Juvenile mitral stenosis
Introduction
The original descriptions of the natural history of mitral stenosis (MS) from developed countries noted a minimum latent period of two decades between an initial attack of rheumatic fever (RF) and the development of symptoms of MS [1,2]. Longitudinal echocardiographic studies reported an average decline in mitral valve area (MVA) of approximately 0.09 cm2/year [3]. As RF commonly develops between the ages of 5 and 15 years, it was extremely rare to encounter symptomatic severe MS in childhood or adolescence in the developed countries even during the era when RF was rampant.
In 1963, Sujoy Roy et al drew attention to the occurrence of symptomatic severe MS of rheumatic aetiology in India among individuals who were below 20 years of age [4]. They coined the term “juvenile mitral stenosis”. Their classic paper described how juvenile mitral stenosis (MS) differed from the presentation in adults. Further, they drew attention to the fact that congestive heart failure (CHF) in those <20 years was not synonymous with active rheumatic carditis but could be a result of mechanical consequences of severe MS which needed to be relieved.
Epidemiology and aetiopathogenesis
Juvenile MS has been predominantly reported from developing countries in South-East Asia [4-7], Africa [8] and the Middle East [9]. Exact figures on the prevalence and incidence are not available. Based on echocardiographic studies, the prevalence of rheumatic heart disease (RHD) was 20 to 30 per 1,000 [10] school children in these countries. Thus, there is a large latent pool of RHD. In developing countries, as many as 25% of patients with pure or predominant MS are below 20 years of age [11] and 10% are <12 years [5]. Unlike the female preponderance in adults, juvenile MS affects both sexes equally or has a slight male preponderance (1.2-1.6:1) (Table 1). It has been postulated that hormonal changes after puberty may increase the incidence of cicatrisation of the mitral valve.
Studies of juvenile MS [4-9] have noted a short latent time interval with rapid progression of MS leading to serious disability early in life. Roy et al noted that 70% of the patients with a history of RF had developed symptoms of MS within five years of the first attack [4]. In another study, this duration was less than two years in 71% cases and in 5% of cases it was as short as three months [7]. The youngest reported case of rheumatic MS was in a two-and-a-half-year-old boy who underwent percutaneous transvenous mitral commissurotomy (PTMC) at the age of four [11]. It is believed that, on average, it takes at least two years for the development of severe MS in these countries [12]. What leads to the early appearance and rapid progression of MS in this group of patients is not clear. Recurrent attacks of RF were reported in 28-30% of cases (Table 1). The initial attacks of rheumatic carditis may have been more severe possibly due to the late or inadequate treatment of acute streptococcal pharyngitis [9]. Malnutrition (low intake of proteins and micronutrients), overcrowding, low socioeconomic status, inadequate penicillin prophylaxis and increased genetic susceptibility linked to the human leukocyte antigen (HLA) system may have been contributory [6,13].
The term juvenile MS is reserved for MS that is rheumatic in origin. Other rare causes of MS that are seen in the young are congenital MS, systemic lupus erythematosus, mucopolysaccharidoses of the Hunter-Hurler phenotype, Fabry disease, etc. With the advent of modern-day imaging (namely echocardiography), differentiation of the aetiology has been simplified.
Pathologic changes
The left atrium (LA) is only moderately enlarged and less than ideal at the time of surgery in half the cases [14]. This is due to the rapid progression of the disease that gives no time for LA distension. As a result, the brunt of the back pressure falls on the pulmonary vasculature, leading to high degrees of pulmonary venous and arterial hypertension. The pulmonary arteries are dilated and turgid with relatively small aorta. Histopathological changes on lung biopsy specimens noted pronounced medial hypertrophy and intimal thickening of small muscular pulmonary arteries, arterioles and venules, alveolar capillary sclerosis, and hypertrophic smooth-muscle bands in the distal respiratory passages [4].
