she underwent multiple angioplasties with implantation of four drug-eluting stents (DES). The patient tolerated the procedure well and was discharged home on clopidogrel and rivaroxaban. At the one-month follow-up visit, she reported several episodes of accidental fall at home and great difficulty in managing her therapy. She appeared confused regarding the type and the number of pills to take daily, with no care-giver available to assist her. What is the best course of action in a case scenario like this? In the light of her overall clinical and social history, is oral anticoagulation (OAC) really indicated for this patient?
This clinical case is characterised by several critical issues that contribute to making OAC extremely challenging in subjects older than 80 years of age. This subset of patients is becoming more and more prevalent in hospital departments and outpatient clinics due to the progressive ageing of the global population, with the proportion of individuals aged ≥85 years being expected to increase threefold worldwide by the year 2035 [1]. The prevalence of AF rises steadily from 0.4-1% among the general population, and to 8% by age 80 years [2]. Stroke risk from AF increases exponentially with age, with an estimated 1.45-fold increase for each subsequent decade [3], reaching an annual risk as high as 23.5% in patients with AF aged 80 to 90 years [4]. Compared with non-AF-related strokes, AF-related strokes are almost twice as likely to be fatal and to cause severe disability in survivors, increasing the length of hospital stay and reducing the likelihood of patients returning to their own home, with associated significantly higher mean direct costs per patient [5].
Adherence to stroke prevention guidelines and OAC can significantly reduce the incidence and severity of cerebrovascular accidents. Emerging data from clinical trials and real-world registries demonstrate that the benefits of anticoagulation compared to no therapy are maintained also in elderly patients, with a clinical advantage of non-vitamin K antagonist oral anticoagulants (NOACs) compared to conventional anticoagulation with vitamin K antagonists (VKAs). However, many physicians raise concerns about whether these favourable results can be reproduced in the elderly population in daily clinical practice, due to the burden of associated comorbidities and additional risk factors. The presence of CKD with related decreased renal clearance of medications, multiple drug therapy, poor nutritional status with low body weight, and a higher chance of falls are all factors that increase the risk of both stroke and bleeding in older patients.
In this paper we will review the risk/benefit profile of different anticoagulation strategies implemented for prevention of thromboembolic risk in elderly patients with AF, we will offer some insights on the reluctance in prescribing OAC in clinical practice, and we will suggest a protocol that can assist in the management of these challenging patients.
Before the era of NOACs
Before the advent of NOACs (2011), the conventional anticoagulation therapy for very old patients with AF at risk of arterial thromboembolic complications was VKAs, with the target international normalised ratio (INR) value ranging between 2.0 and 3.0. Compared to no treatment, warfarin decreases the risk of stroke by approximately 2/3 and death by 1/4, whereas antiplatelet therapy decreases stroke risk by only 22% [5]. It is significant to note that it has been reported that ASA has no discernible protective effect against stroke and is associated with more serious adverse events compared with warfarin in a small randomised trial of octogenarians with AF [6]. For the prevention of strokes, VKAs’ superior efficacy compared to antiplatelet therapy is proven, even when clopidogrel is added to aspirin [7]. In subjects with AF aged 75 years or older, treatment with warfarin is associated with a 52% reduction in the risk of fatal or disabling stroke, intracranial haemorrhage (ICH), or clinically significant arterial embolism, compared with aspirin 75 mg daily [8]. These benefits were also confirmed for patients older than 80 years, who experienced a reduction in cardiovascular events with VKAs compared to aspirin after a one-year follow-up period (6% vs. 13%; p<0.01) [7]. Despite compelling evidence of the benefits of warfarin on stroke prevention, observational studies show that during the last decade there has been a high rate of under-prescription due to its well-known drawbacks, i.e., unpredictable pharmacodynamics and pharmacokinetics, narrow therapeutic window, and numerous drug-drug and food-drug interactions, requiring frequent laboratory monitoring and dose adjustments. Observational studies have shown that worldwide time in therapeutic range (TTR) is suboptimal, with the lowest TTR associated with the greatest risk of stroke and bleeding [9]. The major concern of VKA therapy, however, is the risk of ICH, the most devastating form of haemorrhage, which rises strikingly at the age of 80 and beyond, especially in the first month of therapy. Moreover, older age is associated with greater risk of traumatic ICH, due to a greater risk of falls.
The bleeding risk in the elderly with AF is therefore often perceived as inappropriately high, so that OAC is thought to be not indicated by clinicians or is refused by patients. However, a well-managed warfarin treatment - based on vigilance, adherence and reduction of modifiable haemorrhagic risk factors - is still beneficial, with an acceptable safety profile. A higher risk of accidental falls in very old individuals is not a reason to avoid OAC, since it has been shown that AF patients at higher risk of stroke would have to fall >295 times/year before the risk of traumatic ICH would exceed the risk of ischaemic stroke; yet, in older persons who fall, the mean number of falls is only 1.8/year [10].
