Introduction
Atrial fibrillation (AF) and peripheral arterial disease (PAD) are widely prevalent and incident among the general population [1,2]. Both conditions have similar epidemiologic patterns and common risk factors [1,2]. In addition, as is commonly known, AF as well as PAD are equally associated with increased morbidity and mortality [1,2]. In recent years, accumulating evidence suggests a strong relationship between AF and atherosclerotic vascular disease [3,4]. Indeed, it has been suggested that a common underlying pathway exists between AF and clinical manifestations of atherosclerotic disease [5], which explains the large epidemiological evidence.
PAD represents one of the main signs that underline the presence of systemic atherosclerosis. Moreover, PAD is considered to be one of the main risk factors increasing the long-term risk of developing stroke and thromboembolic events in AF patients [6]. Hence, it is currently included in the main clinical score used in the baseline risk assessment for newly diagnosed AF patients, the CHA2DS2-VASc score [6]. Beyond this, the relationship between AF and PAD seems to be more substantial, with a bi-directional relationship between the two conditions that ultimately amplifies exponentially the risk associated with both conditions. The aim of this summary is to provide a brief review of the most relevant evidence related to the relationship between AF and PAD.
A bi-causal relationship
To date, several studies have sought to investigate the relationship between AF and PAD (Table 1). In most cases, however, they were subgroup or post hoc analyses derived from various kinds of studies. The challenging issue for most of the studies reporting about this relationship is the assessment of PAD. Indeed, PAD can often be asymptomatic and, even if use of the ankle-brachial index (ABI) is the recommended diagnostic tool, PAD is often assessed only on the basis of symptomatic status (i.e., the presence of claudicatio intermittens) or recorded in the clinical history if collected during the clinical interview. Among the reported studies, only one was specifically designed to investigate the association between AF and PAD. The “Atrial Fibrillation Registry for Ankle-brachial Index Prevalence Assessment: Collaborative Italian Study” (ARAPACIS) was an Italian observational study aiming to report the prevalence of PAD as assessed by an ABI ≤0.90 in a cohort of patients diagnosed with non-valvular AF [7]. Even though in the other studies the prevalence of PAD in AF patients had great variability [8,9] (Table 1), the ARAPACIS study demonstrated that, when using a well-established diagnostic tool, PAD affects many AF patients, with a total prevalence up to 21% [7], significantly higher than that reported in the general population [2]. Moreover, 10% of patients were found to have an ABI ≥1.40 [7]. Similarly, the prevalence of AF in patients with PAD (11.5%) [10] largely exceeds that reported in the general population [1].
Table 1. Main studies reporting about the relationship between AF and PAD.
Study | Year | Location | Study type | Cohort characteristics | N | Condition prevalence | Results |
|---|---|---|---|---|---|---|---|
Frost [15] | 2000 | Denmark | Nationwide cohort | All patients discharged with an AF diagnosis and without any history of stroke | 27,202 | PAD: 4.2% | PAD increases independently risk of stroke both for men and women |
Goto [10] | 2008 | Multinational | Observational prospective | Outpatients with established atherosclerotic disease or multiple risk factors | 7,716* | AF: 11.5% | Concomitant AF and PAD increase the risk of major adverse events, in particular CV death |
Naccarelli [8] | 2009 | USA | Insurance database | Patients diagnosed with AF or atrial flutter | 222,605** | PAD: 12.2% | - |
Olesen [16] | 2012 | Denmark | Nationwide cohort | Patients diagnosed with non-valvular AF | 87,202 | PAD: 2.9% | PAD increases independently risk of stroke and TE |
Violi [7,17] | 2013/2016 | Italy | Observational prospective | Patients with non-valvular AF | 2,027 | PAD: 21% | Presence of PAD is independently associated with an increased risk for MI, CV events and CV death. |
