Keywords
pulmonary hypertension, right ventricle, transcatheter therapy, tricuspid regurgitation, tricuspid valve
Introduction
Tricuspid regurgitation (TR) is an important yet often underappreciated disease in cardiology practice. In up to 90% of patients in Western countries moderate-to-severe TR is secondary or functional, most often due to left-sided heart disease and other causes of pulmonary hypertension [1]. In these cases, functional TR results from tricuspid annular dilatation in the setting of right ventricular remodelling, mainly due to pressure or volume overload (Figure 1). Up to 50% of patients undergoing mitral valve surgery for functional ischaemic mitral regurgitation have moderate or severe TR, and a substantial proportion of patients develop TR late after previous left-heart surgery [2]. Given the overall increase in life expectancy, improved treatment options for patients with ischaemic or non-ischaemic cardiomyopathy and expanding transcatheter interventions for mitral valve pathologies in patients at increased surgical risk, the number of patients presenting with relevant TR may increase in the coming decades. Importantly, recent publications have challenged the long-held misperception that functional moderate-to-severe TR will resolve once successful treatment of mitral regurgitation has been carried out. About 70% of patients with a more than moderate TR before mitral valve edge-to-edge repair still had moderate-to-severe TR after two years of follow-up [3]. The prevalence of coexisting moderate-to-severe TR is between 11% and 26% in patients undergoing transcatheter aortic valve replacement (TAVR) for native aortic valve stenosis, and a reduction in echocardiographic TR grade has been observed in 15% to 50% following TAVR in these patients [4]. Conflicting results have been reported regarding the prognostic impact of TR following TAVR and surgical aortic valve replacement. Interestingly, a recent analysis in patients at high surgical risk undergoing valve-in-valve TAVR for degenerated surgical aortic bioprostheses suggested a significant reduction in TR severity and lack of prognostic impact of preprocedural TR at short- and long-term follow-up [4].
Figure 1. Causes of organic and functional TR.
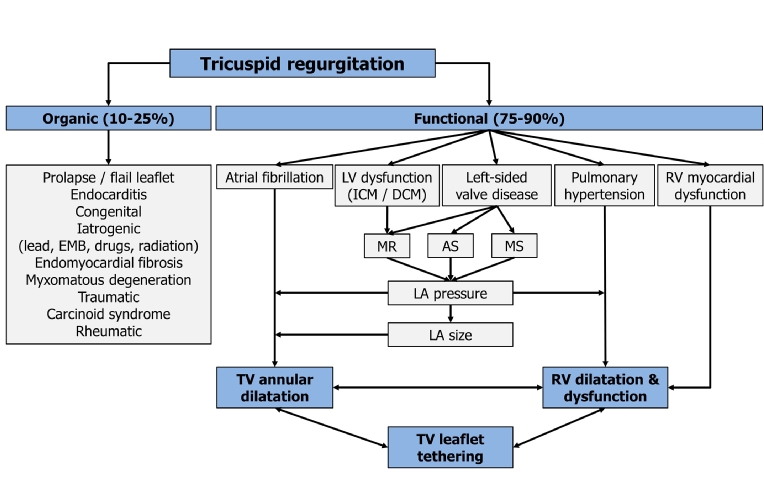
AS: aortic stenosis; DCM: dilated cardiomyopathy; EMB: endomyocardial biopsy; ICM: ischaemic cardiomyopathy; LA: left atrial; LV: left ventricular; MR: mitral regurgitation, MS: mitral stenosis; RV: right ventricular; TV: tricuspid valve
Medical treatment options for functional TR
Frequently, patients with moderate-to-severe TR present late in the natural history of the disease with exercise limitation and other symptoms of right heart failure difficult to treat by medical therapy. In the diagnostic and treatment process, the underlying cause of functional TR needs to be identified and treated, if possible. Patients with ischaemic or non-ischaemic cardiomyopathy are managed according to current heart failure guidelines [5]. If there is clinical suspicion of pulmonary hypertension, further differential diagnosis, including right heart catheterisation, is mandatory to classify pulmonary hypertension and identify patients who might benefit from specific drug therapy (e.g., calcium channel blockers, endothelin receptor antagonists, phosphodiesterase type five inhibitors and guanylate cyclase stimulators, prostacyclin analogues and prostacyclin receptor agonists). Oral anticoagulation should be initiated in patients with right heart chamber dilatation due to (repeated) pulmonary embolism. Nevertheless, in a considerable number of patients medical treatment options of TR are limited for the following reasons: often, an impairment of kidney function in patients with advanced TR limits further escalations of diuretic dose to improve symptoms of volume overload. If kidney function is preserved, adding an aldosterone antagonist represents a possible treatment option, in particular for patients with hepatic congestion and secondary hyperaldosteronism. However, the benefit either of aldosterone antagonists or of angiotensin-converting enzyme inhibitors/angiotensin II receptor blockers on right ventricular remodelling or functional improvement in patients with severe functional TR has not been validated in clinical studies.