The era of NOACs
According to current guidelines, NOACs, including the direct thrombin inhibitor dabigatran, and the direct factor Xa inhibitors rivaroxaban, apixaban, and edoxaban, are recommended over VKAs in patients with non-valvular atrial fibrillation (i.e., AF in the absence of mechanical prosthetic heart valves and in the absence of moderate to severe mitral stenosis) [11,12]. NOACs have an improved efficacy/safety ratio, predictable effect without the need for routine blood level monitoring, and fewer food and drug interactions compared with VKAs. The phase III NOACs trials have shown that these drugs are associated with an approximately 12% decrease in mortality compared to warfarin, a 20% decrease in stroke or systemic embolisation, and - most impressively - a 50% reduction in ICH, though there is an increased incidence of gastrointestinal bleeding [13].
Despite the absence of clinical trials that specifically assess the efficacy and safety of NOACs in elderly people, the size of the patient group older than 75 years enrolled in the studies supports the conclusion that trial results will also apply to older patients [14]. In addition, it should be considered that the absolute risk reduction obtainable will be higher in elderly than in younger patients, due to the increased overall risk in the former group, preserving the favourable balance between risks and benefits of treatment demonstrated in younger individuals [15,16].
Remarkably, the bleeding profile of NOACs is more favourable than that of warfarin. This is true in particular for intracranial and other life-threatening haemorrhagic events. Not only is there non-inferiority or even superiority for the overall incidence of bleeding, but the outcome of bleeding complications under NOAC treatment has also been demonstrated to be more benign compared to bleedings that develop during VKA treatment [12]. Also, fewer bleeding events under NOAC therapy will lead to fewer changes or interruptions in anticoagulant therapy, with subsequent reduction in early and late mortality.
The benefit from NOAC therapy (best shown for edoxaban and apixaban) is evident also in frail very old patients; thus, frailty and an increased risk of falling per se should not be considered exclusion criteria for anticoagulation [17]. Nevertheless, as more patients will start using one of the NOACs, the number of bleeding-related events is expected to increase [14].
Older patients and comorbidities
In the elderly patient, the balance between thrombotic and haemorrhagic risk is strongly affected by the presence of comorbidities; we will focus our attention on CKD, cancer and coronary heart disease (CAD), which are the most challenging scenarios encountered in clinical practice.
CKD
A retrospective cohort study assessing the safety of NOACs in patients older than 75 years managed in a single healthcare system has found that all major bleeding episodes were associated with a decline in glomerular filtration rate (GFR) compared to baseline, highlighting the importance of monitoring renal function regularly, at least once every six months according to current guidelines [12]. This is even more critical in the very elderly among whom the prevalence of CKD is 25-30% and can even exceed 50% in those with associated cardiovascular and metabolic diseases [18].
NOACs are metabolised mainly by the kidneys; consequently, more frequent monitoring should be considered in subjects with reduced or variable renal function and if acute conditions develop that can impact on an already impaired baseline GFR. Appropriate NOACs prescription and dosage adjustment according to renal function can significantly reduce the risk of bleeding. In patients with CKD stage 4, i.e., CrCl 15-30 mL/min, rivaroxaban, apixaban, and edoxaban are approved in Europe at a reduced dose regimen, even though current ESC guidelines recommend against their administration in such a clinical condition [11][12] (Table 1). Patients with end-stage renal disease, i.e., CrCl <15 mL/min, are not eligible to receive NOACs, but can be treated with VKA. However, clinicians should be aware that warfarin therapy in patients with CKD has been associated with possible development of vascular calcification, faster progression of renal dysfunction and the potential for supratherapeutic INRs, and low bone density [13].
Table 1. Dose adjustment for dabigatran, rivaroxaban, apixaban, edoxaban and warfarin according to age, renal function, weight and hepatic function.
