Introduction
Peripheral artery disease (PAD) caused by systemic atherosclerosis is the major risk factor for cardiovascular disease and limb loss. Peripheral arterial disease of the lower extremities (LEAD) is characterised by reduced blood flow to the lower extremities and inadequate oxygen delivery due to narrowing of the arterial tree. Severe limb ischaemia (SLI) and intermittent claudication (IC) are the main clinical presentations in LEAD [1].
LEAD affects 12-14% of the general population and the prevalence is age-dependent [2]. In current epidemiology studies, it has been shown that the prevalence of this disease is 18% in the population aged between 60 and 90 years and that of intermittent claudication is 7% [2]. LEAD is seen more frequently in males and elderly individuals and is associated with increased mortality and morbidity [3], as well as with a high risk of cardiovascular events including major limb loss, myocardial infarction, stroke, and death [4].
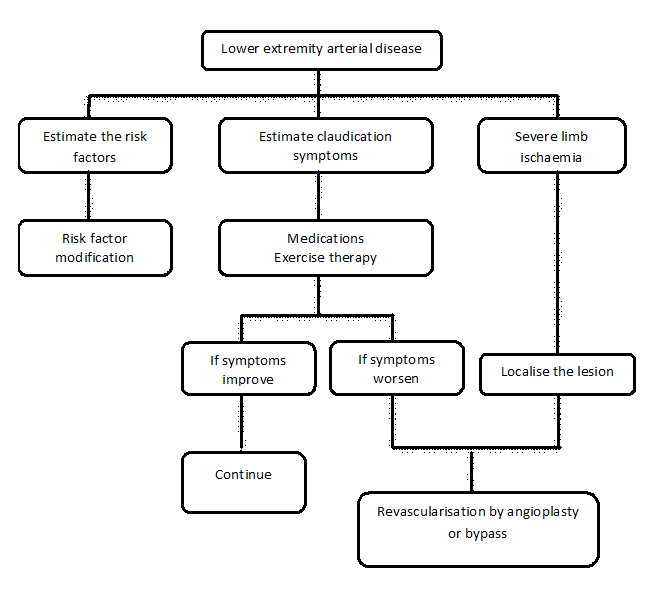
The majority of treatment strategies for LEAD are geared towards impeding progression of the disease and increasing blood flow [1] and should be based on the relief of clinical symptoms, to improve quality of life, to prevent progression of the disease, and to improve the short- and long-term prognosis. More than 70% of patients diagnosed with LEAD remain stable or improve with conservative management comprising lifestyle modifications and pharmacologic agents. The first-line treatment for LEAD focuses on behavioural changes, with reducing risk factors by healthy diet, smoking cessation, improving exercise performance [1,5], and medications such as antiplatelets (clopidogrel and/or aspirin), lipid-lowering agents (statins), antihypertensive agents and oral hypoglycaemic drugs or insulin. If behavioural changes and medications fail, the next line of therapy is revascularisation through endovascular or open surgery. Without successful revascularisation, those with critical ischaemia have a limb loss (amputation) rate of 80-90% in one year [6]. In the last decade, endovascular revascularisation for patients with LEAD has developed rapidly. Currently, due to low morbidity and mortality as compared with vascular surgery, many clinical centres prefer endovascular approaches for the treatment of LEAD; however, they retain the option of surgery in case of failure. Figure 1 summarises the treatment strategies for LEAD.
Figure 1. Treatment strategies for lower extremity peripheral artery disease.
Comparative difference of indications for medical, interventional and surgical treatment.
Medical treatment is used to reduce risk factors. It has been suggested that statins significantly improve the cardiovascular (CV) prognosis and can limit adverse limb events in patients with LEAD unless they are contraindicated or not tolerated. Thus, an ESC current guideline (diagnosis and treatment of peripheral arterial diseases) recommends them in all patients with LEAD [5]. Patients with hypertension and/or diabetes mellitus with LEAD will benefit from angiotensin-converting enzyme inhibitors (ACEIs)/angiotensin receptor blockers (ARBs) or calcium antagonists (CAs) because of their peripheral vasodilation effects and the improvement in walking distance. Beta-blockers (especially nebivolol and metoprolol) are indicated for patients with concomitant heart failure and LEAD. Antiplatelet agents are used in all patients with LEAD to prevent limb-related and general CV events. In addition, two drugs, such as pentoxifylline (by reducing blood viscosity) and cilostazol (a phosphodiesterase III inhibitor), may alleviate the symptoms of claudication caused by LEAD.