Atrial thrombi (0-4%) [6,7] and mitral valve (MV) calcification (0-6%) are rare. The MV is often tightly stenosed (<0.5 cm2) [6]. Subvalvular fusion is found in 30-50% cases [5-7]. Two unique types of pathology have been noted in juvenile MS. First are elastic valves that stretch like India rubber [7]. Second are “button-hole”, densely sclerotic valves with fibrous tissue that need great force to fracture. However, the most common pathologies are commissural, cuspal or chordal fusion or a combination thereof [6,7]. With improvement in socioeconomic conditions, there is an attenuation in the disease severity with the emergence of a milder pathologic variety which is restricted to commissural fusion alone (with reduction in the combined type of stenosis) [15].
Clinical features
Most have moderate to severe exertional dyspnoea which may progress to paroxysmal nocturnal dyspnoea and acute pulmonary oedema (Table 1). Haemoptysis (sometimes frightening) may occur. Congestive heart failure is seen in one third to half of patients. There is failure to thrive. Thromboembolism is rare (1%). An interesting symptom is the presence of angina (13-16%). This is related to severe pulmonary hypertension which leads to subendocardial ischaemia of the right ventricle and low cardiac output. Approximately 9% are asymptomatic.
Table 1. Comparison of adult and juvenile MS series
| Author, year | Wood P, 1954 [1] | Roy et al, 1963 [4] | Shrivastava et al, 1991 [5] | Paul AT, 1967 [7] | Cherian et al, 1964 [6] |
| Country | England | India | India | Sri Lanka | India |
| Number | 300 | 108 | 125 | 100 | 126 |
| Age (years) | 37.6 (adults) | <20 | <12 | <16 | <20 |
| Male:Female ratio | 1:4 to 1:7 | 1.6:1 | 1.4:1 | 1:1 | 1.3:1 |
| Past history of RF | 68% | 66% | 51% | 55% | 53% |
| Multiple attacks of RF | 31.5% | 28% | NA | 30% | 12% |
Dyspnoea (mod-severe) | 80% | 78% | 73.6% | 67% | 75% |
| PND | 35% | 16% | 24% | 8% | NA |
| Haemoptysis | 42.5% | 27% | 16% | 10% | 18% |
| Chest pain/angina | 8.4% | 12% | 9% | 16% | 22% |
| Embolism | 13% | 2% | 1% | 1% | 1% |
| Rheumatic activity | NA | 22% | NA | 56% (biopsy) | 54% (biopsy) |
| Asymptomatic | NA | 9% | 1% | 5% | 0% |
| CHF | NA | 45% | 29% | 37% | 37% |
| Atrial fibrillation | 39% | 6% | 0% | 2% | 2% |
CHF: congestive heart failure; PND: paroxysmal nocturnal dyspnoea; NA: not available; RF: rheumatic fever
Physically, almost all cases have stunted growth [7]. They have a fragile look [4]. Severe pulmonary hypertension and arterial desaturation lead to a dusky complexion. Signs of CHF in the form of raised jugular venous pressure (JVP), enlarged liver and pedal oedema may be present. Sinus tachycardia is common whereas atrial fibrillation (AF) is rare (2-6%). Otherwise, the cardiac findings of juvenile MS are similar to their adult counterparts. The first heart sound is invariably loud with an early opening snap due to the low prevalence of leaflet calcification and immobility. A long mid-diastolic murmur with presystolic accentuation is heard at the apex. The majority (70-80%) have evidence of pulmonary hypertension in the form of parasternal heave and a loud P2. Rarely, a Graham Steell murmur of pulmonary regurgitation may be heard. A pansystolic murmur of functional tricuspid regurgitation is often heard in the tricuspid area.
Investigation
The electrocardiogram shows sinus rhythm in most patients [4-7]. AF is rare as the relatively small LA has not attained the critical mass needed for persistent AF to develop. The axis is rightward in more than 75% of cases. Left atrial enlargement (P mitrale) and right ventricular hypertrophy (dominant R in V1 or dominant S in V5) are common. A minority (11-13%) will show right atrial enlargement as well. Chest X-ray reveals mild-moderate cardiomegaly. The LA is universally enlarged. No MV calcification is seen. Moderate to severe pulmonary arterial hypertension is common and a majority will show hilar congestion.