| CLINICAL OBSERVATION | DABIGATRAN | RIVAROXABAN | APIXABAN | EDOXABAN | WARFARIN |
|---|---|---|---|---|---|
| AGE | <75 years: 150 mg b.i.d. 75-80 years: 150 mg b.i.d. (110 mg b.i.d. should be considered when the risk of stroke is low and the bleeding risk is high). >80 years: 110 mg b.i.d. | No dose adjustment is required. | Recommended dose: 5 mg b.i.d. 2.5 mg b.i.d. in case of at least 2 of the following characteristics: age ≥80 years, body weight ≤60 kg, or serum creatinine ≥1.5 mg/dL. No dose adjustment is required based on age, unless criteria for dose reduction are met. | No dose adjustment is required. | Consider lower initiation and maintenance doses of warfarin in patients ≥60 years. |
| RENAL FUNCTION | CrCl 30-50 mL/min: recommended dose is 150 mg b.i.d. (110 mg b.i.d. for patients with high risk of bleeding). CrCl <30 mL/min: contraindicated. | CrCl 15-49 mL/min: 15 mg o.d. CrCl <15 mL/min: not recommended. | Recommended dose: 5 mg b.i.d. No dose adjustment is necessary in patients with mild or moderate renal impairment, unless criteria for dose reduction are met. CrCl 15-29 mL/min: 2.5 mg b.i.d. CrCl <15 ml/min or dialysis: not recommended. | CrCl 15-49 mL/min: 30 mg o.d. CrCl <15 mL/min: not recommended | No dose adjustment required. |
| WEIGHT | No dose adjustment is necessary based on body weight. However, close clinical follow-up is required for patients with body weight <50 kg. | No dose adjustment is necessary based on body weight. | No dose adjustment is required based on body weight, unless criteria for dose reduction are met. | Body weight >60 kg: 60 mg o.d. Body weight ≤60 kg: 30 mg o.d. | Lower initial dose (2.5-5 mg) is recommended if weight <60 kg. |
| HEPATIC FUNCTION | Administration in patients with moderate hepatic impairment showed large inter-subject variability but no evidence of consistent change in exposure. | Avoid use in patients with Child-Pugh B and C hepatic impairment, or any degree of hepatic disease associated with coagulopathy. | Not recommended in patients with severe hepatic impairment. Dosing recommendation cannot be provided in patients with moderate hepatic impairment. | Not recommended in patients with moderate or severe hepatic impairment. | Caution needed in patients with moderate-to-severe hepatic impairment. |
Cancer
As cancer is more common among older individuals, it is not unusual to be faced with an elderly patient with AF associated with an oncological disease. Given that cancer patients are at elevated risk of both thrombotic and bleeding complications, the decisions about whether to initiate OAC, and which anticoagulant to use, are not easy ones.
Among cancer patients with AF, use of warfarin is complicated by the possibility of metabolic interactions with chemotherapeutic and antibiotic agents, by chemotherapy-induced thrombocytopaenia, and by the frequent need for invasive procedures or surgery. Despite a lack of data coming from randomised clinical trials, observational studies show that the use of NOACs is not rare among cancer patients with AF, with evidence of lower or similar rates of bleeding and strokes compared with warfarin users, and lower rates of venous thromboembolic complications [19]. However, caution must be exercised with chemotherapeutic and antimicrobial agents that can modify the metabolism of NOACs through P-glycoprotein (P-gp) and CYP3A4-type cytochrome interaction.
CAD
The clinical scenario in which percutaneous coronary intervention (PCI) is necessary in patients with CAD and concomitant AF poses a common treatment dilemma concerning the selection of an effective and safe antithrombotic strategy. Current guidelines and preliminary data coming from ongoing prospective trials suggest that triple therapy (TT) - the concurrent use of an oral anticoagulant and dual antiplatelet therapy - is associated with an early reduction in thromboembolic and mortality risks in patients ≥75 years at the cost of a progressive increase in the risk of major bleeding. Thus, an accurate assessment of thromboembolic and haemorrhagic risk for each individual patient is of the utmost importance. The use of TT in elderly patients with AF undergoing PCI is not discouraged, despite increasing the rate of bleeding events [20].
Discussion
Age remains one of the strongest risk factors for stroke in patients with AF. However, real-world data suggest that a significant proportion of older patients are still not receiving stroke prophylaxis treatment in line with guideline recommendations, even in the absence of contraindications to OAC [21]. A possible explanation for this persistent reluctance may be that the very old patient requires a comprehensive time-consuming assessment, that includes looking for the possible presence of major geriatric syndromes, evaluation of social conditions and review of the family support system. Despite compelling evidence that associated risk factors should not always be considered exclusion criteria for OAC, their presence influences the management of anticoagulant treatment and can induce clinicians to underestimate its net clinical benefits.
Currently, there are no scoring systems specifically developed and validated for guiding OAC in the elderly. Guideline-recommended scoring systems used to assess ischaemic and haemorrhagic risks have many limitations, especially when applied to the very old patient. In particular, the HAS-BLED score does not provide a specific assessment of the risk of ICH, which is possibly the greatest concern in the older population. At least 80% of predicted major bleeds are extracranial haemorrhages (especially gastrointestinal) that carry low mortality rates (<5-6%), are rarely the cause of disability, and are easily manageable. Therefore, AF patients in whom the risk of ICH plus fatal or disabling extracranial haemorrhage is likely to exceed the risk of fatal and disabling ischaemic stroke cannot be identified through risk scores presently recommended by guidelines.