Exercise therapy is effective in patients with LEAD and improves quality of life and symptoms, as well as increasing walking distance. Walking exercise has even been compared to angioplasty and the results have been found to be similar [7]. If exercise therapy is impossible due to the severity of the disease in chronic limb-threatening ischaemia, it should be considered after successful revascularisation.
The selection of a proper treatment method for the revascularisation of LEAD is determined depending on anatomic position, comorbidities, risk factors, doctor's expertise and patient's choice. Revascularisation should be considered in order to improve symptoms of patients with IC and should be considered in patients who are showing signs of PAD in their other limb. Catheter-based endovascular approaches for LEAD offer the advantages of less morbidity, faster recovery, shorter hospital stay, less cost, and availability in outpatient facilities. Percutaneous balloon angioplasty and/or stenting is indicated for short-segment stenosis, whereas multi-segment and long-segment disease as well as occlusions are most effectively treated with operative revascularisation [5,8].
Using thromboendarterectomy is an excellent option for type I atherosclerosis (blue toe syndrome) because the offending degenerated atheroma is removed, leaving a clean, glistening surface that is soon covered by new functional endothelium. However, thromboendarteractomy should not be performed in distal external iliac and femoral lesions. A suitable choice in these types of patient is aortobifemoral bypass. In high-risk patients who are less likely to tolerate an abdominal operation, an extra-anatomic bypass may be used. This procedure is suitable for the treatment of aortic graft infections; however, the long-term patency of an extra-anatomic bypass is distinctly inferior to that of an aortobifemoral bypass. The anatomical location and extension of arterial lesions has an impact on revascularisation options. Therefore, the peripheral vascular tree can be conveniently divided into three regions: aortoiliac, femoropopliteal, and infrapopliteal.
Aortoiliac lesions
Aortoiliac occlusive lesions (AIL) occur commonly and are exposed easily by palpation of the femoral pulses. Any appreciable reduction in the femoral pulse indicates that there is a proximal obstruction. Obstructive lesions may be present in the infrarenal aorta, common iliac, internal iliac or external iliac artery, or combinations of the above-mentioned vessels. Patients with AIL usually have a poorer prognosis than those with other LEAD.
Although open surgical interventions have been considered the gold standard for the treatment of these patients, nowadays most specialists tend to favour endovascular interventions due to the low mortality and morbidity and higher patency rate in short- and long-term follow-up in selected patients. According to the anatomical lesion type, interventional treatment is primarily recommended for TransAtlantic Inter-Society Consensus (TASC) II type A-C lesions in the aortoiliac arteries in patients with IC. In the case of a TASC II type D lesion, interventional treatment should be attempted first, when there are severe comorbid diseases and the operator has excellent experience [1]. In these situations, the success rate of interventional treatment is more than 90%, and mortality and morbidity are low. Interventional procedures in the iliac artery result in better patency rates than surgical treatment [9].
Balloon angioplasty
A stenotic or occlusive lesion of the aortoiliac artery can be treated successfully by percutaneous transluminal angioplasty (PTA) alone. When PTA alone is technically unsuccessful, additional stent placement is indicated. PTA with selective stenting (SS) and primary stenting (PS) resulted in analogous outcomes at the stage of peripheral arterial occlusive disease according to Rutherford's criteria, improving clinical symptoms, and increasing quality of life as well as patency rate. The STAG trial showed that PS placement for iliac artery lesions (occlusions) improved technical success and lowered major procedural complications (mainly distal embolisation) when compared with balloon angioplasty [10]. If the lesion is shorter than 3 cm, and/or there is concentric stenosis, favourable results may be expected with balloon angioplasty only. That is why PTA is an appropriate choice, provided it is performed by a skilled vascular interventionist and the arterial disease is localised in a short vessel segment. If there is a lesion with a long segment or multiple stenosis, occlusion or calcification, using balloon angioplasty only will not give a favourable outcome due to the risk of dissection or other complications. In these cases, stent replacement is recommended [1].
Stent placement
Stent placement is effective in iliac artery lesions because the long-term patency rate of iliac artery stents is excellent. Stenting in the iliac artery has low complications and it is not invasive. Stent placement can be used either as a primary treatment or after angioplasty in selected patients. In general, the more extensive and complex the occlusive disease, the more likely a primary stent placement will improve patency due to the lower risk of embolisation, arterial rupture, or dissection than using balloon angioplasty only. In ostial lesions with high calcium, using balloon-expandable stents is more effective because of the greater radial strength and resistance to crush which allows successful expansion and retention of vessel diameter. A recent randomised controlled trial (RCT) by Goode et al demonstrated that primary stenting can improve the technical success rate and lower complications in patients with iliac artery occlusive lesions more than balloon angioplasty [10]. In addition, primary stenting should be considered in complex lesions such as calcified, eccentric, or ulcerated lesions, or in plaque with spontaneous dissection. Ye et al reported that the long-term patency rate of primary stent placement was better than selective stent placement for common and external iliac artery cases [11].
