Keywords: atrial fibrillation, cardio-oncology, cardiotoxicity, chemotherapy, hypertension, ischaemic heart disease, radiotherapy
Introduction
As the improvement in cancer patients’ survival has increased the clinical relevance of preventing and monitoring cardiovascular (CV) complications derived from oncological therapies and cardio-oncology has been proposed as a new approach to improve patients ́ prognosis [1]. Nowadays, much of the focus has been on the early detection and prevention of heart failure (HF); however, some of the old and new cancer therapies are associated with the development of a broad range of CV toxicities [2]. The present article summarises multidisciplinary expert-based recommendations on the diagnosis, monitoring and treatment of the more prevalent non-HF toxicities including arrhythmias, hypertension and ischaemic heart disease.
Cancer and cardiac arrhythmias
Both the development of new anti-cancer drugs and the improvements in cancer prognosis have dramatically increased the prevalence of cardiac arrhythmias (CA) in cancer patients [3, 4]. However, their incidence is probably underestimated in the literature because patients with pre-existing cardiac diseases have frequently been excluded from oncological trials. A detailed description of the drugs related to CA and their incidence is outside the objectives of this document. For more detail, the reader is referred to recently published reviews [3-7].
The mechanisms by which cancer treatments cause CA are still poorly understood. The individual risk of clinically relevant CA depends on the risk of the specific therapeutic scheme, the presence of comorbidities and the inflammation and metabolic changes induced by cancer itself. In addition, arrhythmic risk is higher in cancer patients according to age (2/3 of patients are over 65) [3,4] and the presence of pre-existing CV risk factors or CV diseases (there is a 30% prevalence of pre-existing CV diseases in cancer patients) [8]. Management of cancer-induced CA is challenging and requires a close collaboration between cardiologists and oncologists. This is particularly relevant for atrial fibrillation (AF) and QT prolongation, the two main arrhythmic issues in these patients. Table 1 summarises the drugs associated with a greater incidence of AF and QT prolongation.
Table 1. Cancer drugs associated with the development of atrial fibrillation, QT prolongation and hypertension and general rules to guide management.
| Cancer therapy induced | Cancer drug agents |
General rules to guide management: early cardio-oncology consultation |
|---|---|---|
| Atrial fibrillation |
Alkylating agents Cisplatin, ifosfamide, cyclophosphamide, melphalan Anthracyclines Antimetabolites Capecitabine, 5-FU, gemcitabine IL-2 Interferons Rituximab Small molecule TKIs Ponatinib, sorafenib, sunitinib, ibrutinib Topoisomerase II inhibitors Amsacrine, etoposide Taxanes |
|
| QTc prolongation |
Anthracyclines Histone deacetylase inhibitors Depsipeptide, belinostat, vorinostat Chemicals: arsenic trioxide Tyrosine kinase inhibitors Axitinib, bosutinib, cabozantinib, crizotinib, dabrafenib, dasatinib, lapatinib, nilotinib, pazopanib, ponatinib, sorafenib, sunitinib, vandetanib, vemurafenib BRAF inhibitor: vemurafenib CDK4/6 inhibitors: ribociclib |
|
| Hypertension |
Monoclonal antibody-based TKI Bevacizumab, ado-trastuzumab emtansine Monoclonal antibodies Alemtuzumab, ibritumomab, ofatumumab, rituximab mTor inhibitors: everolimus, temsirolimus Tyrosine kinase inhibitors Pazopanib, ponatinib, sorafenib, sunitinib, axitinib, abozantinib, ibrutinib, nilotinib, ramucirumab, regorafenib, trametinib, vandetanib, ziv-aflibercept Proteasome inhibitors Bortezomib, carfilzomib Antimetabolites Decitabine |
|
| Coronary artery disease |
Fluoropyrimidines 5-fluorouracil, capecitabine Platinum compounds cisplatin Taxanes Paclitaxel, docetaxel VEGFi Bevacizumab, sorafenib, sunitinib Etoposide Bleomycin |
|
The incidence of AF is substantially higher among cancer patients than in the general population [3,5,6]; however, a prior history of AF, or the potential for developing AF, should not prevent physicians from considering any anti-cancer therapy if indicated. As in other high-risk populations, early identification and management of comorbidities and educational programmes are essential to reduce the incidence of AF [9]. Postoperative AF is a frequent well-recognised complication of cancer surgery with an overall incidence of 20%. In high-risk patients, prophylactic treatment with beta-blockers or losartan may help to minimise postoperative AF, particularly after lung cancer surgery [3,5].
