Background
Peripheral arterial disease (PAD) is usually caused by atherosclerosis of the major vessels supplying the lower extremities. Approximately 10% of the world’s population have PAD [1]. The majority of people with PAD are asymptomatic. Some people with PAD, however, have limited walking ability and therefore reduced quality of life. In cases where medical management is not suitable or effective or in severe cases of PAD, endovascular and surgical interventions are indicated.
Different endovascular techniques are available for the management of peripheral stenosis and/or occlusions. Standard interventional treatment options, besides plain old balloon angioplasty (POBA), are drug-coated balloons (DCB), bare metal stents (BMS), and drug-eluting stents (DES), as well as numerous atherectomy devices. In addition, stent implantation increases the patency rate compared to POBA and probably also to DCB; thus, it is often used, particularly in long and/or calcified lesions [2].
Lower extremity artery disease (LEAD)
Most patients with LEAD are asymptomatic. Walking capacity must be assessed to detect clinically masked LEAD. Even asymptomatic patients with LEAD are at high risk of cardiovascular (CV) events and will benefit from most CV preventive strategies, especially strict control of risk factors. Antithrombotic therapies are indicated in patients with symptomatic LEAD. There is no proven benefit for their use in asymptomatic patients. Data from anatomical imaging tests should always be analysed in conjunction with symptoms and haemodynamic tests prior to treatment decision. Early recognition of tissue loss and/or infection is important, and referral to a vascular specialist is mandatory for limb salvage using a multidisciplinary approach. Acute limb ischaemia with neurological deficit mandates urgent revascularisation [3].
Management strategy for intermittent claudication
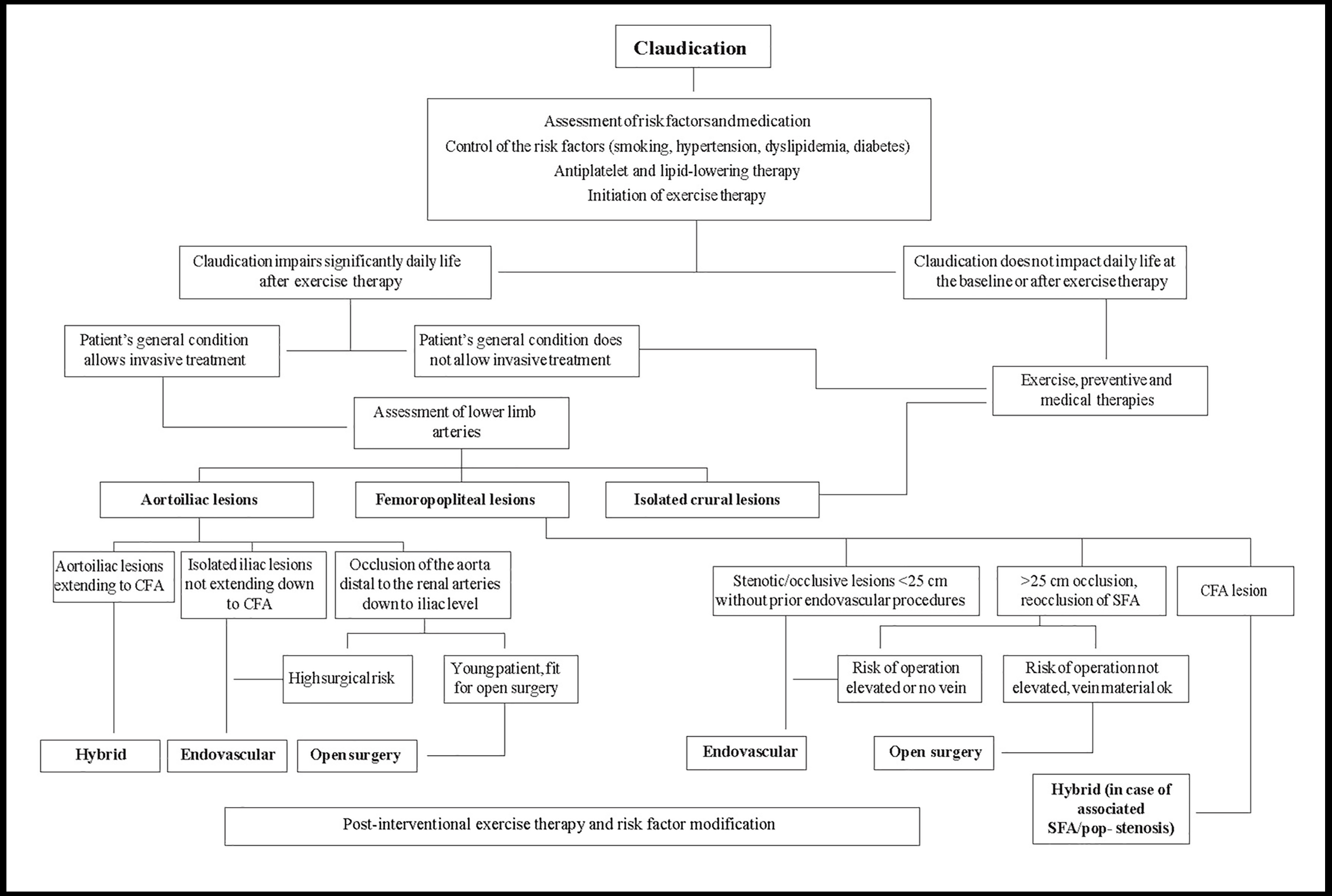
Several studies have demonstrated the efficacy of endovascular therapy and open surgery for symptom relief and quality of life (QOL) in claudicants. However, these interventions have limited durability and may be associated with mortality and morbidity. Thus, they should be restricted to patients who do not respond favourably to medical treatment or when disabling symptoms substantially alter activities of daily life (Figure 1) [3].
