Keywords
alternative therapies, cell-based therapy, critical limb ischemia, gene therapy, lower extremity arterial disease, limitations, regeneration
Background
Because of the increasing burden of arteriosclerotic vascular disease and the associated challenge to healthcare systems, the field of lower extremity arterial disease (LEAD) has recently gained momentum. As a result of the cumulating multimorbidity in an ageing population, LEAD has been spreading significantly over Europe with almost 40 million individuals suffering [1]. The progression of peripheral artery disease (PAD) is even accelerated in certain comorbidities such as diabetes and renal insufficiency which themselves constitute rising entities in the western world nowadays. Even today, exercise therapy and medical secondary prevention remain the therapeutic cornerstones [2]. Despite these, over time LEAD often progresses to refractory chronic limb-threatening ischaemia (CLTI) lacking options for revascularisation. In Europe, the incidence of CLTI accounts for 0.5 to 1 cases per 1,000 per year, and CLTI individuals are prone to limb loss and related mortality (25% and 30% per year, respectively) [1]. Consequently, the individual faces impaired quality of life, amputation, working disability, and associated morbidity and mortality, and the medico-social system faces increased healthcare costs.
Revascularisation of relevant stenosis/occlusions is key to enhancing quality of life. Modern strategies comprise endovascular, open surgical and hybrid procedures [2]. Independent of the revascularisation strategy, diffuse disease progression as well as restenosis/occlusion remain the Achilles’ heel in revascularisation despite increasing understanding of disease and technical advances. The risk of restenosis increases by the location of target lesion from proximal to distal. Below-the-knee bypassing also shows limited patency. The ultimate option of pharmacological vasodilation of the microcirculation is barely supported by satisfying evidence and is not recommended [2].
Given the unmet dilemma of the lack of treatment options particularly in refractory CLTI, alternative (regenerative) treatment options seem promising. As discussed below, the present options have been of limited success in the clinical arena. Future research identifying novel therapeutic options may lead to other therapeutic options with prolonged relief of symptoms and/or improvement of vascular prognosis.
Alternative therapies in LEAD/CLTI – status quo
Triggered by promising preclinical and early-phase, small-scale clinical studies, the concept of ischaemic tissue repair or regeneration has been developed eagerly. The therapeutic goal is to stimulate the expansion of existing vessels (vasculogenesis), the growth of new vasculature (neovascularisation) and/or the formation of collaterals (arteriogenesis) to improve perfusion of ischaemic limbs. For this purpose, the application of angiogenic growth factors by means of virus and/or proteins as well as stem or progenitor cells has predominantly been advanced. The underlying reasons for the failure of randomised large-scale studies in gene and cell-based therapies appear to be multifaceted and complex. While intense debate is ongoing and research activity to improve these alternative concepts and to overcome identified limitations is on the way, at present the ESC does not recommend using gene and cell-based therapies (class III). Below we will summarise the current status of these therapies, discuss conceptual limitations, and address potential solutions for practising physicians interested in vascular medicine. The focus will be exclusively on randomised, controlled phase II/III studies.
Gene and protein therapy
Stemming from preclinical work and their involvement in tissue ischaemia, several candidate genes have been proposed for angiogenic gene therapy in LEAD/CLTI patients. Conceptually, there are two established ways of utilising the proangiogenic capacity of a therapeutic factor in man - the encoding plasmid/virus vs. the synthesised protein.
Vascular endothelial growth factor (VEGF)
The isoforms VEGF165 and VEGF121 have been evaluated in LEAD/CLTI patients. While there was more angiographic vascularity after intra-arterial application of AdVEGF165 in one study [3], studies using intramuscular injection of VEGF were overtly negative (RAVE trial). Notably, Kusumanto et al reported an improvement of secondary endpoints (clinical and functional improvement) with VEGF165 plasmid in diabetic PAD patients [4].
Fibroblast growth factor (FGF)
Of the FGF family, only FGF1 and FGF2 have been studied in human cardiovascular gene therapy. The intramuscular injection of FGF1 plasmid did not improve the primary endpoint in the PM 202 (pO2) and the TALISMAN 201 study (ulcer healing), but as a positive signal the amputation rate was improved in FGF1-treated no-option CLTI patients [5]. Infusion of FGF2 increased walking time while quality of life and ankle brachial index (ABI) remained unchanged (TRAFFIC) [6].
