Epidemiology and burden of atrial fibrillation with respect to the old/very old population
Prevalence
The increased prevalence of non-valvular atrial fibrillation (NVAF) among the elderly is well recognised [1]. In the last two decades, NVAF has become one of the most important public health issues and represents an important cause of healthcare expenditure in western countries [2]. NVAF influences quality of life significantly as a result of its anatomic, haemodynamic, and haemocoagulation consequences. In addition, NVAF is frequently associated with disturbing symptoms and critical socioeconomic problems, such as permanent disability, cognitive disturbance, and hospitalisation. Because of comorbidities and frailty, NVAF has a more negative impact on the health of elderly patients compared to young patients. The prevalence of NVAF in the general adult population of Europe is more than double that reported just one decade earlier, ranging from 1.9% in Italy, Iceland, and England, to 2.3% in Germany and 2.9% in Sweden [2]. However, with the aging of the population, the incidence and prevalence of NVAF double for each advancing decade of life [1]. In Spain, the prevalence of NVAF has progressively increased with age and reached 6.3% for those older than 75 years [3]. In the USA, the prevalence of NVAF in Medicare beneficiaries older than 65 years is estimated to be 8.6% and its incidence has continued to be much higher in the elderly population (18.8 per 1,000 person-years in those aged 70-74 years versus 53.9 per 1,000 person-years in those aged 84-89 years) [4]. In Germany, subjects aged 75-84 years show an NVAF incidence of 18.6 per 1,000 person-years [2]. The prevalence of NVAF was found to be between 10% and 17% of those aged 80 years or older. NVAF has a greater prevalence in men, with a male to female ratio of approximately 2:1. However, in the elderly population women represent the bulk of patients with NVAF due to their longer survival [2]. Epidemiological surveys might underestimate the real prevalence and incidence of NVAF since a substantial proportion of elderly patients are asymptomatic, with NVAF often discovered at a routine check or only at the time of stroke, or even after stroke occurrence [5].
In the European Union, there will be approximately 14 million NVAF patients among individuals aged >55 years in the year 2030, and the number of new NVAF cases will be 120,000–215,000 per year. To these figures must be added a further 280,000–340,000 new ischaemic strokes, and 3.5-4 million hospitalisations or emergency care consultations for NVAF [2,6]. It is expected that in the USA more than 12.1 million people will have NVAF in 2030, and half of NVAF patients in the USA will be older than 80 years in the year 2050 [7].
Socioeconomic burden
Ischaemic stroke risk is increased fivefold in patients with NVAF. NVAF accounts for 36% of ischaemic strokes in elderly individuals >80 years. Elderly patients >80 years with ischaemic stroke and NVAF are significantly (p<0.0005) more likely to be chronically disabled, bedridden, to require constant nursing care, frequent and longer hospital stays, and device implantations, and to suffer more from the adverse effects of NVAF therapy [8]. The socioeconomic burden of NVAF in the European Union countries is considerable. A study analysing the costs of NVAF in five European countries showed that NVAF was associated with average healthcare costs from 1,010 euros per patient per year in Poland up to 3,225 euros per patient per year in Italy [9]. Moreover, the total annual costs for treating AF range from 272 million euros in Greece up to 3,286 million euros in Italy, while in Spain the total annual cost for treating AF was 1,545 million euros [9]. These figures and costs are expected to double with the increasing number of geriatric patients with NVAF. Really, the magnitude of these data seems to confer an endemic dimension to this healthcare problem, implying not only a greater engagement of physicians but also a significant effort of healthcare systems to improve NVAF prevention and its treatment, and to facilitate the organisation of social interventions for the cure of its consequences.
Causes and risk factors for atrial fibrillation in the old/very old population
In the elderly, the causes and risk factors for atrial fibrillation are the sum effects of the associated comorbidities and age-related changes of biological and physiological conditions.