Current indications for surgical treatment of functional TR
Current guidelines for the management of valvular heart disease provide recommendations to guide surgical interventions in primary and secondary TR [6]. Endorsements on transcatheter techniques targeting TR are not yet available, owing to the fact that interventional TR treatment is still in early clinical testing. The optimal timing of surgical intervention for TR remains controversial. However, delayed surgery must be avoided given the risk of irreversible right ventricular damage, organ failure and poor results of later surgical intervention. The 2017 ESC/EACTS Guidelines for the management of valvular heart disease provide a class I (level of evidence: C) recommendation for tricuspid valve surgery in patients with severe functional TR undergoing left-sided valve surgery [6]. Concomitant treatment of TR during left-sided heart surgery does not confer an increased risk of postoperative morbidity, mortality or permanent pacemaker requirement. According to a recent meta-analysis, concomitant tricuspid valve repair during left-sided valve surgery was associated with a reduction in cardiac-related mortality and improved echocardiographic outcomes for TR after a mean weighted follow-up of six years as compared to cases without concomitant tricuspid valve repair [7]. A class IIa (level of evidence: C) recommendation is given for tricuspid valve surgery in patients with mild or moderate functional TR with a dilated tricuspid annulus (i.e., ≥40 mm or >21 mm/m2 by 2D echocardiography) undergoing left-sided valve surgery [6]. The latter recommendation justifying tricuspid valve surgery in patients with moderate or even mild TR appreciates the increased risk of progressive TR and right heart failure once tricuspid annular dilatation has occurred. Tricuspid valve surgery may be considered in patients undergoing left-sided valve surgery with mild or moderate secondary tricuspid regurgitation even in the absence of annular dilatation when previous recent right heart failure has been documented (class of recommendation: IIb, level of evidence: C) [6]. In addition, the guidelines provide a class IIa (level of evidence: C) recommendation for tricuspid valve surgery in patients with severe TR after previous left-sided surgery if patients are symptomatic, or progressive right ventricular dilatation or dysfunction is evident. However, before a decision is made for reoperation on the tricuspid valve in cases of persistent TR after left-sided heart surgery, recurrent left-sided valve dysfunction, severe right or left ventricular dysfunction, and severe pulmonary vascular disease or hypertension have to be ruled out.
Surgical repair techniques for functional TR
Surgical tricuspid valve repair with annuloplasty is the preferred treatment for functional TR in patients with suitable anatomy, preserved right ventricular function and acceptable surgical risk [6,8]. Tricuspid valve annuloplasty techniques aim to target the pathophysiological hallmark of functional TR, i.e., dilatation of the tricuspid annulus mainly along its posterior portion with an increased size of the tricuspid valve orifice and alterations of annular geometry. Since the 1960s, several surgical annuloplasty methods have been described, including suture, band and ring techniques. Amongst them, suture bicuspidisation, better known as the Kay procedure, intends to reduce TR by obliterating the annular segment corresponding to the posterior leaflet through placement of pledget-supported mattress sutures in the annulus [9]. As a result, the tricuspid annular circumference is reduced, and the tricuspid valve is converted into a smaller but competent mitral-like valve. During De Vega annuloplasty, two C-shaped suture lines are placed along the base of the anterior and posterior tricuspid valve leaflet, starting at the anteroseptal commissure and ending beyond the origin of the coronary sinus. At the anteroseptal and posteroseptal commissures the sutures lines are anchored with pledgets and, finally, tied to reduce the tricuspid valve orifice diameter [9].