O’Neal [11] | 2014 | USA | Observational prospective | Adult patients (45-84 years) without any history of CV disease with available ABI | 774* | - | PAD independently increases the risk of incident AF, particularly in elderly patients. Risk increases with progressively lower ABI |
Chang [13] | 2016 | Taiwan | Nationwide cohort | Patients with newly diagnosed AF and without any history of PAD, HF and stroke | 3,841 | - | Final adjusted Cox regression analysis found that AF independently increases the risk of PAD |
Griffin [12] | 2016 | USA | Prospective population cohort | Elderly subjects with risk factors for cardiovascular disease and without history of AF | 1,056* | - | Final adjusted Cox regression analysis found that PAD independently increases the risk of AF |
Lin [14] | 2016 | Taiwan | Nationwide Cohort | Adult patients (age ≥20 years) Without history of valvular disease | 555,113 | AF: 0.7% PAD: 4.3% AF & PAD: 0.08% | After multiple adjustments: PAD increases risk of AF; AF patients have higher rate of incident PAD, but the risk seems dependent on other risk factors; coexistence of AF and PAD is associated with higher risk for CV death and other adverse events |
Perera [9] | 2017 | Multinational | RCT | Subjects with AF and at least 1 additional risk factor | 7,554 | PAD: 3% | PAD independently increases the risk for all-cause death after full adjustment |
Proietti [18] | 2017 | Europe | Observational prospective | Patients with recent diagnosis of AF | 2,975 | PAD: 11% | PAD is associated with multiple comorbidities. Patients with concomitant PAD reported higher rates of CV and all-cause death, but the risk appears to be mediated by other comorbidities |
Schuyler Jones [19] | 2014 | Multinational | RCT | Patients with non-valvular AF | 14,264 | PAD: 5.9% | PAD patients reported higher rates of MI, CV and all-cause death. This difference in risk was attenuated after adjustments and no longer significant |
Hu [20] | 2017 | Multinational | RCT | Patients with non-valvular AF | 18,201 | PAD: 4.9% | PAD increased independently risk for CV and all-cause death. A non-significant trend for higher risk of stroke/TE was also found |
*PAD subset. **AF subset.
ABI: ankle-brachial index; AF: atrial fibrillation; CV: cardiovascular; HF: heart failure; MI: myocardial infarction; PAD: peripheral arterial disease; RCT: randomised controlled trial; TE: thromboembolic events
Furthermore, several studies have reported that PAD is independently associated with an increased risk of incident AF and, conversely, that the presence of AF is associated with a higher risk of reporting PAD, even though this risk seems partially mediated by other concomitant risk factors. An analysis derived from the “Multi-Ethnic Study of Atherosclerosis” (MESA) study found that patients with PAD, assessed by ABI measurement (defined as <1.00 and >1.4), have an increased risk for incident AF (hazard ratio [HR] 1.5, 95% confidence interval [CI]: 1.1-2.0) [11]. This relationship remains unchanged across various subgroup analyses (sex and race), while it seems more likely to be found in older patients (HR 1.7, 95% CI: 1.3-2.3 for patients ≥62 years old). Examining separately the ABI cut-offs, an ABI <1.0 was confirmed as significantly associated with a risk of incident AF (HR 1.5, 95% CI: 1.1-2.0), while with an ABI >1.4, despite showing a trend towards a higher risk, the association remains non-significant due to the very low number of patients (HR 1.8, 95% CI: 0.65-4.8) [11]. An additional analysis showed that, in patients with an ABI <1.0, progressively decreasing ABI was inversely associated with a higher risk of newly diagnosed AF (HR 1.1, 95% CI: 1.04-1.2 for each 0.1 decrease in ABI) [11].
Griffin and colleagues further verified this relationship in elderly patients [12]. Indeed, in an analysis from the Cardiovascular Health Study (CHS), which enrolled elderly patients (≥65 years old) affected with multiple cardiovascular risk factors, it was confirmed that the presence of PAD (defined as <1.00 and >1.4) was associated with an increased risk for incident AF (HR 1.25, 95% CI: 1.10-1.42) [12], with a similar inverse “dose-effect” relationship between lowering ABI and AF risk [12], as shown in the MESA study.