As a general rule, in patients with pre-existing AF who need cancer therapy, a cardio-oncology consultation is recommended to optimise AF treatment [9]. The diagnosis of a new AF should also trigger a priority cardio-oncology consultation to decide the best treatment strategy and to avoid unnecessary interruptions of potentially lifesaving oncologic drugs. Heart rate control is the preferred option, given that rhythm control has a limited success during cancer treatment. In the absence of specific risk prediction scores, antithrombotic therapy should be guided by the CHA2DS2-VASc and the HAS-BLED scores [9,10]. Traditionally, the use of vitamin K antagonists (VKAs) is not recommended during active cancer treatment due to the difficulty of ensuring stable international normalised ratio (INR) levels. In valvular AF, the decision to keep VKAs or switch to low molecular weight heparins should be individualised. In this scenario, VKAs remain a reasonable option in patients with stable INRs and a low bleeding risk. Preliminary evidence with direct oral anticoagulants (DOACs) in non-valvular AF is promising in active cancer; however, we need more information to confirm DOACs as a safe alternative [10]. DOAC selection should be individualised based on the potential for drug-drug interactions and comorbidities [10].
A rigorous evaluation of the QT interval is mandatory in patients who receive potential QT-prolonging drugs to minimise the risk of arrhythmias and to prevent unnecessary interruptions of cancer therapies (Table 1) [3,4,6,7]. Initial assessment should include a baseline electrocardiogram (ECG) and regular monitoring of the QT interval during therapy using the Fridericia correction formula. Withdrawal of anti-cancer drugs or administration under hospital monitoring should be considered if the corrected QT interval (QTc) is >500 ms or has increased by more than 60 ms from baseline [3,4,6,7].
Cancer and hypertension
Hypertension (HTN) is the most common comorbidity reported in oncological patients [2-4]. More than one third of cancer patients develop HTN during therapy and this percentage is higher in an aged population [2-4,8]. Oncological therapies may cause HTN through different mechanisms, although the most frequent are vascular endothelial growth factor signalling pathway inhibitors (VEGFi) and proteasome inhibitors (Table 1). It has been reported that VEGFi induce new HTN or destabilise previously controlled HTN in 11 to 45% of patients during the first months of therapy, by inhibiting the production of nitric oxide in the arterial wall [2-4]. HTN was also reported in around 10% of patients treated with proteasome inhibitors. These drugs reduce the vasodilator response to acetylcholine, favouring vasoconstriction and vasospasm as the main mechanism to induce HTN [2-4]. Although HTN is considered an undesirable side effect of cancer therapy, the increase in blood pressure (BP) levels has been shown to predict the efficacy of some cancer therapies, whereas optimal BP control improves treatment tolerance without reducing efficacy [3,4]. BP should be properly managed and monitored before and during cancer treatment, following the pharmacological and dietary recommendations established for the general population [3,4,11]. Early diagnosis and treatment is essential because HTN is a major risk factor for the development of HF and CA, and suboptimal BP control may lead to premature discontinuation of chemotherapy (Table 1) [3,4,11].
As a general rule, the BP target in cancer patients with uncomplicated HTN is <130/80 mmHg [11]. With the exception of low-risk patients or frail older patients, the initiation of treatment with two drugs improves the speed and efficiency of BP control. As in the general population, renin-angiotensin system blockers (RAS) with dihydropyridine calcium channel blockers is the preferred combination [3,4,11]. In patients at risk of HF or AF, RAS and beta-blockers are considered the drugs of choice given their protective profile. If BP is not controlled by the above treatments, the use of a three-drug therapy or the addition of spironolactone is recommended unless contraindicated. Another consideration in choosing antihypertensive agents is to minimise harmful drug-drug interactions [3,4,11].