Selective stent placement after angioplasty is suitable for more than 30% residual stenosis or in flow-limiting dissections. Initial technical success for iliac stenting varies from 90% to 100% and depends on the extent of the disease, with more complex lesions having lower initial technical success rates. The success rate with PTA in focal segmental occlusive lesions in LEAD is up to 94%, with 5-year patency rates of 78-88%. In iliac arterial occlusions, PTA gives success rates of 91-98%, with 5-year patency rates of 52-87%. Patients treated with PTA and selective stent placement in the iliac artery had a better outcome on short- and long-term prognosis for symptomatic success compared with patients treated with primary stent placement according to the results of the Dutch iliac stent trial [12]. Compared to PTA alone, stenting improves 3-year patency by 26% in iliac artery lesions; however, if patients continue to smoke, the patency rates are reduced by approximately 50% [13]. For complete occlusion of the aorto-bi-iliac artery, bypass surgery is indicated [13]; however, stent replacement, which has a 1-year patency rate of 85% and a 3-year patency rate of 66%, should be considered in high-risk patients in surgery [14].
Surgery
Although endovascular treatment has many advantages in these vascular lesions, surgery continues to have an important role for selective patients. Bypass surgery is indicated for total occlusion of the aorto-bi-iliac artery. In patients presenting with CLI due to AIL and who are suitable either for surgery or angioplasty, a strategy of bypass surgery and balloon angioplasty is associated with similar outcomes in terms of amputation-free survival. However, surgery is more expensive than angioplasty in the short term [15]. Extension of the disease to the subrenal aorta and the pelvic circulation status influence the choice between end-to-end and end-to-side aortic graft configuration; however, there are no clear differences in the long-term outcomes between the above-mentioned configurations.
In a systematic review conducted by Indes et al comparing the clinical outcomes of endovascular treatment and open bypass surgery for AIL, it was shown that the endovascular choice was associated with shorter hospital stays, lower complications, and reduced 1-month mortality, whilst open bypass surgery was associated with higher primary and secondary patency rates in terms of short- and long-term prognosis [16]. Another study, by Salvatori et al, demonstrated that the 5-year primary patency rate with extra-anatomic bypass for AIL was 19-50% for axillobifemoral bypass and 44-85% for femorofemoral bypass [17].
Aortobifemoral bypass is considered the gold standard for patients with low surgical risk, limiting claudication, and TASC II type C-D lesions. In addition, aortoiliac occlusive patients with a high surgical cardiovascular risk and limb-threatening ischaemia with TASC II type C-D lesions can be considered for extra-anatomic bypass [1].
Femoropopliteal lesions
Femoropoliteal lesions (FPL) are common in intermittent claudication. If the blood flow in the profound femoral artery is normal, claudication may be relieved with exercise therapy. If revascularisation is indicated, the first choice should be endovascular therapy with PTA and/or stenting.
The interventional method is indicated when stenotic lesions are of <10 cm in length and occlusive lesions are of <5 cm in length [5]. However, in cases of stenosis/occlusion more than 10 cm, preference should be given to bypass surgery due to the better long-term patency rate. Bypass surgery is the gold standard for the treatment of critically stenotic or occlusive lesions in the femoropopliteal artery (FPA), especially using the greater saphenous vein.
Balloon angioplasty
In general, patients with FPL will benefit from balloon angioplasty. Kasapis et al reported that the success rate was 98-100% in the stenotic group and 81-94% in the occlusion group with IC. In patients with severe limb ischaemia, these rates were 69-88% and 62-75%, respectively [18]. It was reported that the 1-year patency rate was much lower when using balloon angioplasty if the length of the lesion was >10 cm. Also, the long-term patency rate of balloon angioplasty in the FPA was lower compared to that of surgical bypass using veins. Complete occlusive lesion and severe limb ischaemia patients had worse outcomes than claudication patients with simple stenosis. The 3-year patency rates of stenosis and occlusion were 61% and 48%, respectively, in the intermittent claudication patient group, and 43% and 30%, respectively, in the severe limb ischaemia group [1,18].