Hepatocyte growth factor (HGF)
HGF is a potent mitogen with angiogenic properties. In LEAD/CLTI patients, intramuscular injection of HGF plasmids led to an increased transcutaneous oxygen pressure (TcPO2) at higher doses while secondary endpoints (ABI, toe brachial index [TBI], claudication, wound healing, major amputation) were unchanged (HGF-STAT) [7]. Similarly, Shigematsu et al reported a functional and clinical improvement (pain, ulcer size, ABI, quality of life) after HGF injection of Rutherford 4/5 CLI patients [8]. In a more severely sick collective of Rutherford 5/6 CLI patients, HGF therapy resulted in improved rest pain and improved TBI, whereas wound healing and amputations were unchanged (HGF-0205) [9]. Use of another HGF vector (VM202, genomic complementary DNA hybrid of the human HGF gene, HGF-X7) has been feasible and safe at the phase I level, but the outcomes from phase II/III studies are pending.
Developmentally regulated endothelial locus (Del-1)
Del-1 is an extracellular matrix protein expressed during embryonic development and ischaemia. In PAD patients, the intramuscular injection of Del-1 plasmid was safe but did not show a significant improvement of either endpoint compared to controls (DELTA-1) [10].
Hypoxia-inducible factor-1α (HIF-1α)
HIF-1α regulates cellular responses to hypoxia and is involved in angiogenesis. The intramuscular injection of adenoviral HIF-1α in claudication PAD patients was not effective (WALK) [11].
Critical appraisal
Despite prior evidence on the safety and bioactivity of angiogenic gene therapy, randomised controlled trials failed to generate convincing evidence. The following aspects, at least in part, may be considered:
- Preclinical animal models do not properly model elderly, multi-morbid cardiovascular patients who are generally enrolled in gene therapy trials. Species-, age-, and health-related differences may have impaired efficacy in man.
- Vectors (virus, plasmid) may fail to transfect (i.e., transfer and integration of carried genomic sequence into local tissue). Currently available vectors entail a signature of advantages and disadvantages (e.g., transfection efficacy, immunogenicity, adversity, risk of malignancy). The ideal vector may be tissue specific and remains yet to be determined.
- Ideal route of application is unknown.
- Precise “dosing” is difficult. Optimal dose, timing and repetition are undetermined. A prolonged angiogenic stimulation and/or repetitive treatment may be worthwhile.
- We may be lacking accurate endpoints as the established measures mainly reflect bulk blood flow from conduction vessels. A measure of the microcirculation and/or tissue perfusion is required.
- The majority of angiogenic gene therapy studies have been conducted in “end-stage” or “no-option” patients due to risk/benefit considerations. There may, however, be a threshold of disease severity, when the effect of a therapy cannot be leveraged anymore.
Cell-based therapy
Different populations of stem and progenitor cells have been evaluated in the preclinical setting for the indication of LEAD/CLTI. After some success in early-phase clinical studies, the confirmation of their therapeutic value in subsequent randomised controlled phase II/III trials is pending. Given the fact that many studies, at least in part, have generated interesting insights or surrogates, another look at favourable signals may be worthwhile.
Bone marrow-derived mononuclear cells (BM-MNC)
In a multicentre study conducted by Iafrati et al, percutaneous BM-MNC injection showed trends towards improvement in amputation, pain, quality of life, Rutherford classification, and ABI in no-option CLI patients compared to controls [12]. Formally missing its primary endpoint (change of ABI), intra-arterial application of cells for CLTI led to improved ulcer healing and reduced leg pain in CLI patients (PROVASA) [13]. In a recent meta-analysis summarising 201 patients, the amputation rate was reduced and deferred after intramuscular BM-MNC injection in Rutherford 4/5 CLI patients [14]. In another smaller RCT studying CLTI patients with foot ulcers, local BM-MNC injection was associated with limb salvage compared to controls. This effect was associated with improved ABI and TBI [15]. In the recent JUVENTAS trial, which evaluated intra-arterial application of BM-MNC in 160 patients with non-revascularisable limb ischaemia, neither any primary nor any secondary endpoint was met [16].
Peripheral blood-derived mononuclear cells (PB-MNC)
Mononuclear cells can also be isolated from peripheral blood, but only after mobilising the cells from the bone marrow to the blood stream do sufficient numbers become available. PB-MNC mobilised by granulocyte colony-stimulating factor (G-CSF) and locally injected into ischaemic limbs resulted in better ulcer healing as well as improved ABI and limb perfusion in diabetic patients [17]. Another study on diabetic patients also showed attenuation of ischaemic clinical signs (pain, neurological symptoms, wound healing), improved perfusion (ABI) and also fewer lower-limb amputations [18].