In a large population cohort study from Taiwan, Chang et al verified, conversely, the association between the presence of AF and the risk of incident AF [13]. Based on the ICD-9-CM codes, patients diagnosed with AF but with no history of PAD, heart failure or stroke were identified and compared with no AF patients. Over almost five years of follow-up observation, the presence of AF was found to be independently associated with incident PAD (HR 1.31, 95% CI: 1.19-1.45) [13]. This relationship was verified across gender and age subgroups [13]. Analysing the interaction between the presence of AF and other comorbidities/risk factors (hypertension, diabetes, hypercholesterolaemia, pulmonary disease, coronary artery disease, asthma), it was found that AF and other comorbidities had a similar increased risk for PAD when taken individually (HR 2.29 and HR 2.88, respectively), but when found together they increased exponentially the risk of PAD (HR 3.89, 95% CI: 2.82-5.36) [13].
Subsequently, another study derived from the same database further deepened the analysis about the relationship between AF and PAD [14]. All the patients available were divided into four groups: i) one control group without AF or PAD; ii) one group with AF only; iii) one group with PAD only; iv) one group with coexistent AF and PAD. Over 10 years of follow-up, Lin and colleagues found that the incidence rates for AF in PAD patients were higher than those in non-PAD patients, as well as the incidence rates for PAD in AF patients compared to non-AF ones [14]. Univariate Cox analysis found an increased risk both for incident AF in PAD patients and for incident PAD in AF patients. After full adjustments, increased risk for incident AF remained independently associated with PAD presence (HR 1.29, 95% CI: 1.17-1.42), while the risk for incident PAD became non-significant (HR 1.00, 95% CI: 0.89-1.11), seemingly mediated by other risk factors [14].
A dangerous combination
Moving from the epidemiological and pathophysiological link between AF and PAD to long-term follow-up observation, the relationship between these two conditions becomes stronger and more likely dangerous. Since 2000, Frost and colleagues, exploring the association between AF and incident stroke occurrence, found that, despite a not particularly high prevalence, PAD was independently associated with stroke occurrence, both in male and female subjects (HR 1.3, 95% CI: 1.0-1.7 and HR 1.3, 95% CI: 1.0-1.6, respectively) [15].
A subgroup analysis from the Reduction of Atherothrombosis for Continued Health (REACH) registry found that, when combined in the same subject, AF and PAD caused a significant increase in rates of cardiovascular (CV) death, as well as in rates of heart failure occurrence and the combined outcomes of CV death/myocardial infarction (MI)/stroke or hospitalisation for vascular events [10]. Moreover, in a large nationwide study derived from the Danish registers, PAD was significantly associated with an increased risk for stroke and thromboembolic events, even over a 12-year follow-up [16].
Follow-up analysis of the ARAPACIS study further extended the evidence about the association between AF and PAD in determining adverse outcomes. Indeed, an ABI ≤0.90 was found to be significantly associated with incident MI (HR 2.62, 95% CI: 1.32-5.20), vascular death (HR 2.24, 95% CI: 1.34-3.73) and the composite outcome of any vascular event (HR 1.50, 95% CI: 1.11-2.03) over a three-year follow-up period [17].
Expanding their analysis, Lin and colleagues also found that the coexistence of AF and PAD brought a relevant increase in risk for all the major adverse outcomes, carrying, after full adjustment, at least a twofold higher risk for CV death (HR 5.04, 95% CI: 3.00-8.47) than in patients with AF or PAD only [14].