Currently, tricuspid valve annuloplasty is usually performed by implantation of an undersized ring (rigid, semi-rigid, flexible) or a flexible band (Figure 2). Compared with the use of flexible annuloplasty bands, implantation of a rigid or semi-rigid ring is associated with a reduced incidence of late, recurrent tricuspid regurgitation. However, use of a rigid ring may increase the risk of subsequent ring dehiscence [10]. Annuloplasty rings or bands do not cover the whole tricuspid annulus, but remain open at the septal annulus to avoid injury to the conduction system. In recent years, refined tricuspid annuloplasty rings resembling the complex three-dimensional configuration of the native tricuspid annulus have been developed [9]. Retrospective analyses have suggested that ring annuloplasty is superior to suture annuloplasty in terms of residual and recurrent TR, redo surgery and, possibly, survival [9]. The choice of annuloplasty ring (rigid vs. flexible vs. three-dimensional) for tricuspid valve repair is still a matter of debate.
Figure 2. Intraoperative surgical view of the tricuspid valve.
Left: annuloplasty ring sutures are placed along the annulus of the tricuspid valve starting from the septal leaflet (S), across the posterior part of the annulus (P) followed by the anterior part (A).
Right: implantation of an annuloplasty ring into the tricuspid valve. The septal annulus remains uncovered to avoid passing the anchoring sutures close to the conduction system.
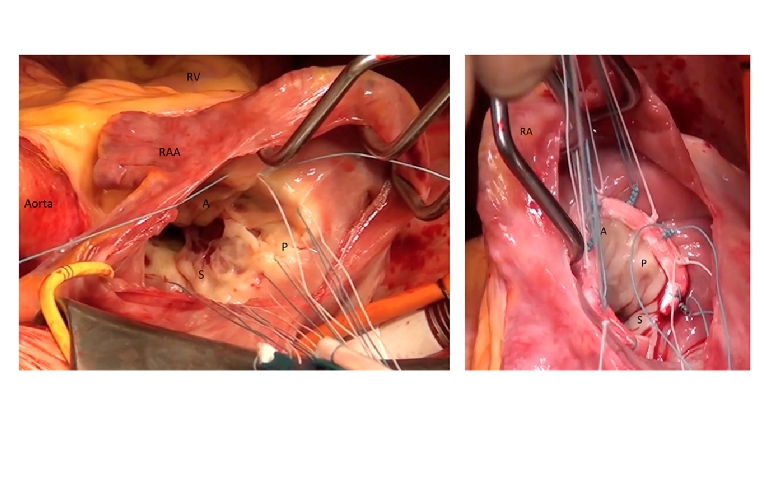
RA: right atrium; RAA: right atrial appendage; RV: right ventricle
Tricuspid valve repair is associated with lower perioperative mortality as compared to valve replacement in patients with functional TR and, therefore, is generally the preferred surgical treatment option [8]. Tricuspid valve replacement should be considered when valve repair is technically not feasible or predictably not durable (e.g., in patients with profound right ventricular remodelling or dysfunction, or higher pulmonary artery pressures). Studies comparing bioprosthetic and mechanical valves for tricuspid valve replacement indicated similar long-term outcomes. Given the lower risk of thromboembolism, many surgeons favour bioprosthetic valves; however, the choice of prosthesis type should be individualised, taking into consideration the patient’s age, condition, and concomitant cardiac or non-cardiac diseases [9].
Limitations for surgical TR repair in clinical practice
Concomitant tricuspid valve repair for functional TR at the time of left-sided valve surgery is not associated with increased mortality if right ventricular function and dimensions are preserved and the patient does not suffer severe heart failure. However, repeat surgery for symptomatic TR late after left-sided valve surgery is associated with substantially increased perioperative morbidity and mortality, with early mortality rates up to 20% even in experienced centres. The increased risk of reoperation on the tricuspid valve has been attributed mainly to the late referral and consequently poor clinical condition of the patient. In these cases, the risk of repeat surgery has to be weighed against the modest functional outcome observed in many patients and the comprehensive preoperative evaluation of left and right ventricular function, pulmonary hypertension, kidney and liver function. Minimally invasive tricuspid valve operations through a right thoracotomy may provide an alternative surgical treatment option associated with excellent early outcome even in high-risk patients undergoing elective reoperative tricuspid valve repair for TR after left-sided heart surgery [11].