Associated comorbid conditions predispose to NVAF
Previous studies highlighted the effects of the common clinical risk factors for NVAF. They can contribute to NVAF individually, or as combined effects. In the Framingham cohort and after age adjustment, hypertension, diabetes mellitus, coronary artery disease and smoking were the main risk factors for NVAF. The combined effects of these factors could explain 44% of the burden of NVAF in men and 58% in women [1].
Hypertension, which is the most prevalent cardiovascular disease, is considered an important modifiable risk factor for NVAF [2]. Poorly controlled hypertension has a greater risk for inducing NVAF. Furthermore, in patients with NVAF, hypertension worsens the stroke rate by an additional two to threefold [10].
Chronic heart failure is a complex cardiovascular disease of increasing incidence among the elderly. NVAF and heart failure have been recognised as the two epidemics of modern cardiovascular medicine. Both conditions frequently coexist because heart failure is a strong risk factor for NVAF. The risk of NVAF increases 4.5 to 5.9-fold in the presence of heart failure. NVAF prevalence increases as heart failure severity worsens. NVAF has been estimated to occur in 5% to 10% of patients with mild heart failure, 10% to 26% of those with moderate disease, and in up to 50% with advanced heart failure. It is well known to increase atrial pressure, which gradually leads to atrial dilatation and fibrosis and precipitates the electrical and structural changes that provide the basis for NVAF development [11].
Ischaemic heart disease and renal impairment have an incremental prevalence among the elderly, are known as risk factors for NVAF, and are common underlying causes of death among patients with NVAF. In NVAF patients, the presence of coronary artery disease has been shown to be related to recurrent NVAF episodes, to the presence of symptoms (including arrhythmia, heart failure, and angina symptoms), and to increased risk of death. Therefore, coronary artery disease plays an important role in the mortality and quality of life of patients with AF.
Diabetes mellitus and thyroid disease are relatively common diseases of the elderly and both are well recognised endocrine diseases which show a significant association with NVAF. Significant valvular disease in the elderly can induce atrial fibrillation by its direct mechanical effects that result in an increment of atrial pressure and volume. High body mass index, sleep apnoea and chronic lung disease have been associated with the development and progression of NVAF.
Age-related changes predispose to NVAF
The aging heart, characterised by myocardial fibrosis and atrial dilation, provides an ideal soil and substrate for NVAF to flourish. NVAF induces electrical and structural remodelling in the atria by shortening, mismatching, and lengthening the effective refractory period (increase of dispersion), depressing the intra-atrial conduction, and deprivation of its contractile function. Thus, NVAF begets NVAF [12].
Aging is accompanied by a continuous process of degeneration in living organisms, leading to attenuation of most biochemical and physiological functions and indicators.
The majority of elderly individuals live with one or more comorbidities. The atrial myocardium undergoes electrical and structural remodelling with age, both of which play an important role in the induction and/or perpetuation of NVAF. The longer time during which the atrial myocardium is exposed to the effects of external stressors and risk factors would explain the association of age with NVAF [13].
Aging by itself exerts structural changes of the atrial bundles which are characterised by accumulation of the fibrous tissue that is interspersed between myocytes. The excessive accumulation of fibrillary collagen in the extracellular space may result from the gradual age-dependent cardiomyocyte loss (replacement fibrosis), or it may be accelerated by chronic diseases such as hypertension, myocarditis, and congestive heart failure (reactive fibrosis). Fibrosis of the atria in the aging heart is the hallmark substrate for NVAF [13].
Arterial stiffness and left ventricular diastolic dysfunction have been considered as part of the normal aging process. However, arterial stiffness predicts coronary artery disease, stroke and mortality. When using pulse pressure as a marker of arterial stiffness, participants of the Framingham Heart Study were found to have an increased risk for NVAF as well. Left ventricular diastolic dysfunction induces left ventricular relaxation abnormalities that could lead to higher atrial pressures during atrial diastole by reducing passive left atrial emptying. In the long term this might lead to left atrial and pulmonary vein dilatation and potentiate electrical and structural remodelling, aggravating the risk of having NVAF [14].
