Definition
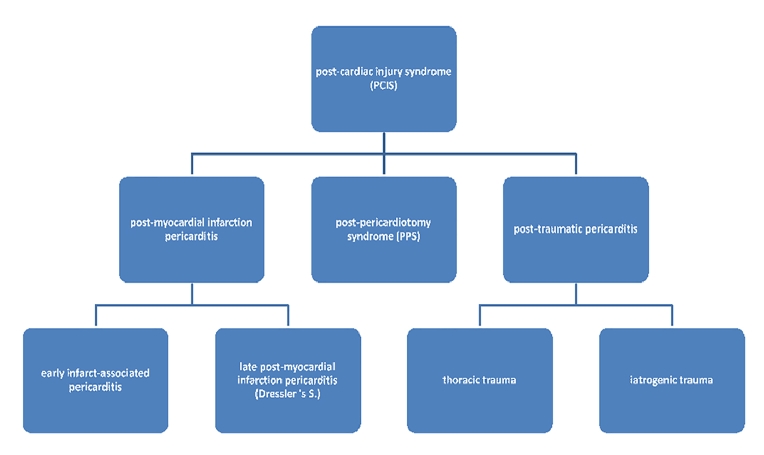
Post-cardiac injury syndrome (PCIS) refers to an aetiologic heterogenous group of autoimmune-mediated conditions of pericardial, epicardial, and myocardial inflammation. It is important to realise that inflammation is not confined to the pericardium and epicardium but also affects the myocardium to some extent. Nevertheless, symptoms of epicardial and pericardial inflammation, such as pain, effusions, and fever, dominate the clinical picture. Signs of ventricular dysfunction such as heart failure, ventricular arrhythmias or severe conduction abnormalities are very rare. If present, they rather reflect worsening of pre-existing heart failure by a second systemic inflammatory hit. By definition, post-cardiac injury syndromes include: post-myocardial infarction pericarditis triggered by ischaemic myocardial necrosis, post-pericardiotomy syndrome (PPS) after surgical trauma and, finally, post-traumatic pericarditis due to either iatrogenic or accidental injury [1] (Figure 1).
Figure 1. Post-cardiac injury syndrome and its subentities.
Epidemiology and clinical picture
A prospective cohort study of 1,162 cases identified post-cardiac injury as the leading cause of pericarditis, accounting for 21% of patients [2].
Post-myocardial infarction pericarditis can be differentiated into early infarct-associated pericarditis and late post-myocardial infarction pericarditis, also known as Dressler’s syndrome. The former occurs within five days after myocardial infarction. With an incidence of <5% it has become quite rare in developed countries. A major risk factor is delayed angioplasty. The risk of recurrence however is very low. Dressler’s syndrome, on the other hand, occurs two to eight weeks after infarction and is also very rare nowadays in clinical settings. Prior to the reperfusion era it had been reported with an incidence of 5%. Newer studies have reported the de facto disappearance of Dressler’s syndrome, a fact that is not yet fully understood. On the one hand, this observation might reflect under-reporting of subclinical cases. However, diminution of infarct size by means of early reperfusion and immune-modulatory effects of standard-of-care drugs, such as ACE inhibitors, some beta-blockers, statins, and aspirin, mainly explain these observations. Importantly, and in marked contrast to early infarct-associated pericarditis, Dressler’s syndrome implies a relevant risk of recurrence. Nevertheless, complications such as late cardiac tamponade and constrictive pericarditis are rare for early infarct-associated pericarditis as well as for Dressler’s syndrome [3,4].
Post-pericardiotomy syndrome (PPS) has been found to be a relatively frequent complication of cardiac surgery with an incidence of 10-40% in patients undergoing cardiac operation. This wide range of incidence largely reflects inhomogeneous diagnostic criteria. PPS classically occurs days to several weeks after cardiac surgery [5–7]. It is of importance to distinguish PPS from uncomplicated postoperative effusion, which affects up to 85% of patients undergoing cardiac surgery. Uncomplicated postoperative effusions lack signs of systemic inflammation [8].
Last, but not least, post-traumatic pericarditis spans entities ranging from accidental blunt or sharp cardiac trauma to a wide variety of iatrogenic trauma. Accidental trauma-associated pericarditis is either rare or – more likely – under-reported. Accordingly, there are no systematic and valid data available in this context. Pericarditis due to iatrogenic trauma, on the other hand, has an incidence of 0.5% to 5% depending on the interventional trigger. Percutaneous coronary interventions, pacemaker lead insertions, radiofrequency ablation and even Swan-Ganz catheterisation can predispose to myocardial injury of various degrees and may culminate in post-traumatic pericarditis [9-11].