However, based on our own observations in clinical practice, there is yet another growing group of elderly patients presenting with massive TR and intractable symptoms of right heart failure without prior left-sided heart surgery. The clinical features of these patients are rather heterogeneous, but almost all of them display functional TR, about one third have an ejection fraction <40%, severe mitral regurgitation is seen in about 60% and there is a high prevalence of atrial fibrillation [12,13]. Often, right heart chambers and tricuspid annulus are severely dilated, and right ventricular function is frequently impaired in this group of patients. In addition, a significant proportion of these patients displays pacemaker or implantable cardioverter-defibrillator leads across the tricuspid valve, chronic pulmonary disease, pulmonary hypertension and an impairment in kidney function. These patients are highly symptomatic with more than 95% presenting in NYHA functional Class III or IV despite optimal medical therapy and face recurrent hospitalisations for decompensated right heart failure. Up until recently, conventional and/or minimally invasive tricuspid valve surgery was offered to these patients next to medical treatment, however at a significant operative risk. Nowadays, emerging transcatheter therapies may provide an alternative treatment option for these patients if surgical risk exceeds the potential benefit.
Emerging transcatheter treatment options for functional TR
In recent years, several devices have entered preclinical and early clinical testing for transcatheter treatment of functional TR. Overall, the number of patients treated in compassionate use or clinical feasibility programmes is still limited and data on long-term haemodynamic consequences are missing. Nevertheless, early signs of safety, feasibility and clinical benefit for some devices have aroused great interest among cardiac specialists. Current studies aim to address the question whether interventional TR treatment provides an alternative therapeutic option for patients with severe functional TR, intractable symptoms of right heart failure and prohibitive surgical risk based on the conclusive decision of a multidisciplinary Heart Team (Table 1). Current existing transcatheter therapies for functional TR can be divided into three groups according to their mode of action and anatomic therapeutic target (Figure 3). The present article provides a short summary of devices which have been applied in patients with severe functional TR and for whom published data (at least case records) are available.
Table 1. Comparison of procedural aspects between surgical and transcatheter tricuspid valve repair.
| Procedural aspects | Surgical TVR | Transcatheter TVR |
|---|---|---|
| Acute and midterm procedural success | + | - |
| Data on long-term durability | + | - |
| Operative risk in elderly patients | substantial | low |
| Feasible in advanced RV failure | + | - |
| Widespread use/availability | + | - |
| Invasiveness of the procedure | - | + |
| Feasible despite impaired echo imaging | + | - |
“+” indicates an advantage and “-” indicates a disadvantage of surgical or transcatheter TVR.
RV: right ventricular; TVR: tricuspid valve repair
Figure 3. Transcatheter techniques targeting functional TR.
Transcatheter devices in early clinical testing can be grouped according to their mechanism of action into annuloplasty devices, coaptation devices and heterotopic caval valve implantation.
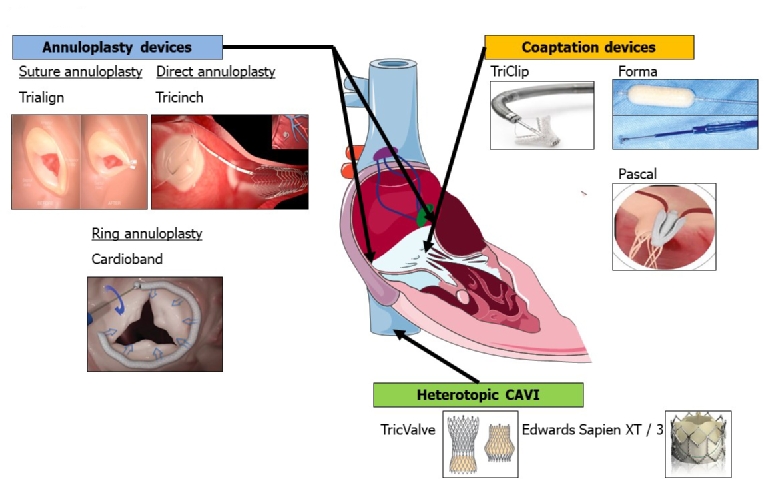
Percutaneous tricuspid valve annuloplasty devices
The first group comprises percutaneous tricuspid valve annuloplasty devices, based either on transcatheter suture or ring implantation techniques.