Comparison of bone marrow-derived vs. peripheral blood-derived mononuclear cells (BM-MNC vs. PB-MNC)
Other remarkable studies in the field were designed to compare different types of mononuclear cells. Intramuscular treatment of CLI patients with BM-MNC was superior to PB-MNC as assessed by TcPO2, rest pain, and pain-free walking time four weeks after intramuscular treatment (TACT) [19]. For some measures, this effect lasted for two years of follow-up (leg pain, walking distance, ulcer size), while ABI and TcPO2 were not different anymore [20]. As assessed by skin temperature, ABI and rest pain BM-MNC were more effective than PB-MNC in patients with arteriosclerosis obliterans, while there was no difference in TcPO2, pain-free walking distance and amputation rates [21]. Heterogenous data were generated in a small collective treated with MNC because of inflammatory vasculitis and necrotic and painful digits. While there was no effect on TcPO2 and thermogram, blood flow measured by digital subtraction angiography (DSA) was increased with BM-MNC [22]. Of note, comparing the efficacy of bone marrow mesenchymal stem cells (BM-MSC) vs. BM-MNC in type 2 diabetes CLI patients with foot ulcers, the effect of BM-MSC on various measures was stronger (painless walking time, ABI, TcPO2, MR angiography) [23].
Marker-selected progenitor cells
Enriching progenitor cells based on certain surface markers was previously thought to increase their therapeutic efficacy. The local injection of different doses of CD34+ cells exerted favourable trends but did not reach significance in amputation rates at six or 12 months (ACT34-CLI) [24]. CD90+-enriched so-called tissue repair cells did not substantially affect wound healing in diabetic foot patients [25]. Further characterising CD34+ cells based on aldehyde dehydrogenase brightness did attenuate amputation rates relevantly when intramuscularly injected in CLI patients (CLI-101) [26]. Aiming at a broader strategy, Powell et al used a patient-specific, expanded, multicellular therapy (so-called Ixmyelocel-T) in Rutherford 4-6 CLI patients. This intramuscular therapy was associated with fewer treatment failures of CLI as determined by major amputations, all-cause mortality and doubling of wound surface (RESTORE-CLI) [27].
Critical appraisal
Similar to the situation with gene therapy, conceptual and practical limitations have been identified that may have resulted in mixed results and, in turn, hampered the clinical establishment of cell-based therapies:
- Altered viability and retention of injected cells after local application.
- Impaired functionality and, in turn, the regenerative capacity of autologous stem/progenitor cells harvested from elderly, cardiovascular patients.
- Difficulties with timing, dosing and repetition of cell therapies.
Intensive scientific efforts are being undertaken to overcome some of these limitations to leverage the full therapeutic potential. Modification of stem/progenitor cells prior to injection, combination with gene therapy, co-application with biomaterials, integration into tissue engineering and others are under investigation to improve the therapeutic potential [3]. For now, cell-based therapies are not recommended for PAD/CLI.
Emerging therapeutic options on the horizon
While medical needs in LEAD/CLTI remain unmet, the holy grail in cardiovascular medicine is to recover or rebuild ischaemic tissue by biological means (i.e., ischaemic tissue repair or regeneration). Alternative strategies targeting ischaemic tissue have been proposed (Figure 1, Table 1), but the majority have not fulfilled their expectations at the bedside (i.e., translation). Based on preclinical and early-phase clinical work, the following novel therapeutic options are about to emerge in clinical practice:
Figure 1. Alternative therapeutics.
This schematic provides an overview of established and emerging options for lower extremity arterial disease (LEAD) and chronic limb-threatening ischaemia (CLTI). The combination of options may have synergistic effects.
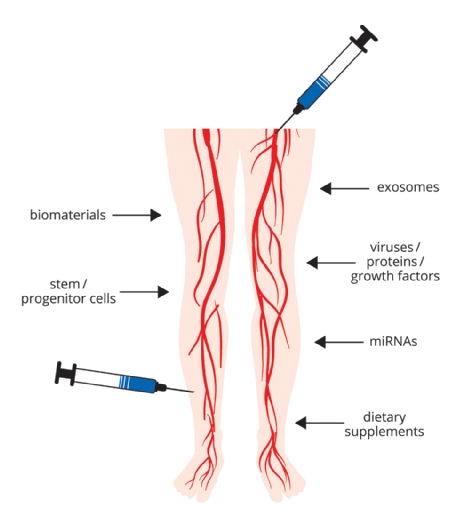
Table 1. Emerging therapeutic options.
Glossary of emerging therapeutic targets and mechanisms of action.