Cancer and ischaemic heart disease
Several cancer therapies can lead to cardiac ischaemia or, in the worst cases, result in myocardial infarction. The mechanisms by which drugs can cause vascular toxicity range from a direct vasospastic effect to endothelial injury or premature atherosclerosis. The most notorious cancer therapeutics resulting in myocardial ischaemia are fluoropyrimidines including 5-fluorouracil (5-FU) and its oral precursor capecitabine. Both agents can lead to coronary artery spasms and endothelial dysfunction. In a prospective study including 367 patients, the use of high-dose 5-FU resulted in cardiac events in 28 patients (7.6%). The majority of the events consisted of unstable angina and/or hypotension. Sudden cardiac death was observed in four patients [12]. In a more recent study including 358 patients receiving 5-FU, 21 of them (5.9%) had ischaemia at rest. In the patients with no ischaemia at rest, 16 (6.9%) subsequently had exercise-induced ischaemia. In 14 of them, a second exercise test after stopping 5-FU showed no ischaemia anymore. The majority of patients with a positive exercise test had no symptoms, so-called silent ischaemia [13]. In some patients, 5-FU, but also capecitabine, can lead to fatal myocardial infarction [3].
Another class of drugs associated with vascular toxicity is cisplatin. Arterial thrombosis may develop in 2-8% of patients treated with cisplatin [3]. Furthermore, VEGFi, such as bevacizumab and sunitinib, have also been associated with arterial thrombosis ranging from 1-4%, depending on the underlying disease, with the highest percentage in patients with metastatic disease [2,3].
There are no specific recommendations for ischaemic heart disease prophylaxis in cancer patients. As in those without cancer, preventive strategies should be based on a strict CV risk factor control and clinical surveillance. When required, testing for the detection of ischaemia (electrocardiogram [ECG] or echo stress test) and coronary artery disease (cardiac CT) are recommended. For patients with angina, treatment optimisation is recommended, as well as control of precipitating factors such as anaemia. Nitroglycerin or calcium antagonist administration can prevent recurrence of vasospastic angina if there are no oncologic therapeutic alternatives [14]. In patients with persistent symptoms, an eventual revascularisation may facilitate therapy tolerance. Revascularisation strategies should be individualised as well as the need for antithrombotic therapy due to the high risk of bleeding and thrombocytopaenia in this population [3] (Table 1).
Radiotherapy CV side effects
Radiotherapy plays an important role as an adjuvant therapy in the majority of malignancies and improves disease-free and overall survival. However, the benefits of radiotherapy may be offset by side effects, which may only become apparent many years after the initial treatment. Radiation exposure of the thoracic region may lead to pericardial disease, macrovascular and microvascular coronary artery disease (CAD), valvular heart disease, conduction diseases, autonomic dysfunction and myocardial fibrosis [3,15]. Additionally, many cancer patients receive multimodal treatment in which radiotherapy poses an additional risk of CV complications [16].
Acute pericarditis has become unusual after radiotherapy due to the lower doses used and new techniques to keep the heart out of the radiation field. Treatment of acute pericarditis does not differ from other aetiologies and includes the use of colchicine and NSAIDs as first-line therapy [3]. Chronic pericarditis occurs mostly in patients after high-dose radiotherapy and may lead to pericardial effusion and, ultimately, to a constrictive pericarditis. If pericardial thickening is severe enough to cause a symptomatic chronic constrictive pericarditis, early pericardiectomy is indicated, even if the cardiac surgery risk is higher than in other aetiologies [3,15].