We believe that it would be beneficial to develop a new scoring system dedicated to the elderly patient with AF that should take into account all the risk factors specific to this population subset. This would lead to a more rational and standardised approach to this subset population (Figure 1).
FIGURE 1. Proposed algorithm to evaluate the very old patient with AF and validated scores available.
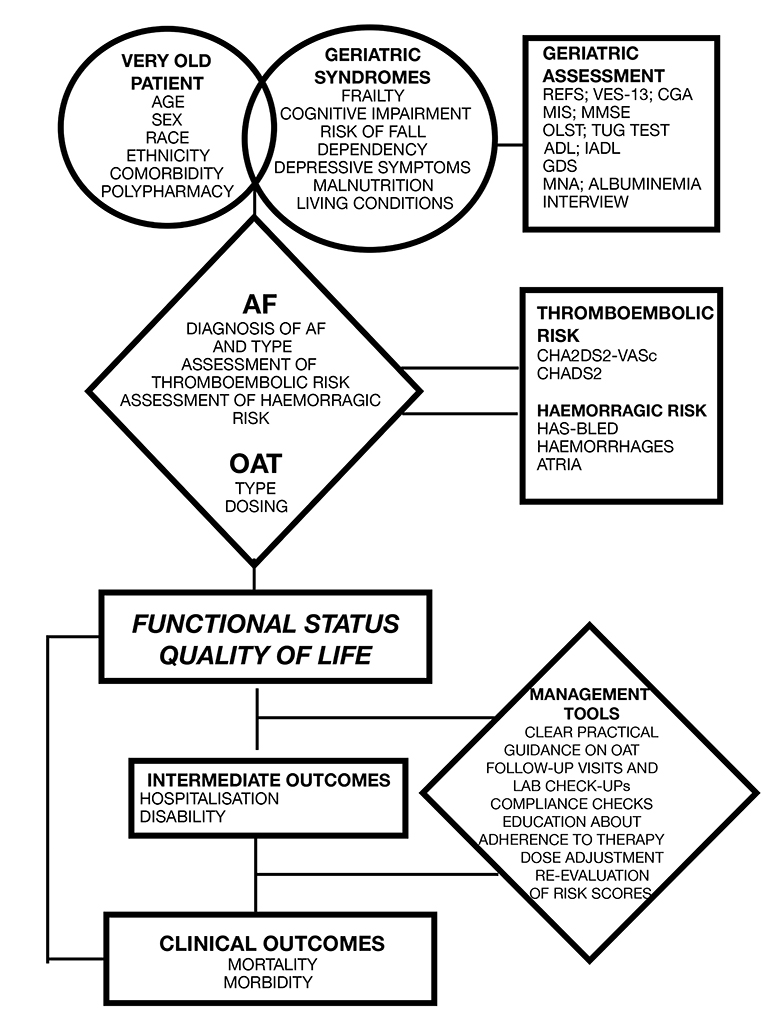
REFS: Reported Edmonton Frail Scale; VES-13: Vulnerable Elders Survey; CGA: Comprehensive Geriatric Assessment; MIS: Memory Impairment Screen; MMSE: Mini Mental State Examination; OLST: One Legged Stance Test; TUG: Timed Up and Go; ADL: Activity of Daily Living; IADL: Instrumental Activity of Daily Living; GDS: Geriatric Depression Scale; MNA: Mini Nutritional Assessment
Moreover, risk is not static and, particularly in the case of bleeding, many potentially dangerous conditions can be acted upon. To improve the net clinical benefit and the safety of OAC, efforts should be made in recognising early and minimising the impact of each modifiable risk factor (e.g., inadequate blood pressure control, deteriorated renal function, presence of haematologic disorders, suboptimal nutritional status, lack of social support) through an accurate initial assessment and appropriately scheduled follow-up. Close periodic re-evaluation of the very old patient is essential due to frequent changes of clinical status, and in order to assess treatment adherence, to verify the need for dose adjustment, and to educate the patient on a continuing basis about the importance of OAC.
Advances in the management of OAC, in association with the improved safety performance of NOACs, will boost clinicians’ confidence in prescribing anticoagulant therapy even in the most challenging scenarios, when evidence-based data predict a net clinical benefit.
Conclusions
In conclusion, except when there is an unacceptably high risk of major bleeding, anticoagulation is required in most elderly patients with AF to prevent the risk of stroke. NOACs have been shown to be associated with the best efficacy and safety profiles compared to VKAs in very old patients with non-valvular atrial fibrillation.