Figure 1. Management of patients with intermittent claudication (related to atherosclerotic lower extremity artery disease).
With permission from [3].
EVT: endovascular therapy; GSV: great saphenous vein
A systematic review of 12 trials (1,548 patients) comparing medical therapy, endovascular therapy and open surgery in claudicants showed that, compared with the former, each of the other alternatives was associated with improved claudication symptoms and QOL [4].
Compared with endovascular therapy, open surgery may be associated with longer hospital stays and higher complication rates but results in more durable patency [4].
Revascularisation for intermittent claudication
In patients with intermittent claudication, cardiovascular prevention and exercise training are the cornerstones of management. If activities of daily life are severely compromised, revascularisation can be proposed, along with exercise therapy. The anatomical location and extension of arterial lesions has an impact on revascularisation options (Figure 1) [3].
Aorto-iliac lesions
Isolated aorto-iliac lesions are a common cause of claudication. In the case of short stenosis/occlusion (<5 cm) of iliac arteries, endovascular therapy gives good long-term patency (>90% over 5 years) with a low risk of complications [5].
In cases of iliofemoral lesions, a hybrid procedure is indicated, usually endarterectomy or bypass at the femoral level combined with endovascular therapy of the iliac arteries, even with long occlusions. If the occlusion extends to the infrarenal aorta, covered endovascular reconstruction of an aortic bifurcation can be considered [3]. One- and two-year primary patency is 87% and 82%, respectively [6].
If the occlusion comprises the aorta up to the renal arteries and iliac arteries, aortobifemoral bypass surgery is indicated in fit patients with severe life-limiting claudication. In these extensive lesions, endovascular therapy may be an option, but it is not free of perioperative risk and long-term occlusion [3].
Femoropopliteal lesions
Femoropopliteal lesions are common in claudicants. If the circulation to the profunda femoris artery is normal, there is a good possibility that the claudication will be relieved with supervised exercise training; intervention is mostly unnecessary. If revascularisation is needed, endovascular therapy is the first choice in stenosis/occlusions <25 cm. If the occlusion/stenosis is >25 cm, endovascular recanalisation is still possible, but better long-term patency is achieved with surgical bypass, especially when using the great saphenous vein (GSV). No head-to-head trials comparing endovascular therapy and surgery are yet available [3].
The challenge of endovascular therapy is the long-term patency and durability of stents in the femoropopliteal region, where the artery is very mobile.
Several new endovascular solutions, such as atherectomy devices, drug-eluting balloons and new stent designs have been shown to improve long-term patency [3].
Infrapopliteal disease
Extended infrapopliteal artery disease is mainly seen in diabetic patients, often associated with superficial femoral artery (SFA) lesions (inflow disease). In stenotic lesions and short occlusions, endovascular therapy can be the first choice. In long occlusions of crural arteries, bypass with an autologous vein gives superior long-term patency and leg survival. If the patient has increased risk for surgery or does not have an autologous vein, endovascular therapy can be attempted [3].
Chronic limb-threatening ischaemia (CLTI)
Chronic limb-threatening ischaemia specifies clinical patterns with a vulnerable limb viability related to several factors. The risk is stratified according to the severity of ischaemia, wounds and infection. The treatment here is urgent to avoid limb loss, though some patients can keep their legs for long periods of time even in the absence of revascularisation [7].