Transthoracic echocardiography is the imaging modality of choice for confirming the diagnosis, quantifying severity and assessing suitability for PTMC (Figure 1). It helps to rule out other causes of MS (e.g., congenital MS). The echocardiographic features of juvenile MS are similar to those of adult MS (“hockey stick” appearance of anterior mitral leaflet, immobility of posterior leaflet and “fish-mouth” narrowing of the valve orifice) with a few notable exceptions. MV calcification is absent. Significant subvalvular disease is noted. Minute orifices (as small as 0.5 cm2) are common. The transmitral gradients are high owing to tight stenosis and tachycardia. The left atrium is not markedly enlarged. The right atrium (RA) and right ventricle (RV) are enlarged due to severe pulmonary arterial hypertension (PAH). The RV systolic pressure may be suprasystemic. As transthoracic images are optimal and AF is rare, transoesophageal echocardiography is usually not required.
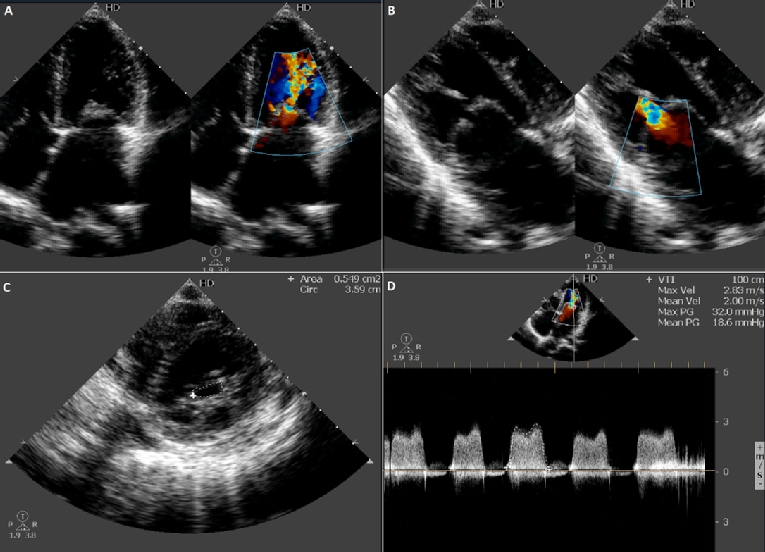
Figure 1. Two-dimensional echocardiography of a 14-year-old boy with severe mitral stenosis. A) Apical four-chamber view with dilatation of LA and RA and thickened AML and PML. B) Parasternal long-axis view shows significant subvalvular disease. C) Planimetry of mitral valve with area of 0.5 cm2. D) Continuous wave Doppler across MV with a mean gradient of 18 mmHg.
Routine cardiac catheterisation is not required except at the time of PTMC. The pulmonary artery pressure (PAP) and the pulmonary capillary wedge pressure (PCWP) are markedly elevated. The pulmonary vascular resistance is grossly abnormal in two thirds of cases [4]. The PAH is partly passive and partly vasoreactive. The cardiac output is normal in the majority of cases [4]. Interestingly, LV wall motion abnormalities in the form of global hypokinesia and segmental posterobasal hypokinesia have been reported as early as 12 years of age [5].
Management
The medical management of juvenile MS is similar to that of adults in the form of diuretics and rate-controlling drugs. Long-term penicillin prophylaxis is essential. The indications for intervention in MS are well established in adults. Intervention is indicated for symptomatic patients with an MVA <1.5 cm2 and for asymptomatic patients with an MVA of <1.0 cm2. These criteria, based on MVA, may be used for older children (>14 years) and adolescents. However, such thresholds (based on MVA) have not been validated in younger children (<12 years). It has been suggested that the MVA be indexed to body surface area (BSA). The American Heart Association [16] has recommended the following criteria for interventions in children with respiratory symptoms and failure to thrive: 1) peak transmitral gradient ≥20 mmHg; 2) mean transmitral gradient ≥15 mmHg; 3) near-systemic pulmonary artery pressure; and 4) calculated MVA <1 cm2/m2.