An analysis from the EURObservational Research Programme in the AF pilot registry, an observational registry about AF patients held by the European Society of Cardiology, reported that AF patients with concomitant PAD had higher rates of CV and all-cause death, even though the Cox regression analysis found a non-significant trend in the association between PAD and the occurrence of all-cause death (HR 1.375, 95% CI: 0.931-2.030; p=0.1096), after adjustment for concomitant risk factors and comorbidities [18]. A fully adjusted analysis, comprising CV prevention drugs, further mitigated the association between PAD and all-cause death, conversely underlining a relevant role of statins in reducing mortality in PAD patients, with no difference in risk for the use of antiplatelet agents (mostly aspirin) [18].
In recent years, as is commonly known, non-vitamin K antagonist oral anticoagulants (NOACs) entered daily clinical practice in treating AF patients [1]. Despite this, there are few data about the use of NOACs in patients with AF and PAD, except for some subgroup analyses derived from the NOACs phase III trials, both reporting quite a low prevalence rate [19,20]. In the “Rivaroxaban Once daily, oral, direct factor Xa inhibition compared with vitamin K antagonism for prevention of stroke and Embolism Trial in Atrial Fibrillation” (ROCKET-AF) study, despite a higher rate of major adverse events, PAD was not associated with any difference in risk after adjustment [19]. Comparing the use of rivaroxaban and warfarin interacting with the presence of PAD, the use of rivaroxaban was associated with an increased risk of bleeding outcomes (HR 1.40, 95% CI: 1.06-1.86 for major and clinically relevant non-major bleeding for rivaroxaban vs. warfarin in PAD patients) [19].
In the post hoc analysis from the “Apixaban for Reduction in Stroke and Other Thromboembolic Events in Atrial Fibrillation” (ARISTOTLE) trial, the presence of PAD was associated with an increased risk of CV and all-cause death (HR 1.44 and HR 1.36, respectively), with a non-significant trend for higher risk in stroke and thromboembolic events (HR 1.32, 95% CI: 0.93-1.88; p=0.1227), probably due to the low absolute number of events in the PAD group [20]. The use of apixaban compared to warfarin in PAD patients was found to be associated with no difference in terms of bleeding outcome occurrences, which were instead strongly reduced in non-PAD patients [20].
