Abbreviations
C-O cardio-oncology
CAD coronary artery disease
CCT cardiac computed tomography
CMR cardiac magnetic resonance
CTRCD cancer therapy-related cardiac dysfunction
EF ejection fraction
ERNA equilibrium-gated radionuclide angiography
GLS global longitudinal strain
IMT intima-media thickness
LGE late gadolinium enhancement
LV left ventricular
PET positron emission tomography
SPECT single photon emission cardiac tomography
Keywords: cardiac oncology, carotid artery stenosis, ejection fraction, global longitudinal strain, heart failure, pericarditis
Introduction
The cardiac imaging applied to cardio-oncology (C-O) includes standard and advanced (speckle tracking and three-dimensional [3D]) echocardiography, cardiac magnetic resonance (CMR), cardiac computed tomography (CCT) and cardiac radionuclide imaging (equilibrium-gated radionuclide angiography [ERNA], single photon emission cardiac tomography [SPECT] and positron emission tomography [PET]). The choice of one technique over another depends on two main points: the need to achieve an early diagnosis in order to begin a timely management, and the type of cardiotoxicity which is suspected. Accordingly, in cancer patients the imaging tools are used to detect the following:
- Subclinical and clinical heart failure
- Coronary artery disease
- Valvular heart disease
- Vascular complications
- Pericardial damage
Diagnosis of subclinical and clinical heart failure
The need for a timely diagnosis of subclinical and clinical heart failure by using cardiac imaging has been addressed by the Expert Consensus of the ASE and EACVI [1] and more recently reinforced by the ESC Position Paper on cancer treatments and cardiovascular toxicity [2]. The quickest and most available imaging tool in detecting cancer therapy-related cardiac dysfunction (CTRCD) is transthoracic echocardiography [1]. Standard Doppler echocardiography examination enables the identification of abnormalities of left ventricular (LV) systolic and diastolic function as well as of the involvement of the right ventricular (RV) chamber. The changes of LV diastolic parameters do not appear to be decisive in the oncologic setting, mainly because of their load dependence which is often evident in patients undergoing anthracycline therapy and suffering from vomiting, diarrhoea and anaemia [1]. The most used parameter in C-O is LV ejection fraction (EF), obtainable by 2D echocardiographic measurements of LV internal chamber volumes at end-diastole and end-systole. Traditionally, an echo determination of LVEF is requested by the oncologists in all cancer patients at baseline, i.e., before starting anti-cancer therapy, and in any situation in which the suspicion of heart failure is plausible, during and after completion of the anti-cancer therapy. 2D-derived LVEF is also used to start cardioprotection and to establish the interruption from anti-cancer therapies. A drop in LVEF from baseline greater than 10 percentage points, to a value <53%, necessitates the interruption of the ongoing anti-cancer therapy.
The use of 2D-derived LVEF over time, i.e., at baseline and every three months during chemotherapy, has been proposed by Cardinale et al [3] for an early detection of anthracycline cardiotoxicity and subsequent improvement of LV systolic function by heart failure therapy. However, the diagnostic accuracy of 2D LVEF is limited by its suboptimal biological (day-to-day) reproducibility (about 10%), which has been clearly demonstrated in the general population and in the oncologic setting as well [4]. 2D echocardiography appears to be reliable in the detection of only those LVEF differences over time which are greater than 10%. Accordingly, in oncologic patients, the best determination of LVEF should be performed using 3D echocardiography [1,2] which does not suffer from geometric assumption and is relatively operator-independent. Indeed, the feasibility of the 3D technique can be reduced in some cancer patients because of the negative influence of factors such as concomitant radiotherapy (breast cancer, lymphoma) and surgery (mastectomies of left breast cancer, breast expanders or implants), which make the ultrasound windows under these circumstances suboptimal [5].