PTMC, using the Inoue technique (Inoue balloon [Toray, Tokyo, Japan], Accura balloon [Vascular Concepts Ltd., Bangalore, India]), is the procedure of choice when intervention is indicated. Correction of anaemia, infection, and heart failure stabilisation is advisable prior to PTMC. However, a critically ill child will need intervention on an urgent/emergent basis. PTMC can be performed even in the presence of rheumatic activity if needed. The contraindications to the procedure include bicommissural calcification, LA/LA appendage clot and significant mitral regurgitation (>grade 2/4), the first two being uncommon in juvenile MS. Technically, the procedure is the same as in adults, especially if the patient has attained a height of 140 cm (Figure 2). For younger and shorter patients, some modifications are needed. A paediatric transseptal needle set is preferable. It is preferable to begin balloon dilatation at a size that is 2-4 mm less than the calculated balloon size and progressively increase by 0.5-1 mm [17]. This approach has been shown to reduce the development of severe mitral regurgitation (MR) which is reported in up to 6% of cases [18]. Success rates have ranged from 93-100% [18,19], with almost nil mortality. In children less than 12 years, the success rates are a little lower (93%) [17], but still good. This probably results from the aggressive nature of the disease in this group leading to severe subvalular deformity. The rates of restenosis ranged from 16% at intermediate follow-up (34 months) [17] to 26% on long-term follow-up (8.5±4.8 years) [20]. Thus, these results are comparable to the adult population. Echo score rather than age was a predictor of restenosis [20]. The apprehension that restenosis rates may be higher in juvenile MS (due to smouldering rheumatic activity or the recurrence of rheumatic fever) is ill-founded. Repeat PTMC has been successfully performed in patients with restenosis and suitable anatomy with good success rates [17,19]. However, the need for mitral valve replacement (MVR) rises with restenosis due to a higher incidence of unfavourable valve anatomy.
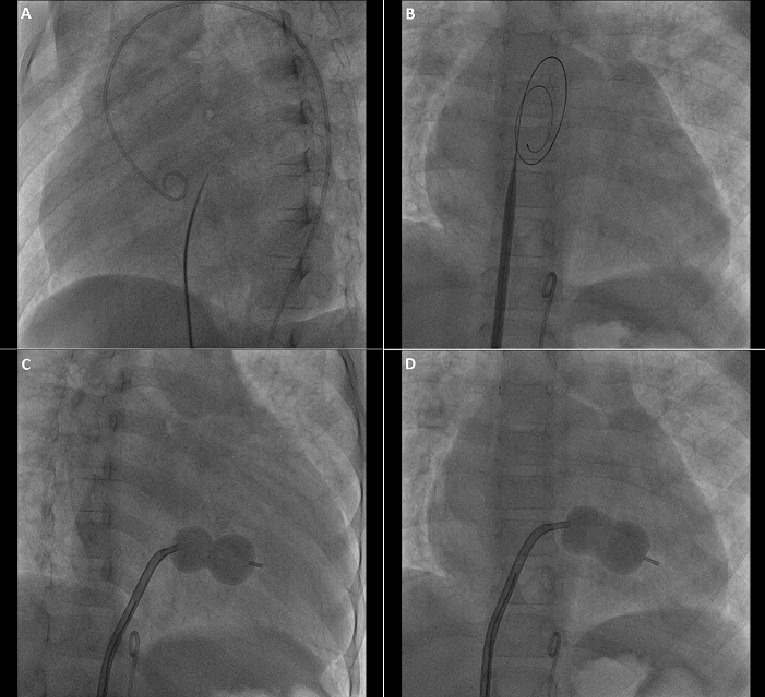
Figure 2. PTMC in a 12-year-old boy, height 124 cm, weight 22 kg. A) Transseptal puncture using a paediatric transseptal needle. B) Septal dilatation using a 14 Fr dilator. C) & D) 22-mm Accura balloon serially inflated to 22 mm, MVA increased from 0.5 cm2 to 1.2 cm2.