TrialignTM
The TrialignTM device (Mitralign Inc., Tewksbury, MA, USA) is a transjugular suture-based tricuspid valve annuloplasty system which aims to reduce tricuspid annular diameter through tissue plication. During the Trialign procedure a pair of polyester pledgets is delivered across the tricuspid annulus from the ventricular to the atrial side in proximity to the anteroposterior and septoposterior commissure, cinched by a polyester suture to obliterate the posterior tricuspid leaflet and, finally, locked on the atrial side. Hence, from a mechanistic point of view, this approach replicates the surgical Kay procedure. The results of the SCOUT (Percutaneous Tricuspid Valve Annuloplasty System [PTVAS] for Symptomatic Chronic Functional Tricuspid Regurgitation) trial suggest that TR reduction by the Trialign device translates into improvements in NYHA functional class, Minnesota Living with Heart Failure Questionnaire score and six-minute walking distance in the intention-to-treat cohort [14]. In one patient, an intraprocedural ST-elevation myocardial infarction occurred due to tenting of the distal right coronary artery in the region of the plication with significant narrowing. Pledget detachment from the annulus was evident in 3/15 patients on follow-up after 30 days. No major adverse events were reported.
TriCinchTM
The TriCinchTM system (4Tech Cardio, Galway, Ireland) aims to reduce the septolateral tricuspid annular diameter by implanting a nitinol coil anchor in the anteroposterior tricuspid annulus and applying tension on the annulus via a Dacron band fixed to a self-expanding nitinol stent in the inferior vena cava. Unpublished data from the PREVENT (Percutaneous Treatment of Tricuspid Valve Regurgitation With the TriCinch System) trial suggested an intraprocedural technical success rate of 81% in 24 patients with acute TR reduction by at least one grade in 94% of patients. Notably, late annular anchor detachment occurred in 23% of patients and an alternative coil anchoring system to improve annular fixation has been developed [15].
CardiobandTM Tricuspid Valve Reconstruction System
The Cardioband implant (Edwards Lifesciences, Irvine, CA, USA) is composed of an adjustable fabric band with up to 17 anchors for annular fixation and was initially developed for patients with functional mitral regurgitation. The Cardioband tricuspid reconstruction system aims to reduce TR via annular reduction and is advanced into the right atrium via transfemoral access. The anchors are deployed on the beating heart along the anterior and posterior portion of the tricuspid annulus. At the end of the procedure, the Cardioband is contracted to reduce the tricuspid annular circumference. The TRI-REPAIR trial examined safety, efficacy and early functional benefits of TR treatment with the Cardioband tricuspid system [15].
Percutaneous tricuspid valve coaptation devices
The second group currently includes three devices designed to improve leaflet coaptation in functional TR (Figure 3).
TriClip
According to recent registry data, the off-label transfer of the MitraClip® technique (Abbott Vascular, Santa Clara, CA, USA) to the tricuspid valve is so far the most common technique applied for interventional TR treatment [13]. In the largest analysis published thus far, tricuspid valve edge-to-edge repair, carried out either in isolation or in combination with mitral valve edge-to-edge repair, led to an improvement in NYHA functional class in 76% of patients and a 29% increase in six-minute walk test distance [12]. In addition, successful TriClip implantation was associated with reduced mortality and less readmission for heart failure. A small coaptation gap size of TR and a central or anteroseptal location of the TR jet on echocardiography independently predicted procedural success of TriClip implantation [12]. Notably, in patients with both significant mitral and tricuspid regurgitation at high surgical risk, combined transcatheter mitral plus tricuspid valve edge-to-edge repair using the MitraClip technique was associated with superior functional improvement (in terms of NYHA class, NT-proBNP levels and six-minute walk test distance) and fewer hospitalisations for heart failure as compared to isolated mitral valve edge-to-edge repair [16]. Whether application of the novel MitraClip XTR system with longer clip arms and longer grippers with two additional rows of frictional elements will improve the technical success rate in patients with functional TR is the subject of current research.