Management of patients with chronic limb-threatening ischaemia
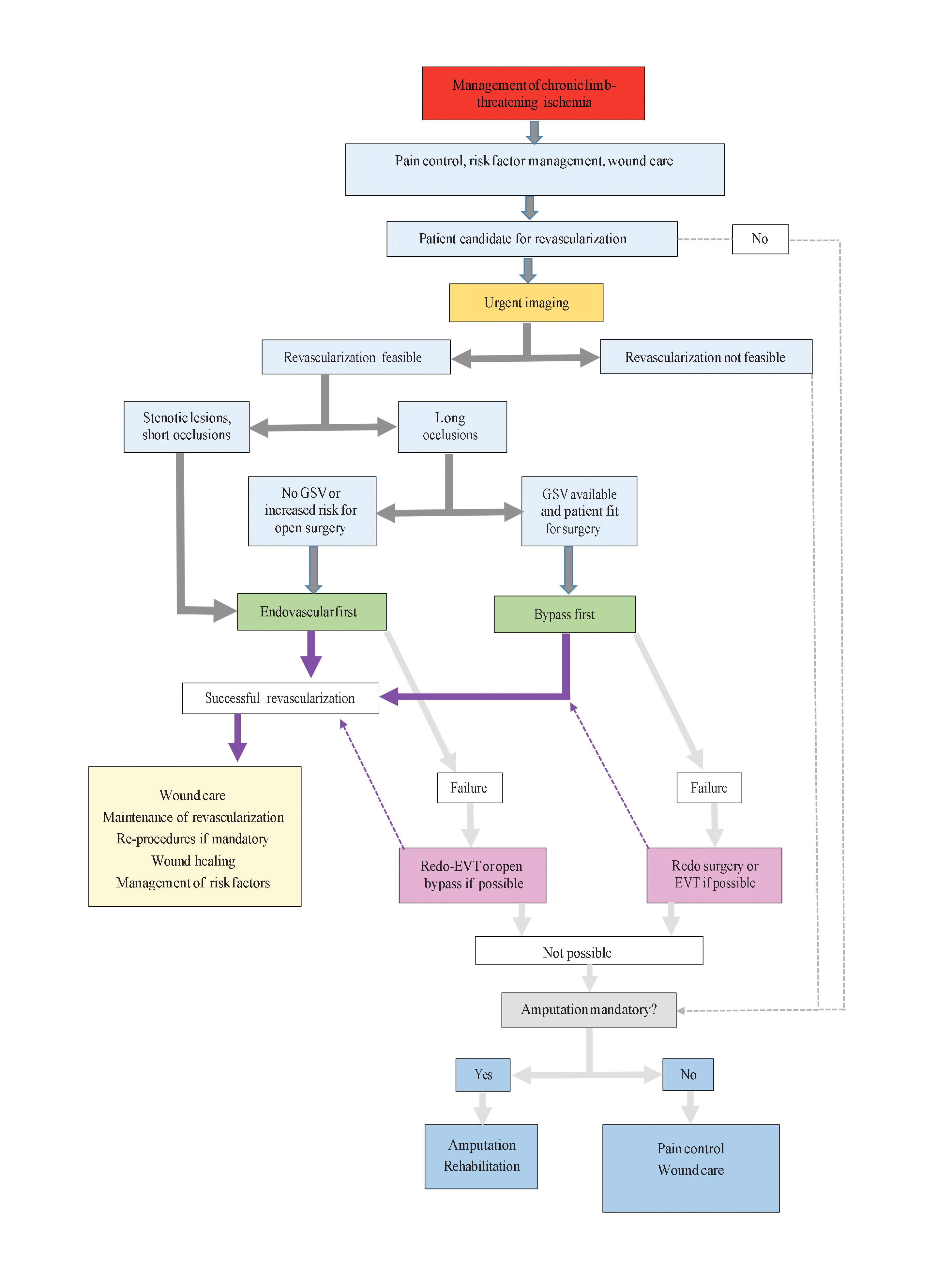
All patients with CLTI must have best medical treatment (BMT) with correction of risk factors. In those with diabetes, glycaemic control is particularly important for improved limb-related outcomes, including lower rates of major amputation and increased patency after infrapopliteal revascularisation [8]. (Figure 2)
Figure 2. Management of patients with chronic limb-threatening ischaemia.
With permission from [3].
CFA: common femoral artery; SFA: superficial femoral artery
Revascularisation
Revascularisation should be attempted as much as possible [3]. So far, only one randomised trial, the BASIL trial, has directly compared endovascular therapy to open surgery in CLTI patients [9]. At two years, there was no significant difference between endovascular therapy and surgery regarding amputation-free survival. In survivors after two years, bypass surgery was associated with improved survival (on average seven months, p=0.02) and amputation-free survival (six months, p=0.06) [10]. These data are challenged by more recent endovascular therapy techniques.