Radiotherapy is an independent risk factor for microvascular endothelial inflammation implicated in the pathogenesis of accelerated atherosclerosis and heart failure, usually associated with preserved ejection fraction [15]. The risk of radiation-induced CAD is directly related to radiation doses, increases over time and is magnified by the presence of CV risk factors. Usually the distribution of CAD reflects the radiation field: typically, lesions are clustered in a proximal distribution, increasing the risk of larger myocardial infarctions even in young patients without traditional cardiac risk factors (Figure 1) [15]. A large Scandinavian case control study in over 2,000 patients with breast cancer showed a linear relationship between mean heart dose and the risk of an ischaemic event. The risk of a major coronary event increased by 7.4% per gray mean dose to the heart and no dose threshold could be observed [17]. The increase started within the first five years after radiotherapy and continued to increase over decades. For example, for a 70-year-old woman without CV risk factors receiving a mean heart dose of 10 gray, her cardiovascular risk increases to a similar extent as having an established CV risk factor without having received radiotherapy. In a recent study; it has been shown that especially the dose received by the LV was a better predictor of acute coronary events compared to the mean heart dose [18]. The increased risk for CAD highlights the importance of assessing CV risk factors in patients treated with radiotherapy; however, it is unclear whether a screening programme (e.g., coronary CT angiography in patients exposed to radiotherapy) may be beneficial. Since treatment with radiotherapy is an important risk factor for CAD, it seems prudent that CV risk factors, including hypercholesterolaemia and hypertension, should be treated aggressively along with lifestyle interventions [2,3,16].
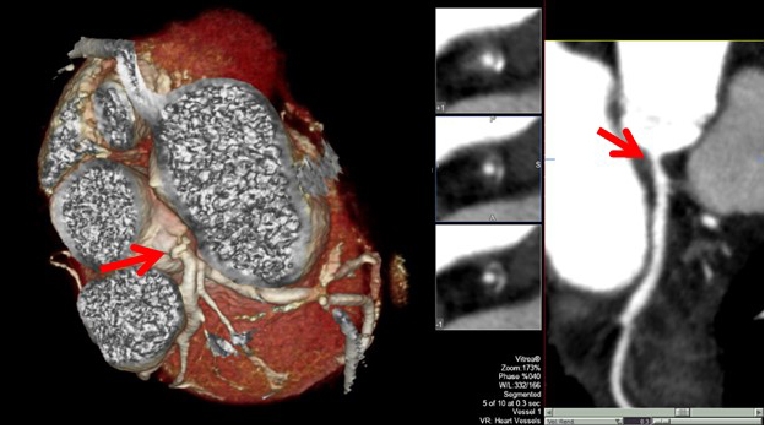
Figure 1. Clinical case. A 42-year-old female presented to the cardio-oncology clinic complaining of mild chest pain and transient dizziness during sports practice. The patient had a history of Hodgkin’s disease treated with anthracycline-based chemotherapy and mediastinal radiotherapy at 16 years old. She had no other CV risk factors. Baseline electrocardiogram revealed sinus rhythm with no associated repolarisation abnormalities. Echocardiographic examination showed an LVEF of 57%. Non-invasive coronary imaging using computed tomography angiography revealed a non-calcified significant lesion at the left main coronary artery. Subsequent cardiac catheterisation revealed severe disease in the left main coronary artery. A drug-eluting stent was placed without associated complications. The patient was discharged under aspirin, prasugrel and atorvastatin. She remains asymptomatic at follow-up.
Mediastinal radiation may also cause both accelerated atherosclerotic calcification and dystrophic calcification of the aorta and great vessels, leading to an excess risk of stroke in long-term survivors across all ages [15,17].
Significant valvular heart disease may occur after radiotherapy, especially with doses exceeding 30 gray. In a retrospective study, 415 patients with Hodgkin’s lymphoma were matched with a general population. It appeared that treated patients were eight times more likely to receive valvular surgery compared to the matched general population [19].
Another chronic problem regarding radiation therapy is cardiac arrhythmias. ECG abnormalities are frequent during and after radiotherapy as well as AF generally related to pericardial inflammation and anti-cancer drugs. Cardiac autonomic dysfunction occurs after mediastinal radiation more frequently in young patients. These patients have an increased prevalence of an elevated resting heart rate, an abnormal heart rate recovery and blunted blood pressure response during all stages of exercise/recovery compared with control subjects. These conditions limit exercise capacity and are associated with increased all-cause mortality and a higher incidence of AF [20].
Conclusions
Cardiovascular toxicity is a reality that impacts on the quality of life and overall survival of cancer patients. Careful analysis of the needs of these patients is mandatory in order to develop preventive strategies focused on the early detection and treatment of CV toxicities. We need cardio-oncology multidisciplinary teams to integrate skills and abilities and to standardise the care process.