Stent placement
In femoropopliteal artery cases, interventional treatment with stent replacement should be preferred even in long and complex lesions due to technological developments and the low risk of the procedure. Thus, interventional recanalisation is recommended for TASC II type A-C lesions in femoropopliteal arteries with IC. Different types of stent have been used in FPL. After stent replacement, restenosis due to neointimal hyperplasia or stent fracture may occur. Primary stent placement is not suitable for short lesions, because there was no observed significant difference in terms of patency rate compared to balloon angioplasty during the long-term follow-up period. In a clinical trial involving 141 patients with intermittent claudication or CLI of less than 5 cm in lesion length randomised to balloon angioplasty or balloon-expandable stent placement, there was a significant difference in the technical success rate (84% vs. 99%) [19]. In one trial conducted by Martin et al it was demonstrated that primary implantation of self-expanding nitinol stents for the treatment of lesions of the superficial femoral artery was associated with superior anatomical and clinical intermediate-term results in comparison with the currently recommended approach of balloon angioplasty with optional secondary stenting [20].
Even though the long-term patency rate of surgical bypass surgery is significantly superior to that of angioplasty for the treatment of femoropopliteal artery lesions, angioplasty for femoropopliteal artery lesions has many advantages in terms of lower morbidity, mortality and shorter hospital stay if the artery remains open for at least 6 months in CLI, which is enough time for wound healing.
Surgery
The superficial femoral artery (SFA) and popliteal artery (PA) are potentially compromised by the high calcium content within the plaque and vessel wall, long length of lesions, and unique dynamic forces found within these arteries; therefore, most of these occlusions are TASC II type C and D lesions, in which surgery is still recommended as the preferred treatment modality. Surgical bypass has been established as the gold standard revascularisation method for the occluded superficial femoral artery (SFA) in IC patients in whom medical management failed or in patients presenting with CLI [15]. The 5-year primary patency rate of femoropopliteal above-the-knee bypass with the autogenous saphenous vein is 70%, while the primary-assisted patency can be improved to approximately 80% [21].
Infrapopliteal artery
Infrapopliteal artery lesions (IPL) are a common occurrence, especially in elderly patients, smokers, and in patients with diabetes mellitus. According to the ESC guidelines (diagnosis and treatment of peripheral arterial diseases), there is increasing evidence to support a recommendation for angioplasty in patients with IPL [5]. Endovascular treatment is indicated in severe claudication that alters lifestyle and does not respond to medical treatment, and for patients who are suffering from CLI in the IPL.
The most important part of the procedure in the infrapopliteal artery is the passage of a guidewire for the blood vessel at the lesion site. Lesion sites can be classified as 1) stenotic lesions: i) without calcification; ii) calcification with an average length of 10 cm or more; 2) occlusion.
Balloon angioplasty
Transluminal balloon angioplasty (TBA) is widely used in IPL with CTO or chronic stenotic lesions. PTA with low-profile balloons is the current gold standard in the endovascular treatment of IPL. The diameter of the balloon is defined according to the diameter of the target arteries and should not exceed 3 mm because of the risk of rupture of the targeted artery; also, the length of the balloon catheter has to include the whole lesion site. In calcified lesions, a cutting balloon catheter can be used with low pressure. In long and multiple lesions, angioplasty will be performed with a long balloon catheter in order to reduce the procedural time and radiation exposure. Short infrapopliteal bifurcation lesions can be treated with cutting balloon angioplasty. However, the patency rate remains low in PTA patients in the infrapopliteal artery due to the small diameter of the vessel and flaccid blood flow. As Romiti et al reported, the patency rate after PTA in the infrapopliteal artery is about 50% [22].
Stent placement
Stenting with self-expanding and balloon-expandable stents is not suitable in the infrapopliteal artery because the lesions are often longer than 10 cm, multiple, the artery is thin, and blood flow is slow. If the lesion does not exceed 50 cm in the infrapopliteal artery, drug-eluting stents can be considered. Furthermore, stenting can be considered in flow-limiting dissections. The DESTINY trial studied DES and BMS in the infrapopliteal artery and demonstrated that the patency rate in one year was better in the DES group (85% vs. 54%, p<0.0001), whereas there was no significant difference observed in terms of late lumen loss, while there was a reduction in mean in-stent diameter stenosis (21% vs. 47%; p<0.0001) [23].
Surgery
In long occlusions of the crural arteries, bypass using an autologous vein gives superior long-term patency and leg survival. There is a great deal of evidence supporting the use of the great saphenous vein for bypass surgery; thus, current ESC guidelines recommend bypass surgery in these patients [5]. Table 1 summarises treatment options for LEAD by anatomical location.