So far, drug-eluting balloons in below-the-knee disease have shown no superiority over POBA [11].
Well-designed prospective studies and randomised controlled trials in CLTI patients are sparse. This may be related to the fact that studies in this specific population are not easy to conduct, due to the lower incidence of CLTI compared to milder forms of PAD, and issues with respect to follow-up of CLTI patients.
Meanwhile, in each anatomical region, both revascularisation options should be individually discussed.
Aorto-iliac disease
CLTI is almost never related to isolated aorto-iliac disease, and downstream lesions are often concomitant. A complete digital subtraction angiography (DSA) down to the plantar arches is required for proper arterial network assessment and procedure planning. Hybrid procedures (e.g., aorto-iliac stenting and distal bypass) should be encouraged in a one-step modality when necessary [12].
Femoropopliteal disease
CLTI is unlikely to be related to isolated superficial femoral artery (SFA) lesions; usually femoropopliteal involvement combined with aorto-iliac or below-the-knee disease is found. In up to 40% of cases, inflow treatment is needed [11]. The revascularisation strategy should be designed according to lesion complexity. If endovascular therapy is chosen first, landing zones for potential bypass grafts should be preserved. When bypass surgery is decided, the bypass should be as short as possible, using the saphenous vein [3].
Endovascular modalities
While a number of endovascular modalities are available, a clear consensus on the superiority of a particular type of treatment has not yet emerged. We review the current data on established endovascular therapies for PAD.
Balloon angioplasty
The introduction of long balloons allows widespread use of percutaneous transluminal angioplasty (PTA) including for long lesions and patients with diffuse disease, especially given the often multilevel and multivessel nature of PAD. Initial success rates are in the 90% range for PTA of the SFA and infrapopliteal arteries [13]. However, long-term vessel patency is a concern, with restenosis occurring in more than 60% of cases at 12-month follow-up [13].
Drug-eluting balloons
Several studies have established the benefit of drug-eluting balloons (DEB) compared to PTA in primary patency for femoropopliteal disease. Lower rates of target lesion revascularisation (TLR) have also been reported with paclitaxel-coated balloon (PCB) use [14].
The LEVANT II trial, a highly powered 54-site randomised controlled trial of 476 patients with symptomatic intermittent claudication or ischaemic pain while at rest and angiographically significant atherosclerosis, showed greater primary patency rates for those who had undergone angioplasty with a PCB as opposed to PTA (65.2% vs. 52.6%, p=0.02), with non-inferior safety outcomes at 12 months. However, TLR and functional outcomes such as rates of reintervention, thrombosis, and amputation were similar among groups [15].
The five-year results of the THUNDER trial demonstrated lower rates of TLR with PCB as compared to PTA (21% vs. 56%; p=0.0005), without significant differences in adverse events, amputation rate, and death [16].
Stenting
Self-expanding nitinol bare metal stents (BMS) improve outcomes compared to PTA in femoropopliteal arterial disease, even in complicated patients with multiple comorbidities. A meta-analysis of patients undergoing both balloon-expandable and self-expanding BMS for infrapopliteal disease showed satisfactory angiographic results, patency, and clinical outcomes over a 12-month period [17].
The Zilver PTX trial demonstrated a higher five-year primary patency rate with paclitaxel-eluting stents compared with PTA or provisional BMS for SFA disease (66.4% vs. 43.4%) [18].
Data from clinical trials reveal that the treatment of infrapopliteal disease with drug-eluting stents (DES) leads to significantly greater patency rates when compared to PTA or BMS. The ACHILLES trial, which included 200 patients with infrapopliteal disease, demonstrated higher primary patency rates with sirolimus-eluting stents (SES) (80.6% vs. 58.1%; p=0.006) and lower TLR rates (10% vs. 16.5%; p=0.26) compared with PTA [19].
Factors influencing the outcome after PTA
The success of PTA depends on the severity of PAD, the type of PAD (stenosis or occlusion), the length of the lesion, the quality of the vessels, concomitant disease (diabetes or coronary artery disease) and persistent risk factors (smoking, blood pressure, dialysis) [20]. Lesions may be suprainguinal (i.e., involving the aorta and iliac arteries), femoropopliteal or infrapopliteal. The effectiveness of PTA is greatest for lesions in the iliac arteries and progressively decreases for more distal vessels [21]. A meta-analysis of six studies reported that the immediate technical success of PTA and stenting for aorto-iliac occlusive disease was 96%, a higher success rate than for PTA alone [22].