Conclusions
Epidemiological data have shown a strong relationship between AF and PAD, underlining how often the two conditions are concomitant. Several studies have documented a clear effect in determining the occurrence of AF in PAD patients, while accumulating evidence suggests a similar effect the other way around, with an increased risk of PAD in AF patients, even though this relationship still deserves further insights, since it appears to be partially mediated by other concomitant risk factors and comorbidities.
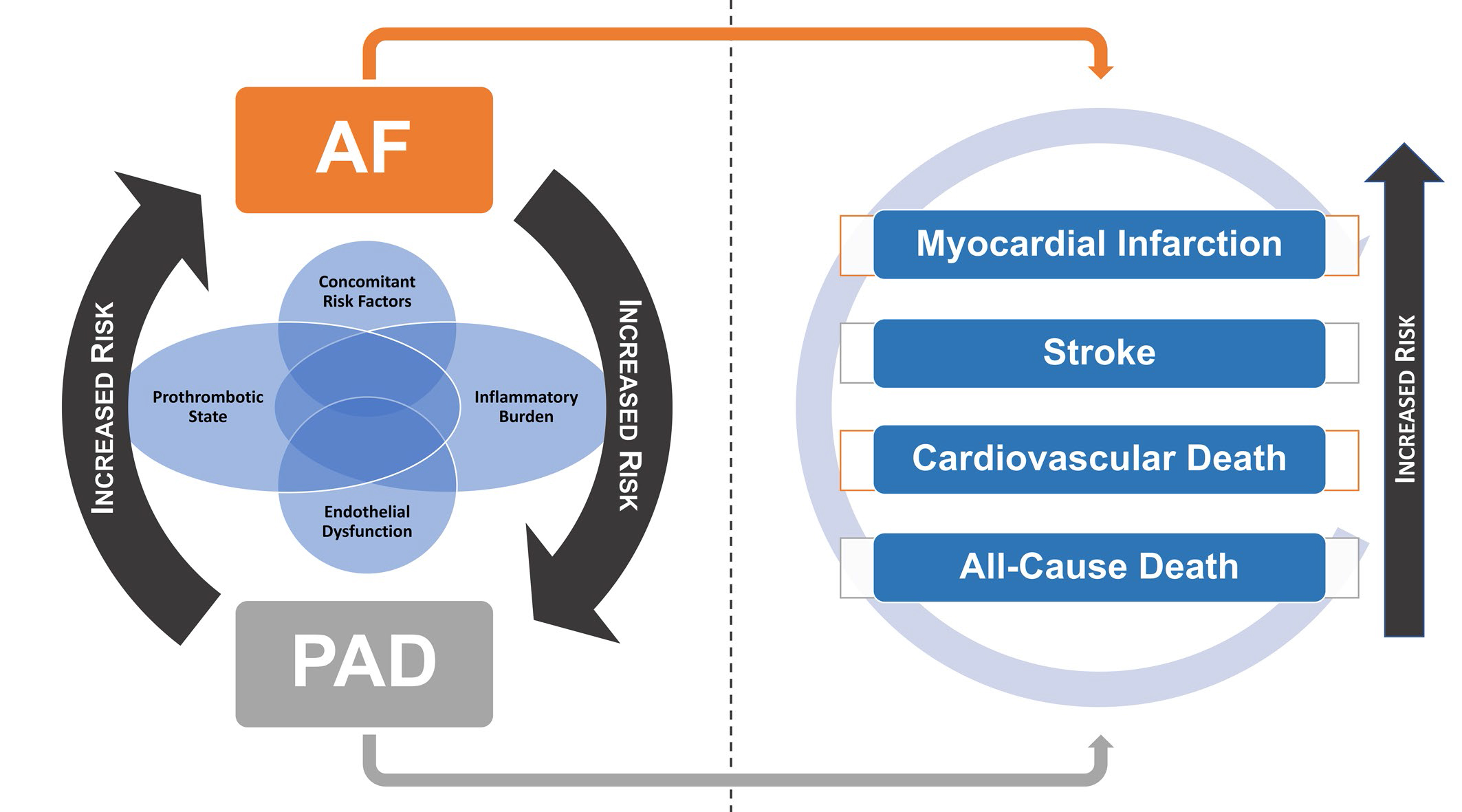
Despite the fact that there are currently no further insights in terms of the mechanistic processes, it seems plausible that the association between AF and PAD, on the background of multiple common risk factors and comorbidities, lies in the mutual ground related to increased levels of inflammation, endothelial dysfunction and a prothrombotic state, which leads to a susceptibility for each condition to increase the risk for the other one [11]. Further data are still needed to elucidate fully the complex mechanisms linking AF and PAD to each other (Figure 1, left panel).
Furthermore, all the data presented clearly underline that the association of AF and PAD elicits a relevant increase in all the major outcomes associated with both conditions independently (stroke, MI, CV death, all-cause death), making their concomitant presence a much feared complication for both AF and PAD patients (Figure 1, right panel).
Figure 1. The complex relationship between AF and PAD and their association in determining major adverse outcomes.
To date, European guidelines about AF and PAD are concordant in indicating the use of oral anticoagulant drugs rather than antiplatelet drugs when the two conditions occur in the same patient [1,2]. Even though current AF guidelines recommend the use of NOACs over warfarin and other vitamin K antagonists, there are no specific data investigating the use of NOACs in PAD patients, while data available from the NOACs trials seem to suggest a possible increase in the risk of bleeding outcomes with no clear advantage in terms of reduction of stroke and other major efficacy outcomes. A clear need for more research is warranted to clarify better this highly relevant issue in the clinical management of these patients.