FORMA
The FORMA repair system (Edwards Lifesciences, Irvine, CA, USA) aims to reduce malcoaptation of the tricuspid leaflets by placing a balloon spacer through the central coaptation line, which reduces the regurgitant orifice area. The device is advanced via the left subclavian or axillary vein. The spacer is stabilised by a rail anchored at the right ventricular apex. Three different balloon sizes (12, 15 and 18 mm) have been implanted. Early results of 18 patients treated on a compassionate use basis yielded encouraging results with successful device implantation in 16 patients and reduction of TR severity in all but one patient with a successfully implanted device [17]. Moreover, functional improvement on follow-up was evident in patients with successful implantation of the FORMA system. One patient was diagnosed with thrombosis of a spacer that occurred during hospitalisation for treatment of pneumonia four months post procedure. The patient had nontherapeutic international normalised ratios on warfarin; following antibiotic treatment and resumption of adequate anticoagulation the thrombus resolved within two months. Recently, the 30-day outcomes of 29 patients treated with the FORMA device within the US Early Feasibility Study were reported. Intraprocedural perforation of the right ventricle occurred in two patients and nine patients had at least one adverse event after 30 days of follow-up, including death, vascular injury, major or life-threatening bleeding and device-related surgery because of device perforation, migration or infection. Technical improvements are currently underway to minimise the risk of such adverse events in upcoming trials [15].
PASCAL
The PASCAL system (Edwards Lifesciences, Irvine, CA, USA) has undergone first-in-human, compassionate use experience in 23 patients with severe mitral regurgitation [18]. Recently, the device was successfully implanted in a patient with severe functional TR for the first time. The novel PASCAL system incorporates a spacer to fill the regurgitant jet area, paddles designed to avoid stress concentration on native leaflets, and clasps which allow for independent leaflet capture. Hence, the PASCAL device in a way integrates the mode of action of the FORMA and the TriClip devices.
Heterotopic caval valve implantation (CAVI)
The third group comprises techniques for heterotopic caval valve implantation, where either two dedicated self-expanding bioprosthetic valves (TricValve; P&F Products & Features Vertriebs GmbH, Vienna, Austria,) or balloon-expandable valves used to treat aortic stenosis (29 mm Edwards SAPIEN XT or SAPIEN 3; Edwards Lifesciences, Irvine, CA, USA) are placed in the inferior and superior vena cava [19]. The use of balloon-expandable valves requires prior implantation of a self-expanding stent in the inferior (and occasionally superior) vena cava to prepare a landing zone, given the large diameter of the vena cava and the hepatic vein confluence. The technique was recently tested in an excess-risk cohort and appeared safe and feasible with a high procedural success rate. Substantial in-hospital and 30 day-mortality rates were attributed to severe and non-device-related comorbidities in these patients [19].
Comparison between treatment modalities for functional TR
A retrospective analysis of patients with isolated TR suggests that severe TR is a serious disease characterised over time by a cumulative high rate of complications. Only 31% of patients were free from cardiac events (i.e., development of heart failure, atrial fibrillation, need for surgery, or death) at 15 years after diagnosis. In these patients, surgical repair of the tricuspid valve was feasible, with low operative mortality and marked symptomatic improvement in 88% of patients, indicating that surgical is superior to medical treatment in patients with isolated severe TR [20]. Prospective data comparing different treatment modalities are lacking. However, with the advent of transcatheter therapies targeting TR in patients at high surgical risk, the question may arise as to whether these techniques are indeed superior to optimal medical therapy alone.
Conclusion
Moderate-to-severe functional TR is a common problem in everyday practice associated with a considerable symptomatic burden and poor prognosis. Medical treatment options for TR are limited. Surgical tricuspid valve repair should be carried out in a timely manner according to recommendations given in current guidelines. A variety of transcatheter devices is currently in clinical testing to provide alternative treatment options for patients deemed at high risk for surgical tricuspid valve repair. Despite promising early data on safety and efficacy, further studies are needed for most of the devices targeting functional TR to validate the long-term effect on functional status and hard clinical endpoints. Finally, the question as to whether transcatheter TR treatment in elderly patients at high surgical risk is superior to medical treatment alone remains to be answered.