Complications related to NVAF in old/very old patients
Ischaemic stroke
NVAF is recognised as a major risk factor for stroke, increasing the risk by fivefold in comparison to that in individuals in normal sinus rhythm [15]. In the general population, the incidence of stroke is age-dependent and doubles for every successive decade after the age of 55 years, reaching around 12% in those 75-84 years old and 17% in those >85 years of age [16]. In ischaemic stroke survivors >65 years, 50% had residual hemiparesis and 30% could not walk without assistance. NVAF-related stroke is associated with double the mortality and more severe functional disability than stroke affecting patients in sinus rhythm. The attributable risk of stroke from NVAF increases significantly with age, from 1.5% for individuals aged 50 to 59 years to 23.5% for individuals aged 80 to 89 years, with about 40% of stroke in patients >80 years due to NVAF [17]. The average stroke rate was reported at 4.1% per year among the primary prevention studies for NVAF and 13% per year among those with prior stroke or transient ischaemic attack (TIA) [17]. In the very old (>90 years), patients with NVAF had a significant increase in the ischaemic stroke rate of 5.75% per year compared to 3.00% per year in those without NVAF (hazard ratio 1.93; 95% confidence interval: 1.74–2.14) [18]. The incidence of thromboembolic (TE) events in patients aged ≥85 years with NVAF was 6.3% per year without oral anticoagulants (OACs) versus 4.3% per year with OACs (2% absolute reduction) [19]. Important risk factors for stroke in the setting of NVAF include advancing age, hypertension, heart failure, diabetes mellitus, previous stroke or TIA, vascular disease and female sex. Additional circulating biomarkers of fibrosis or cardiac stress, including high levels of troponin and brain natriuretic peptide, increase the risk of stroke in the setting of NVAF independent of those well-established clinical characteristics [15]. Age is a detrimental factor for the prevalence of both stroke and NVAF, putting older people with NVAF at the highest risk of stroke compared with younger patients in sinus rhythm. Furthermore, very old age is an independent predictor of worse short- and long-term outcomes in stroke patients. OACs are recommended to reduce ischaemic stroke in these high-risk patients [15].
Major bleeding
Advanced age is a strong independent predictor of a major bleeding event [15]. Major bleeding is of increasing prevalence in the general very elderly population. Intracerebral haemorrhage (ICH) is the most dreadful vascular event with a high fatality rate or severe disability sequelae. A recent retrospective large cohort inpatient database study in the Netherlands demonstrated that the incidence of ICH per 100,000 was 5.9 in those 35-54 years old, 37.2 in those 55-74 years old, and 176.3 in the very elderly population 75-94 years old [20]. In a German study which analysed the database of a regional prospective stroke registry between 2007 and 2009, 34% of 3,448 patients with ICH were aged 80 years or more [21]. Gastrointestinal bleeding is an important and common type of bleeding in the general very elderly population. More than 1% of the general elderly population aged 80 years and older are hospitalised each year because of gastrointestinal bleeding. In the USA, the incidence of lower gastrointestinal bleeding in the general population ranges from 20.5 to 27 per 100,000 persons per year, with a greater than 200-fold increase from the third to the ninth decade of life [22]. The outcome of non-fatal bleeds was also worse at older ages. The proportion of survivors for whom a bleed resulted in a new or sustained increase in disability rose from 3% for people aged younger than 75 to 25% for people aged over 75. Overall, the risk of disabling or fatal bleeding over 10 years was 10 times higher at ages 75 years or older, compared with younger patients. A large real-world study, which included a large cohort of 125,195 elderly patients ≥66 years old with NVAF on OACs, showed that the overall risk of bleeding was 3.8% per year. The risk of major bleeding in patients >75 years was 4.6% versus 2.9% in those <75 years of age [23]. The risk of major bleeding among very elderly patients >90 years with NVAF on OACs reaches about 6.75% per year, and the risk of ICH in those very elderly patients ranges from 0.42-1.63% per year depending on the type of OACs drug used [18]. In very elderly patients with NVAF, the risk of major bleeding was similar in patients on OACs and in those on antiplatelet therapy or without antithrombotic treatment (4.0% per year versus 4.2% per year; p=0.77) [19]. Several factors were found to be independent predictors of major bleeding in very elderly patients with NVAF in several studies. Individual and easily detectable risk factors such as anaemia, thrombocytopaenia, renal impairment, uncontrolled hypertension, history of prior bleeding event, concomitant non-steroidal anti-inflammatory drugs and antiplatelet drugs have recently shown a strong association with major bleeding events in very elderly patients with NVAF [15].