Table 1. Treatment options for LEAD according to the anatomical location of the lesions.
| Lesions |
Balloon angioplasty |
Stent placement |
Surgery |
|---|---|---|---|
|
Aortoiliac lesions |
Lesions less than 3 cm, and/or concentric stenosis |
Calcified, eccentric, ulcerated, or plaque with spontaneous dissection with primary stenting; residual stenosis more than 30% and/or flow-limiting dissections with selective stenting |
Total occlusion and patients with low surgical risk, limiting claudication and TASC II type C-D lesions |
| Femoropopliteal lesions |
Stenotic lesions up to 10 cm in length |
Stenotic lesions of <10 cm in length and occlusive lesions of <5 cm in length |
Critically stenotic or occlusive lesions |
| Infrapopliteal artery |
Calcified lesions with cutting balloon catheter, long and multiple lesions with long balloon catheter, short infrapopliteal bifurcation lesions with cutting balloon angioplasty |
Lesions up to 50 cm in the infrapopliteal artery with drug-eluting stents |
Long occlusions of the crural arteries |
Focusing on decision making for treatment
Precise risk stratification for patients who present with LEAD is important to improve clinical decision making and to determine the most appropriate therapy for individual patients. Elderly patients with diabetes mellitus should be examined regularly due to the high risk of LEAD. Decision making should be based on a multidisciplinary approach. When making a decision, one should take into account the clinical symptoms, prognostic life expectancy, morphological classification of the lesion, comorbidities such as renal disease, coronary artery disease, and diabetes mellitus, and risk factors. Patients with intermittent claudication should be considered for revascularisation to improve the symptoms in the absence of other disease that would limit exercise improvement such as chronic pulmonary disease, angina pectoris, chronic heart failure, or orthopaedic limitations. PTA and/or stenting is recommended for TASC type A-C lesions and bypass is appropriate for patients with TASC type D lesions. As mentioned below, patients with TASC type C/type D iliac lesions, a stenotic ipsilateral superficial femoral artery, ulcer/gangrene, smoking history, and chronic renal failure with haemodialysis should be followed carefully after endovascular iliac revascularisation. These risk factors could be considered indications for primary or selective stenting.
Future prospects of PTA/stenting vs. surgery
Several critical breakthrough technologies account for the remarkable progress in the field of endovascular surgery for the treatment of peripheral artery disease in the past three decades: intra-arterial stents have increased success rates and reduced restenosis, and adjunctive antiplatelet therapy has lowered periprocedural complications. The development of DES is one of the major revolutions in the field of endovascular surgery. The ideal drug to prevent restenosis must have an antiproliferative and antimigratory effect on smooth muscle cells, inhibit the anti-inflammatory response after balloon-induced arterial injury but, on the other hand, it must also enhance re-endothelialisation in order to prevent late thrombosis. Drug-eluting stents with sirolimus have shown better outcomes in short-term and long-term follow-up. Angioplasties with drug-eluting stents minimise neointimal hyperplasia; as a consequence, the restenosis rate may lowered up to 0%. However, one should consider the risk of flow-limiting intimal dissection from the expansion of the balloon catheter. With the present DES the focus has shifted to further development towards increasing the long-term safety and efficacy of the stents. The next generations of DES should further improve endothelialisation.
Despite advances in endovascular treatment and surgery of LEAD, there are still limitations that bring about less than optimal results. Significant calcification, comorbidities, long occlusions, and multiple risk factors can interfere with interventions and worsen outcomes of the procedures. The role of other agents and techniques with potential benefits (e.g., laser ablation of atheroma and thrombus obstructing the inner lumen of the blood vessel, atherectomy devices, cutting balloon angioplasty, arteriovenous fistula, tyrosine kinase inhibitors, abciximab) as well as drug-eluting balloons and new stent designs may be tested in the future and will further advance our knowledge, resulting in improved outcomes both technically and clinically.
Conclusion
The treatment strategy should be started with modification of the risk factors and drug therapy in LEAD. Statins and antiplatelets are indicated in all patients for life unless they have contraindications. ACEIs, ARBs, CAs, beta-blockers, pentoxifylline and cilostazol are indicated in most patients taking into consideration their risk factors and comorbidities. As recommended in current ESC guidelines (diagnosis and treatment of peripheral arterial diseases), cardiovascular prevention and exercise training are the cornerstone for the management of patients with intermittent claudication. If routine activity is severely impaired, revascularisation can be recommended. Revascularisation for LEAD is an appropriate therapy for selected patients with disabling symptoms, after a careful risk-benefit analysis. Treatment should be individualised based on risk factors, comorbid conditions, degree of functional impairment, as well as anatomic factors. Endovascular treatment is favoured with aortoiliac disease and for selected patients with femoropopliteal disease at low risk. In high-risk patients with calcification, small-calibre arteries, diffuse infrainguinal disease and failed prior endovascular procedures, bypass surgery should be preferred.