Women have distinct patterns of ischaemic heart disease (IHD)
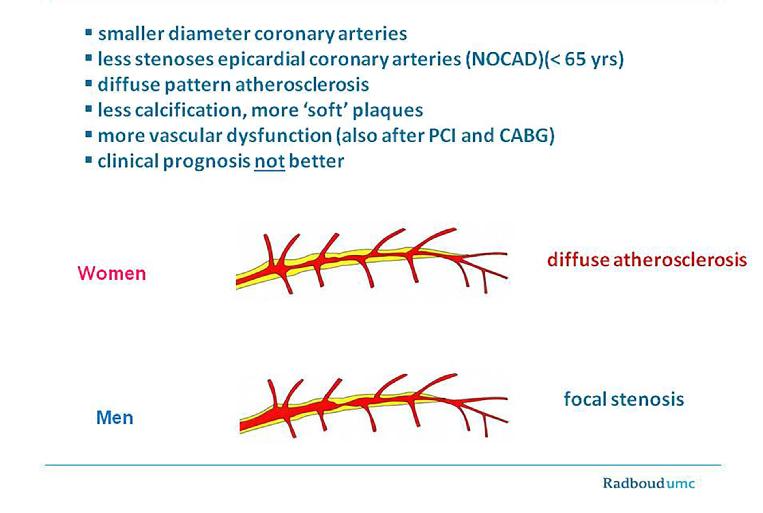
Women have a smaller diameter of the coronary arteries than men, when corrected for body surface area. Postmortem studies and CT angiograms show clinically relevant sex differences in plaque morphology with women having fewer calcifications, less focal obstruction and a more diffuse pattern of atherosclerosis with “outward remodelling” and “soft” plaques than men at all ages (Figure 1) [1,2]. Combined structural and functional disorders of the coronary (micro-) circulation are involved in the various manifestations of IHD [3]. In women, at middle age “functional IHD” is far more prevalent than obstructive coronary artery disease (CAD), which presents at an earlier age in men. In patients with non-obstructive coronary artery disease (NOCAD) at coronary angiography, lesions up to 20% can be present without being noticed, whereas intravascular ultrasound (IVUS) reveals the presence of atherosclerosis [4]. In clinical practice, IVUS is not regularly carried out in these patients and their symptoms are often misinterpreted as being of non-cardiac origin, resulting in undertreatment of symptoms and risk factors. Despite this, NOCAD has a higher five-year event rate in symptomatic women than in men, especially when signs of ischaemia are present [5]. As many cardiologists are still mainly focused on obstructive CAD in the epicardial coronary arteries, more attention is needed to recognise better the more diverse manifestations of IHD in women.
Figure 1. Female pattern of ischaemic heart disease
Symptoms of stable angina in women
The clinical evaluation of symptoms of angina pectoris in women is still considered according to the male standard. Risk estimation charts in the guidelines are primarily based on diagnosing obstructive CAD. In selected patients undergoing PCI for single-vessel disease, the main symptoms of angina are not different among men and women [6]. However, this selection bias in coronary lesions does not reflect daily practice. Whereas classic angina symptoms are more related to obstructive CAD, the combination of NOCAD with endothelial dysfunction frequently occurs in young and middle-aged women, with a more atypical symptom presentation. These symptoms occur both at rest and during exercise and may vary over time. Endothelial dysfunction and coronary spasm fluctuate within days/weeks and may present in a crescendo/decrescendo pattern. Stressful events often aggravate the duration and intensity of symptoms. Gender differences in communication frequently hamper the early recognition of angina in women. Women's communication is more likely to be emotional, subjective, polite, and self-revealing with more concern and awareness for the feelings of others. Men usually report their symptoms in a direct way, while female patients ask more questions, present more and diverse symptoms and give more detailed histories of their activities. This can be misleading to both female patients and their doctors for the correct interpretation of symptoms of IHD. The diverse symptoms of stable angina in women are depicted in Table 1.
Table 1. Classification of angina with female-specific aspects (♀)
Typical angina (definite)
- Meets 3 of the following characteristics:
- oppressive substernal chest discomfort
- provoked by exertion or emotional stress
- relieved by rest and/or nitrates within minutes
- ♀ squeezing, tight, chest discomfort
- ♀ radiation to chest, jaw(s), left armpit and/or left arm, neck, shoulders and inter-scapular area
- ♀ may last longer than minutes
- ♀ crescendo/decrescendo character (spasm)
- ♀ dyspnoea, anxiety, mental stress-related
- ♀ extreme tiredness, also often after angina episode
Atypical angina (probable)
- Meets 2 criteria
- ♀ both typical and atypical symptoms in NOCAD
Non-anginal chest pain
- Lacks or meets only one characteristic criterion
- ♀ beware of cardiac anxiety disorder
Adapted with permission from Montalescot G et al, 2013 ESC guidelines stable CAD [7].