Closed mitral valvotomy (CMV) is indicated if PTMC is not available or the technical expertise for PTMC is lacking. The indications are the same as for PTMC. Mortality rates as low as 1.5% [14] have been reported in experienced centres. However, results of CMV are inferior to PTMC. Open mitral valvotomy (OMV) is indicated in the presence of LA/LAA thrombus or when tricuspid valve repair is needed. If the MV cannot be repaired due to severe deformity or there is pre-existing moderate to severe MR, MVR is indicated. However, MVR is associated with risks of anticoagulation, suboptimal preservation of ventricular function, and reduced survival. Survival at 10 years after MVR and MV repair was 79% and 90%, respectively [21]. Reoperation rates after MV repair are not different from those after MVR. Thus, MV repair is preferred over MVR in this age group.
Prevention and control
Long-term continuous penicillin prophylaxis has been shown to attenuate the prevalence, evolution and clinical spectrum of the sequelae of RF. In addition, with improvement in socioeconomic status, there has been a reduction in the pathologic severity of juvenile MS with a longer latent period and reduced incidence of pulmonary hypertension and CHF [15]. Finally, improvement in nutrition status, personal hygiene and better healthcare delivery for the treatment of streptococcal pharyngitis are the keys to controlling juvenile MS.
Lutembacher's syndrome
In 1916, Rene Lutembacher described the association of atrial septal defect (ASD) and mitral stenosis (MS) that goes by his name - Lutembacher's syndrome (LS) [22]. He believed both lesions to be congenital in origin. The definition has subsequently been broadened. The ASD may be congenital (ostium secundum or sinus venosus type) or acquired (e.g., following PTMC) [20]. The MS is often rheumatic in origin [23] but may be congenital [24]. The valvular pathology has been expanded to include combined mitral stenosis and regurgitation or pure mitral regurgitation [25] (the authors do not favour the inclusion of pure MR). Finally, MS with partial anomalous pulmonary venous connection and intact atrial septum leads to the same pathophysiology, i.e., decompression of the LA [26,27].
LS is rare, occurring in 4-7% of cases of ASD and 0.6-1.2% of cases of MS [23,28]. It has a female preponderance. Its importance lies in the unique haemodynamic consequence. Patients often present in the third decade [23], although this may vary from the first to the seventh decade [23,29]. The natural history of LS depends upon the severity of MS, the size of the ASD, the RV compliance and the pulmonary vascular resistance [23]. If the ASD is restrictive and MS is severe, the clinical course resembles that of MS alone. On the other hand, if the ASD is non-restrictive and MS is mild, the course is that of isolated ASD. The presence of a non-restrictive ASD and severe MS leads to a unique haemodynamic scenario. The ASD should be at least 1.0-1.5 cm [20,29]. The non-restrictive ASD allows the LA to decompress, thereby relieving the pulmonary venous hypertension. In turn, the left to right shunt across the ASD is augmented, leading to increased right ventricle (RV) volume overload. The consequence of this is that symptoms of MS such as exertional dyspnoea, orthopnoea and paroxysmal nocturnal dyspnoea are attenuated and delayed. This is, however, not without consequence. The increased left-to-right shunt leads to progressive enlargement of the RA and RV and earlier development of pulmonary arterial hypertension, causing congestive heart failure. The low cardiac output in severe MS is further reduced by the left-to-right shunt across the ASD [30]. Thus, LS patients tend to present with exercise intolerance, fatigue and palpitations. A history of RF was found in 46% of cases [23]. The JVP is raised with a prominent “A” wave which is a reflection of the elevated LA pressure. In the presence of MR or TR, the “V” wave is also prominent. Cardiac findings of ASD are accentuated whereas those of MS are attenuated. Thus, there is notable cardiomegaly with prominent pulmonary artery pulsations and systolic thrill at the upper sternal border [23,27]. The tricuspid closure sound is loud with a delayed diastolic murmur at the left sternal border [27] and an ejection systolic murmur at base, all indicative of the increased blood flow to the right heart. On the other hand, the apical mid-diastolic murmur of MS is softer with no pre-systolic accentuation and the opening snap is infrequent. If the ASD is restrictive, a continuous murmur may be present.