The 12-month clinical effectiveness of bare metal nitinol stents to treat patients with symptomatic femoropopliteal PAD is acceptable and is impacted by clinical and lesion-specific characteristics. The 12-month patency in a meta-analysis of different studies was 69.8% [17].
PTA with stenting in the infrapopliteal artery is reserved for patients with chronic critical limb ischaemia (CLI). Despite this, there is no real consensus on when patients should receive stents. A recent systematic review and meta-analysis of randomised controlled trials showed that drug-eluting stents in the infrapopliteal arteries were associated with significantly lower rates of restenosis, TLR and amputations, as well as improved wound healing compared with balloon angioplasty and BMS [23].
Hybrid procedures are associated with good long-term outcomes in the treatment of multisegmental chronic peripheral arterial disease. Diabetes mellitus remains a determinant of worse outcome, while the severity of the disease and previous ipsilateral revascularisation are associated with poorer limb salvage [24].
Antithrombotic drugs after endovascular therapy for LEAD
Despite the increasing number of patients receiving endovascular stents due to progressive PAD, the knowledge regarding post-interventional management, and more specifically dual antiplatelet therapy (DAPT) after stent implantation, is still markedly limited.
DAPT is currently recommended for at least one month after intervention, irrespective of the stent type (bare metal vs. drug-eluting) [3]. Stenting below-the-knee arteries is often followed by a longer period of DAPT, but no specific evidence is available [3]. Anticoagulation has been prospectively tested after percutaneous infrainguinal revascularisation. Vascular patency was not improved, while bleeding was significantly increased [25].
Several studies investigated peripheral artery stent implantation outcome after 12 months with a DAPT therapy regime of 4-6 weeks. The reintervention rate in complex lesions after stenting was between 14.2% and 28.3% [26,27].
Furthermore, drug-coated stent studies revealed low reintervention rates with a TLR of 4% in the MAJESTIC trial (single arm) [28] and 9.5% in the Zilver PTX trial [29].
In patients with symptomatic peripheral artery disease, ticagrelor was not shown to be superior to clopidogrel for the reduction of cardiovascular events. Major bleeding occurred at similar rates among the patients in the two trial groups [30].
In patients with coronary artery disease (CAD), the coexistence of LEAD is associated with a worse prognosis irrespective of the clinical presentation. The coexistence of LEAD in patients with CAD may be an argument for prolonged DAPT. The PRODIGY trial tested DAPT duration after acute coronary syndrome (ACS). Prolonged (24 months) vs. short (6 months) DAPT conveyed a lower risk of the primary efficacy endpoint, a composite of death, MI or cerebrovascular accidents, in patients with LEAD. A significant interaction (p=0.01) suggests specific benefits only in patients with concomitant LEAD [31]. In the PEGASUS-TIMI 54 trial, the addition of ticagrelor 90 mg twice a day or 60 mg twice a day on top of low-dose aspirin in stable patients with prior MI (1-3 years) was investigated. Among patients with known LEAD (5% of the entire population), ticagrelor significantly reduced the risk of major adverse limb outcomes (acute limb ischaemia and peripheral revascularisation). Therefore, long-term ticagrelor on top of low-dose aspirin may be considered in LEAD patients with prior MI (<3 years) [32].
In LEAD patients who underwent infrainguinal percutaneous revascularisation, DAPT may be prolonged beyond one month when there is a prior history (<1 year) of ACS and/or percutaneous coronary intervention (PCI) [3].
Antithrombotic therapy in LEAD patients requiring long-term oral anticoagulation
The evidence is scarce to support a specific antithrombotic regimen in patients with LEAD and an indication for oral anticoagulation (OAC). A post hoc analysis of the ROCKET-AF trial reported a significant interaction for major and non-major clinically relevant bleeding in patients with LEAD treated with rivaroxaban vs. warfarin compared to patients without LEAD (p=0.037) [33].
The duration of combined therapy should be as limited as possible (one month), depending on the clinical indication and bleeding risk. The addition of an antiplatelet treatment may depend on concomitant CAD and the need for LEAD endovascular revascularisation. With the exception of below-the-knee stenting or complex lesions at very high risk of thrombosis, triple therapy (i.e., aspirin, clopidogrel and an anticoagulant) is discouraged in this setting [3].
Conclusion
The current landscape for peripheral vascular interventions includes various technologies that may improve clinical outcomes in PAD. The use of DEB increases patency and TLR outcomes in both femoropopliteal and infrapopliteal PAD. Stenting has a rising role in both bail-out and primary interventions and is particularly promising with the development of DES. Further well-designed and highly powered trials are needed to determine the ideal treatment strategies for the management of patients with PAD.