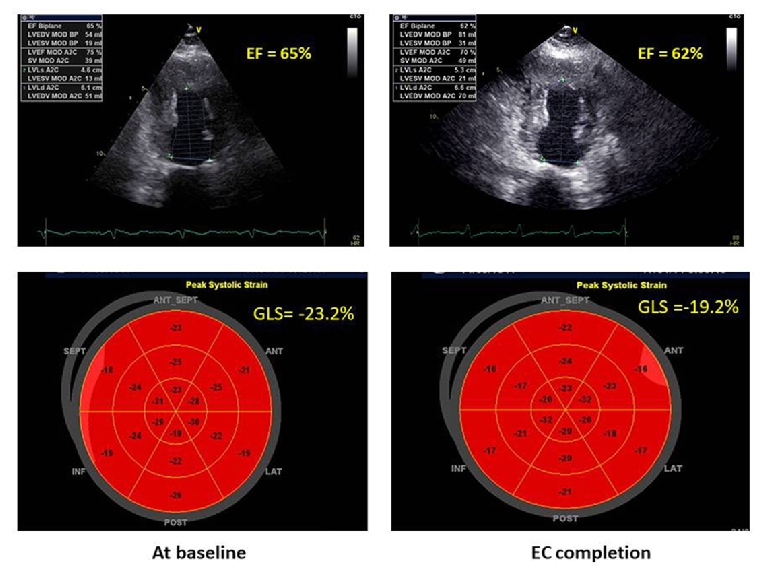
Alternatively, the use of global longitudinal strain (GLS) by speckle tracking echocardiography is strongly recommended [1,2] because of its optimal feasibility and biological reproducibility (about 6%) [6]. Baseline measurements of GLS identify those at high risk of CTRCD in patients undergoing anthracyclines [7]. Moreover, while a relative percentage reduction of GLS <8% from baseline cannot be considered clinically relevant, a GLS reduction from baseline >15% is very likely to be abnormal and could be utilised as the starting point for timely cardioprotection (i.e., beta-blockers and ACE inhibitors) [1]. This strategy could be used in clinical practice to avoid the progression from subclinical CTRCD to overt heart failure in cancer patients, despite the lack of a definite demonstration of its beneficial impact on decision making in this clinical setting. In addition, because of the recognised vendor dependence of speckle tracking echocardiography, the same vendor-specific ultrasound machine should be used for serial determination of GLS in cancer patients [1]. Figure 1 shows a significant drop of GLS (>15% in comparison with baseline values) in a woman with breast cancer during trastuzumab therapy, in the absence of heart failure symptoms. This drop suggests a timely start of cardioprotection in order to avoid the progression of subclinical CTRCD towards overt heart failure.
Figure 1. Changes of LVEF and GLS during chemotherapy in a woman with breast cancer. After the end of EC (epirubicine + cyclophosphamide) cycles, LVEF is still normal but GLS shows a 16% drop from baseline. At this point the patient is asymptomatic but could start cardioprotective therapy according to ASE/EACVI Expert Consensus [1].
Among the other cardiac imaging techniques, CMR is not currently used as a first-line tool for risk stratification of heart failure in cancer patients because of its high cost and limited availability. However, it should be considered, at baseline as well as during/after chemotherapy, in patients with non-diagnostic echocardiograms or in those showing unexplained LV and/or RV enlargement, or morphological abnormalities consistent with infiltrative cardiomyopathies. Late gadolinium enhancement (LGE) CMR can be used in specific cases, allowing the recognition of late enhancement differentiation of non-ischaemic and ischaemic myocardial fibrosis. T1- and T2-weighted CMR sequences can also help to detect intracellular and interstitial oedema, alterations which can precede reduction in both LVEF and GLS. Noteworthy, a subepicardial LGE of the lateral wall has been described in patients treated with trastuzumab [8]. However, this finding has not been confirmed in other studies. LGE CMR can also have value for identifying myocarditis in patients receiving cancer immunotherapy.
Currently, ERNA is still used to detect CTRCD in the United States, but does not have a wide application in Europe.