The ECG shows biatrial enlargement (P mitrale with qR/QR in V1), right-axis deviation and incomplete right bundle branch block. Chest X-ray reveals cardiomegaly (RA and RV enlargement), dilated pulmonary artery and pulmonary plethora. Pulmonary venous hypertension is less prominent. Transthoracic echocardiography remains the imaging modality of choice for the diagnosis of LS (Figure 3). MV calcification is uncommon. The transmitral gradients are low and underestimate the severity of MS. The Doppler pressure half-time method to assess MVA is inaccurate in this setting and overestimates the MVA (in proportion to the magnitude of left-to-right shunt) [31]. Valve areas should be calculated either by planimetry or by the continuity equation [31]. The size of the ASD is calculated in the usual manner.
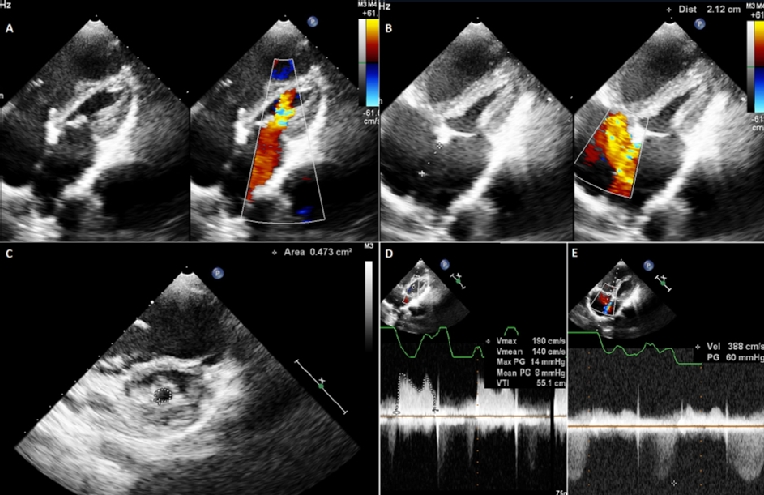
Figure 3. Two-dimensional echocardiography of a 35-year-old female with severe rheumatic MS (MVA 0.47 cm2, panels A & C) with a large ostium secundum ASD (21 mm, panel B). Despite tight MS, the mean gradient across the MV was only 8 mmHg (panel D). The TR jet gradient was 60 mmHg (panel E).
Traditionally, LS has been treated by open heart surgery (OMV/MVR with surgical repair of the ASD). If significant MS is unrecognised/uncorrected at the time of ASD closure, it will lead to catastrophic pulmonary oedema in the postoperative period. Percutaneous closure of ASD (with adequate rims) with PTMC has been reported, thereby allowing a definitive transcatheter repair [32]. It needs to be emphasised that PTMC in the presence of a large ASD is challenging due to the lack of anchoring support of the interatrial septum (IAS) which makes the catheter unstable [33]. LV entry is difficult, and the balloon repeatedly prolapses into the RA. In such a scenario, an over-the-wire technique is useful to track the PTMC balloon. A primary percutaneous approach avoids sternotomy, enabling a future first surgery to be performed at a lower risk in the event of mitral restenosis. However, it precludes a future repeat PTMC. On the other hand, MVR entails a prosthetic valve with its attendant complications and is best avoided in a young patient. OMV will need a repeat procedure in the future for restenosis. Redo surgery has a higher risk whereas redo PTMC (after OMV) is challenging due to the altered geometry of the IAS and higher Wilkins score of the MV. Thus, the choice between a percutaneous and open surgical repair needs to be carefully weighed. However, LS patients are often critically ill with deranged hepatic and renal parameters, making them high risk for open surgical repair. In such a scenario, PTMC is extremely useful in restoring the haemodynamics and rapidly improving the symptoms and metabolic parameters. The ASD may be closed electively at a later date.