Diagnostic criteria
A major drawback in the study, understanding and treatment of PCIS is the lack of reliable and generally accepted diagnostic criteria defining the disease entity with appropriate specificity. This is largely due to its clinical and aetiological heterogeneity and the absence of a single consistent pathognomonic finding. Patients suffering from PCIS frequently seek medical assistance because of chest pain (>80%), low grade fever (50-60%) and dyspnoea (50-60%). Clinical evaluation usually reveals mild to moderate effusions both in the pericardium (>80%) and in the pleural space (>60%), and sometimes pericardial friction rubs (30-60%) on auscultation. Laboratory analysis shows systemic inflammation with elevation of C-reactive protein (CRP) (74%), and elevated blood leucocytes. ECG can provide evidence of typical alterations associated with pericarditis, such as widespread concave ST-segment elevations and PR depressions in multiple leads (except for aVR and sometimes V1, where ST-segment depressions and PR elevations are predominant). Identification and interpretation of the aforementioned findings is, however, often complicated by the underlying pathological condition which triggered PCIS. A diagnosis of PCIS may be considered in patients with chest pain, fever, pericardial or pleural effusions, and a re-emerging systemic inflammatory response syndrome after an appropriate latency period following prior myopericardial or pleural injury. Importantly, alternative causes for inflammation and effusion need to be ruled out before a diagnosis of PCIS can be established [1]. Diagnostic criteria for PPS validated in three large clinical trials encompass five features, two of which have to be met in order to diagnose PPS: fever without alternative causes, pleuritic chest pain, friction rub and evidence of new or worsening pleural or pericardial effusion [5-7, 12].
Aetiology
Damage to pericardial or pleural mesothelial cells in combination with blood entering the pericardial space represents the initial trigger of PCIS. There is evidence for a correlation between elevated antimyocardial antibody titers and the incidence of PCIS after cardiac surgery. Patients undergoing open heart surgery in a small prospective study were tested for baseline and follow-up anti-myosin antibody titers. PPS occurred only in patients with detectable anti-myosin antibody titers and positively correlated with its severity. Based on these observations and insights from animal models, we therefore hypothesise that systemic release of cardiac antigens, together with non-specific, injury-mediated activation of the innate immune system initiate adaptive self-antigen specific responses, as evidenced by an increase of antimyocardial antibody titers and expansion of heart-specific CD4 T cells, which in turn promote recruitment of myelo-derived inflammatory cells to the heart. It is important to realise in this context that the epicardium, pericardium and pleura are much more susceptible to the accumulation of inflammatory cells than the cardiac interstitium, even in the presence of cardiomyocyte specific auto-immune responses. Taken together, the latency period between cardiac injury and PCIS, as well as the generally favourable response to anti-inflammatory drugs, both strongly support the hypothesis of an autoimmune-mediated pathogenesis [1,13,14].
Prevention
Prevention is not an issue in post-myocardial infarction pericarditis and post-cardiac injury pericarditis due to its low incidence. PPS, however, markedly contributes to morbidity in patients after cardiac surgery. Accordingly, most preventive studies have been conducted in the setting of PPS. Meta-analytic pooling showed that perioperative colchicine decreased the risk of PPS (OR 0.38, 0.22-0.65). Administration of methylprednisolone (OR 1.13, 0.57-2.25) and aspirin (OR 1.00, 0.16-6.11), however, showed no benefit [15,16].
The efficacy of colchicine as a potent preventive drug against PPS was confirmed in the randomised, double-blind, multicentre COPPS trial. Patients were randomised on the third postoperative day to receive either placebo or colchicine for one month. An initial loading dose of 1.0 mg twice daily was administered on the first day followed by a maintenance dose of 0.5 mg twice daily with a weight-adjusted dose reduction of 50% for patients weighing below 70 kg. The study showed superiority of colchicine vs. placebo with a number needed to treat (NNT) of 8 (95% CI: 27.3-75.6). Subsequently, the COPPS-2 trial was conducted to gain additional information on efficacy and safety. There was a slight change in the regimen in terms of already starting colchicine administration two to three days before surgery without a loading dose. The COPPS-2 trial strengthened the findings of its predecessor with a NNT of 10. However, an increase of adverse gastrointestinal effects was documented, with a number needed to harm (NNH) of 12. Observed adverse events primarily consisted of gastrointestinal intolerance without serious complications [6,7].
Moreover, a retrospective, single-centre study suggested that in-hospital administration of diclofenac after cardiac surgery protects from PPS (OR 0.34, 0.18-0.65) [17].
Treatment
Mainstays of treatment are anti-inflammatory NSAID and the adjunct use of colchicine, albeit that there are no randomised controlled trials addressing specific treatment approaches for PCIS. Treatment decisions must be taken with care given the potential side effects and risks associated with NSAID and colchicine in cardiac and post-surgery patients. Simple postoperative pericardial effusions without evidence for systemic inflammation do not warrant treatment [18].