Diagnosis of coronary artery disease
The diagnostic capability of rest echocardiography in coronary artery disease (CAD) is limited to the assessment of the presence and magnitude of regional wall motion abnormalities. In asymptomatic patients, moderate-to-severe dyssynergia has been observed in up to 17% of Hodgkin’s lymphoma survivors treated with mediastinal radiotherapy (≥35 Gy) [9]. However, a hypokinetic LV segment is not necessarily related to the presence of CAD but could perhaps reflect a myocardial disease process.
Stress-induced wall motion abnormalities are reliable indicators of inducible ischaemia in patients undergoing anti-cancer therapies which are associated with the development of myocardial ischaemia (e.g., capecitabine, 5-fluorouracil, bevacizumab, sunitinib, sorafenib) and who have an intermediate/high risk of CAD. Accordingly, stress echocardiography represents the first-line cardiac imaging to be utilised. Physical or pharmacological (dobutamine or dipyridamole) testing can be chosen in relation to the exercise capacity of the individual patient at the time of the examination, to achieve a diagnosis of inducible ischaemia [1]. The use of myocardial contrast agents can be promoted in patients with a difficult ultrasonic window, in order to improve the border detection and the identification of LV dyssynergy segments. The main attraction of stress echo in this clinical setting is the lack of radiation exposure, a characteristic shared with stress CMR, which can identify the presence and magnitude of inducible ischaemia with high diagnostic accuracy but is expensive and has poor availability [10].
Nuclear (SPECT and PET) stress testing is a good alternative, in particular when stress echocardiography becomes problematic (e.g., breast cancer patients with implants and mastectomies).
The use of CCT should be limited to identifying coronary atherosclerosis in cancer patients undergoing mediastinal radiotherapy (e.g., because of lymphoma), even several years after the completion of this kind of management [11].
Diagnosis of valvular heart disease
This evaluation is particularly important after radiotherapy which can induce multiple types of valvular damage, including fibrosis and calcification of the aortic root, aortic valve annulus and leaflets, mitral-aortic intervalvular fibrosa, mitral valve annulus and both the base and the mid portion of the mitral valve leaflets [10].
Transthoracic echocardiography is the main tool to identify valvular damage in these patients. The main differential finding between radiotherapy-induced valvular heart disease and rheumatic heart disease is the presence of commissural fusion after radiotherapy, while the involvement of the mitral leaflet tips is an indication of rheumatic disease. 3D echocardiography can be extremely useful to detect commissural fusion, in particular when 2D echocardiography does not allow a complete visualisation of the commissures [10]. Transoesophageal echocardiography needs to be used when transthoracic echocardiography is non-diagnostic or when further diagnostic refinement is required.
In patients with poor quality echocardiographic images or discrepant results, CMR can also be used in order to obtain a comprehensive assessment of valvular heart disease.
Diagnosis of peripheral vascular toxicity
This kind of assessment should be promoted particularly in cancer patients undergoing treatment with tyrosine kinase inhibitors, anti-metabolites, and endothelial growth and hormone factor inhibitors. Clonal haematopoiesis also represents an important risk factor for atherosclerotic vascular damage [12]. However, the most frequent cause of vascular damage in cancer patients is radiotherapy, which can provoke peripheral artery disease (mainly carotid), even several years after its completion.
Carotid artery ultrasonography is particularly recommended to detect the increase of intima-media thickness (IMT) and internal lumen stenosis after radiotherapy. A significant increase of carotid IMT was found in 24 to 42% of patients with Hodgkin’s lymphoma who had undergone radiotherapy five years previously [11].
Contrast-enhanced magnetic resonance angiography and CT angiography can be performed when needed, in particular to evaluate subclavian and aortic diseases related to radiotherapy. This screening can lead to identifying porcelain aorta even 10-20 years after the end of radiotherapy.