Coronary microvascular disease (CMD)
In many women with “normal” coronary arteries, recurrent angina is frequently provoked by exercise, suggesting obstructive CAD. However, the presence of coronary microvascular disease (CMD) is more likely if there is a variable threshold of physical activity that provokes angina, if the chest pain persists for several minutes after effort is interrupted and/or if there is a slow or poor response to short-acting nitrates [7,8]. In addition to exercise-induced angina, (prolonged) chest pain at rest and/or at night/early morning is often reported, which may be provoked by vasospasm. Also, low heart rate activities such as mental arousal or palpitation are more common triggers of angina than in patients with obstructive atherosclerosis [9]. In many patients, dyspnoea on exertion is also reported to be an equivalent of a disturbed oxygen demand, with a general feeling of tiredness and loss of energy. Women with CMD often have concentration problems and are more prone to work-related problems and “burn-out”.
How to identify the female patient at risk for premature IHD
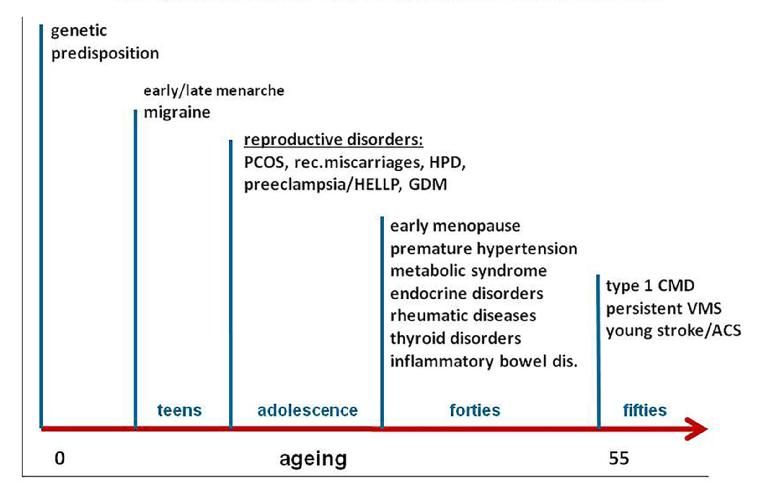
Symptom evaluation alone is not sufficient to distinguish angina pectoris related to endothelial dysfunction/ischaemia from non-cardiac causes of chest pain. The presence of traditional risk factors (RF), female-specific RF and non-traditional risk variables may importantly add to the likelihood of IHD. A positive family risk is an even stronger risk factor for women than for men and importantly adds to risk prediction at a younger age [10]. Migraine is related to an increased family risk for CVD, premature vascular dysfunction and a higher susceptibility for thrombosis and inflammation [11]. Reproductive and pregnancy-related factors predispose to earlier signs of endothelial dysfunction, vascular inflammation and atherosclerosis [12]. In a recent meta-analysis among the various reproductive RF that are known, a previous history of preeclampsia/HELLP was found to be the strongest RF for CVD, with a twofold higher risk [13]. This has now also been incorporated in the 2016 ESC guidelines on CVD prevention [14]. When women are in their forties/early fifties, inflammatory diseases often occur, such as inflammatory bowel disease, thyroid disorders, the metabolic syndrome and rheumatic disorders. These are non-traditional risk variables that may add to a higher susceptibility for endothelial dysfunction [15]. The horizontal life course of women who are more prone to premature signs of IHD is depicted in Figure 2. At older age (>65 years), the traditional RF apparently overrule the abovementioned RF with a shift in the pattern of IHD to more obstructive disease. In parallel, angina symptoms may become more “typical”. However, elderly women often have RF that may enhance diastolic dysfunction, with concomitant symptoms of dyspnoea on exertion. These symptoms can interfere with symptoms of angina and make the diagnosis difficult. The impact of vascular dysfunction and spasm is presumed to decrease at older age, when vascular stiffness and atherosclerosis are more extensively developed.
Figure 2. Female-specific and non-traditional risk factors: risk variables for early endothelial dysfunction/premature CVD.
ACS: acute coronary syndromes; CMD: coronary microvascular dysfunction; CVD: cardiovascular disease; GDM: gestational diabetes mellitus; HELLP: haemolysis, elevated liver enzymes and low platelets syndrome; HPD: hypertensive pregnancy disorders; PCOS: polycystic ovary syndrome; VMS: vasomotor symptoms
Conclusion
Women have different manifestations of IHD during their lifetime.
At younger age, NOCAD and coronary (micro-)vascular dysfunction predominate over obstructive CAD, resulting in a different symptom presentation.
Female-specific RF and non-traditional risk variables may contribute to identifying younger women at increased CVD risk.
Doctors should be aware of the different communication styles among men and women.