Aspirin is the first choice for patients already under antiplatelet therapy. Dosages vary between 750 mg and 1,000 mg every six to eight hours. Known coronary artery disease strongly supports the use of aspirin in such patients. A treatment period of one to two weeks before tapering is usually sufficient. Recurrent symptoms demand prolonged treatment over two to four weeks. Alternatively, ibuprofen 600 mg every eight hours may be prescribed over one to two weeks in case of initial diagnosis or over two to four weeks in case of recurrence. However, it is important to be aware that ibuprofen interferes with the antiplatelet aggregation effects of aspirin in patients after coronary bypass grafting or PCI. Treatment with NSAID is continued until symptoms resolve and CRP normalises followed by subsequent tapering. Careful monitoring of renal and cardiac function is essential. We strongly suggest administration of proton pump inhibitors for adverse gastrointestinal effects prevention. NSAID should be avoided or at least closely monitored in patients with impaired kidney function, volume depletion or known heart failure.
Colchicine has been proven effective for the reduction of recurrences in pericarditis. However, there are insufficient data on its use in PCIS treatment. One retrospective, single-centre study showed a reduction of procedural intervention if PPS was treated with colchicine in combination with an anti-inflammatory drug as compared to anti-inflammatory drugs only (OR 0.43, 0.95-0.99). This study, however, was not designed to evaluate specifically the efficacy of colchicine in PPS or PCIS [19]. Nevertheless, the 2015 European Society of Cardiology guidelines endorsed administration of colchicine in PCIS. Dosing is the same as in PPS prevention with 0.5 mg twice a day and a dose reduction of 50% for patients weighing below 70 kg. Patients should be monitored for creatinine elevation and blood panel changes at two- to three-week intervals. Moreover, we instruct all our patients to be aware of diarrhoea and nausea heralding colchicine overdose.
Glucocorticoid administration is required for a minority of patients with contraindications for aspirin and NSAIDs or in case of refractory symptoms. Data for PCIS are lacking, but best clinical practice suggests similar dosing to that administered in pericarditis, e.g., 0.25 to 0.5 mg/kg/d. If steroids are used, it is of the upmost importance to taper dosages very slowly to avoid recurrences. In patients treated with steroids and NSAID/colchicine, steroids must be tapered before colchicine is stopped.
Recurrence of PCIS has been reported to be lower than recurrences of idiopathic pericarditis, ranging between 10% and 15% [1]. In contrast to idiopathic pericarditis, however, the risk of pericardial constriction is substantial for PCIS, approximating 2.8% over 72 months. Therefore, affected patients need long-term follow-up [20]. Our institutional policy recommends clinical follow-up examination and echocardiography one and three years after PCIS.
In our opinion, the best standard of care includes: colchicine for prevention of PPS, aspirin in combination with colchicine for initial treatment, and glucocorticoids for recurrence or contraindications to aspirin. Proton pump inhibitors are required in combination with aspirin (Table 1).
Table 1. Treatment recommendations.
- PPS Prevention
- Colchicine 0.5 mg twice daily (b.i.d.), starting 3 days before and continuing for 1 month after surgery
- Contraindications to NSAID:
- <70 kg: reduce colchicine to 0.5 mg o.d.
- GFR <30 ml/min or unstable kidney function: no colchicine
- GFR 30–60 ml/min: adjust colchicine dose to kidney function
- PCIS Treatment
- Aspirin 750 mg three times daily (t.i.d.) for 2 weeks + Colchicine 0.5 mg b.i.d. for 6 months + Proton pump inhibitor
- Contraindications to NSAID:
- No aspirin; use prednisolone instead: 0.5 mg/kg o.d. for 1 month, subsequent slow tapering
- Adverse effects of aspirin: consider reducing dose to 500 mg t.i.d.
- <70 kg: reduce colchicine to 0.5 mg o.d.
- GFR <30 ml/min or unstable kidney function: no colchicine
- GFR 30-60 ml/min: adjust colchicine to kidney function
- PCIS Recurrence
- Prednisolone 0.5 mg/kg once daily (o.d.) for 1 month, subsequent very slow tapering + Aspirin 750 mg t.i.d. for 2 weeks + Colchicine 0.5 mg b.i.d. for 6 months + Proton pump inhibitor
- Do not stop colchicine before steroids!
- Contraindications to NSAID:
- No aspirin; consider azathioprine (2 mg/kg o.d.) together with steroids
- Adverse effects of aspirin: consider reduction to 500 mg t.i.d.
- <70 kg: reduce colchicine to 0.5 mg o.d.
- GFR <30 ml/min or unstable kidney function: no colchicine
- GFR 30-60 ml/min: adjust colchicine to kidney function
- Prednisolone 0.5 mg/kg once daily (o.d.) for 1 month, subsequent very slow tapering + Aspirin 750 mg t.i.d. for 2 weeks + Colchicine 0.5 mg b.i.d. for 6 months + Proton pump inhibitor