Target | Mechanism of action |
|---|---|
biomaterial | natural or synthetic compound to enhance delivery and efficacy of alternative therapeutics |
dietary supplements | natural compounds that beneficially affect vascular biology and function, e.g., via NO |
exosome | biological microvesicle containing a multitude of angiogenic factors |
gene therapy (virus, protein, growth factor) | modification of angiogenic genes by use of encoding or virus, recombinant human proteins |
microRNA | small non-coding RNA molecules that regulate the expression of genes in organisms. The therapeutic modulation of miRNA expression by silencing or overexpression influences the expression of target genes |
stem/progenitor cells | structural and/or paracrine induction of neovascularisation |
dietary supplements | natural compounds that beneficially affect vascular function, e.g., via NO |
Nutritional supplements
It is a common notion that vascular structure and function are altered in the diseased vasculature. Endothelial dysfunction, for example, has been extensively proven in cardiovascular patients. By far the most prominent factor involved in vascular homeostasis is nitric oxide (NO). Exercise, modification of cardiovascular risk, and medications of secondary prophylaxis are known to improve NO availability. Several ongoing clinical studies are exploring the dietary supplementation of natural substances that increase NO levels or restore the endothelial function in intermittent claudication PAD: natural precursor of L-arginine L-citrullin (ClinicalTrials.gov Identifier: NCT02521220), Neo40 (ClinicalTrials.gov Identifier: NCT02934438), cocoa (ClinicalTrials.gov Identifier: NCT02876887, NCT01064583), sodium nitrite (ClinicalTrials.gov Identifier: NCT01401517). As the results of these studies are pending, their routine use for the indication of LEAD/CLTI cannot be advocated despite absent negative effects related to NO. Any future proof of benefit will easily outweigh potential risks. Notably, the efficacy of NO modification may be limited to stable PAD while devastating vascular homeostasis in CLTI may not be affected by this strategy anymore.
Exosomes
This emerging concept leverages that subcellular microvesicles, namely exosomes, contain and export a multitude of synthesised factors/proteins from inside the cells to the outer interstitial space. In preclinical work, those exosomes derived from stem cells have been characterised as paracrine effectors of angiogenesis, recovery of injured cells, and attenuation of limb ischaemia. Interestingly, stem cells have been observed to ramp up the release of these microvesicles under hypoxia. Given rapid technical advances in the field with isolation and expansion of stem cells, the viability of the option of reprogramming and the autologous source of material, this therapeutic use of autologous exosomes is exciting. Again, appropriate clinical studies will have to prove the clinical value of this concept.
Small non-coding RNAs
As we have learned in medical school, proteins are coded by our genome. Exciting research in recent years, however, has shaken up this simplistic notion. It has been shown that parts of the ribonucleic acid (RNA) are non-coding, so-called non-coding RNA (ncRNA). While not participating in coding, these RNA subtypes, e.g., microRNAs (miRNAs), are heavily involved in the regulation of the gene expression. Since the milestone identification of miR-92a as a regulator of angiogenesis in ischaemic tissue, several preclinical studies have investigated the regulation and functionality of miRNAs in limb ischaemia. Therapies employing miRNAs aim at suppression or overexpression of specific miRNAs, which in turn modify genes. The systemic usage of miRNAs or their mimics/antagonists, however, is at present limited by degradation (serum nucleases), poor cellular uptake, quick renal clearance and possible side effects. Although advances in RNA technology or cutting-edge modes of delivery (e.g., vesicles, exosomes, biomaterials, microparticles) may facilitate the efficacy of systemic miRNA application, local delivery of miRNA via the intramuscular or transvascular route shows a favourable risk/benefit profile at present despite leakage from injection sites. Silencing of certain miRNAs via local (intramuscular) injection is a feasible strategy to improve angiogenesis. While the therapeutic use of this fascinating concept has already been realised with success in other fields, clinical studies in LEAD/CLTI are to our knowledge still pending.
Conclusion
The growing pandemic of LEAD/CLTI threatens our medical and socioeconomic system. Because standard care, including exercise, medical treatment, and revascularisation, remains ultimately limited, patients continue to face devastating quality of life, working disability, morbidity and mortality. Thus, alternative therapeutics have been developed with great enthusiasm. The predominant strategies, angiogenic gene and protein as well as cell-based therapies, have generated unconvincing results when translated into clinical practice. Although optimisation of study design and the solution of technical limitations may enhance efficacy, neither therapy is recommended for clinical practice at present. Their combination with novel tools such as biomaterials may open new avenues to ischaemic tissue salvage. In addition, emerging therapeutics such as nutritional supplements, miRNAs or exosomes constitute promising options at the early stages of development.