Diagnosis of pericardial damage
Pericardial effusion, cardiac tamponade and pericarditis can appear during several types of chemotherapy but are especially due to radiotherapy. Pericardial damage from radiotherapy includes acute exudative pericarditis, delayed acute pericarditis (within weeks after radiotherapy), characterised by asymptomatic pericardial effusion or symptomatic pericarditis, and delayed chronic pericarditis (several years after radiotherapy completion), due to extensive pericardial fibrous thickening and chronic constriction. Constrictive pericarditis appears to be radiotherapy dose-dependent and is observed in 4 to 20% of patients undergoing mediastinal radiotherapy.
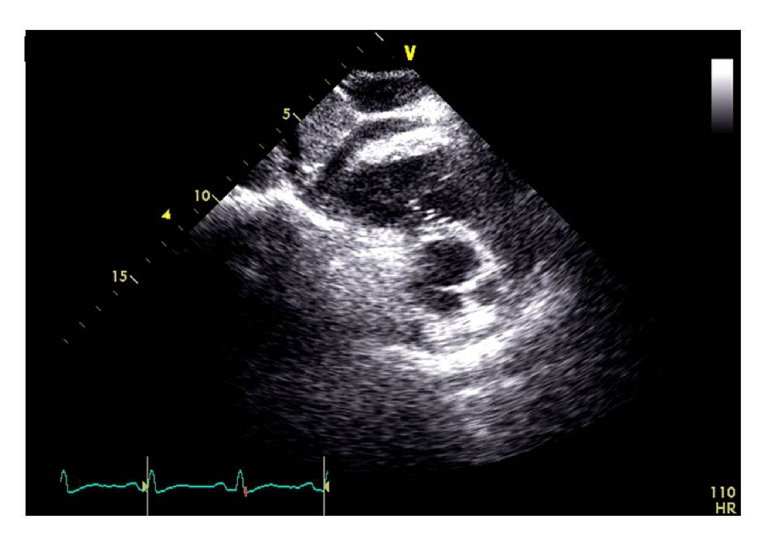
A multi-modality imaging approach to pericardial damage in cancer patients should be performed according to the EACVI position paper on myocardial and pericardial diseases [13]. Echocardiography is the first-line cardiac imaging for the diagnosis of radiotherapy-induced pericarditis. Figure 2 illustrates a significant pericardial thickening (>2 mm) two years after the end of radiotherapy for lymphoma. However, although pericardial thickness is currently considered a main criterion for diagnosing constrictive pericarditis, the range of pericardial thicknesses is highly variable (1–17 mm, mean 4 mm), with up to 20% of patients showing a normal thickness (<2 mm). Cardiac tamponade may be suggested by large fluid accumulation, compression of the cardiac cavities, and right-sided venous congestion. Serial echocardiographic examinations are helpful in patients affected by pericardial effusion or constrictive pericarditis but are less useful for identifying pericardial thickening and calcifications. CMR and cardiac CT, also using LGE (pericardial enhancement suggests residual pericardial inflammation), are more sensitive in detecting specific anatomical abnormalities of the pericardium. End-stage chronic constrictive pericarditis cannot show pericardial LGE on CMR, whereas pericardial enhancement is demonstrative of residual inflammation.
Figure 2. 2D echocardiographic subcostal view showing a significant RV pericardial thickening (>2 mm at the RV free wall) in a patient with lymphoma who had undergone mediastinal radiotherapy two years before the echo examination.
Real-time cine loop CMR can help to assess the influence of respiration on the interventricular septal shape and motion, easily detecting the increased ventricular coupling which occurs under these circumstances [14]. Pericardial calcifications and large pericardial effusion can also be easily identified by non-enhanced CCT images.
Multi-modality imaging approach to cancer patients
The modern multi-modality imaging approach to anti-cancer therapy complications should be focused on the early diagnosis and treatment of cardiovascular complications in individual patients [15]. In this context, echocardiography is the first-line imaging, whereas the application of other imaging tools, mainly CMR, should be modulated whenever needed. In any case, the cardiologic management should always take into account the need for continuing anti-cancer therapy without interruption, aiming at cancer cure and remission, or alternatively support the oncologist's choice between different anti-cancer therapies, in order to maintain survival free from cardiovascular morbidity and mortality.