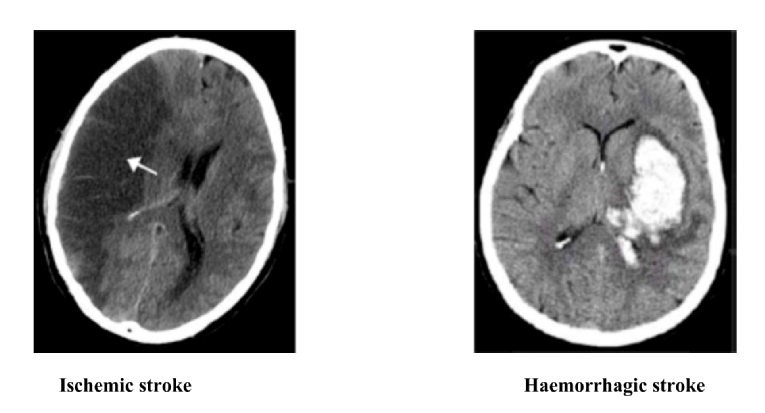
The incidence of ICH and fatal bleeding with OACs increased with age. A delicate balance should be maintained when using OACs to prevent a TE event without inducing major or fatal bleeding (Figure 1).
Figure 1. Very old patients with atrial fibrillation have increased risk of ischaemic stroke and of haemorrhagic stroke; therefore, a good knowledge and careful handling of oral anticoagulants are needed.
Heart failure
Heart failure and NVAF were predicted to become epidemics of the 21st century. In elderly patients, the pathophysiology and risk factors for heart failure and NVAF are closely aligned with a significant burden of comorbidity. NVAF is considered both a cause and consequence of heart failure, with complex interactions leading to impairment of systolic and diastolic function. NVAF is associated with a threefold increased risk of incident heart failure. NVAF can promote the development of heart failure by a number of established mechanisms such as: 1) loss of atrial systole in NVAF which impairs left ventricular filling and can decrease cardiac output by up to 25%, particularly in patients with diastolic dysfunction, and 2) irregular and/or rapid ventricular conduction in NVAF which can lead to left ventricular dysfunction and, in some patients, to tachycardia-induced cardiomyopathy. Restoring sinus rhythm could increase stroke volume and left ventricular emptying even before contractility improves [24].
Cognitive decline and dementia
The increasing prevalence of cognitive decline and dementia in the elderly represents a healthcare challenge and a socioeconomic burden. There is growing evidence that NVAF is a risk factor for cognitive decline and dementia. Several community-based studies show a consistently higher rate of cognitive decline and increased risk of dementia in persons with NVAF. These associations are partly due to the increased risk of clinical stroke in NVAF. Several mechanisms other than clinical stroke might be strongly implicated in the process of NVAF inducing dementia such as the incidence of silent cerebral infarcts, microbleeds and recurrent episodes of cerebral hypoperfusion. Proper oral anticoagulation and improved management of the overall cardiovascular risk and comorbidities in individuals with NVAF offer a reliable hope for reducing the impact of NVAF on cognitive decline and dementia [25].