Introduction
Pericardial involvement in neoplastic diseases can have a varied presentation depending on the underlying aetiology and duration of illness. This includes pericarditis, pericardial effusion, constrictive pericarditis and cardiac tamponade.
Prevalence
Primary tumours of the pericardium are extremely rare and include mesothelioma, sarcoma and teratoma. Secondary involvement of the pericardium from neoplasms of the lung, breast, mediastinal lymphomas and haematological malignancies is more common [1,2,3].
Secondary tumours from the lungs and breast account for more than 30% of reported cases in most series and are the commonest owing to their higher incidence and also to their anatomical proximity, the pericardium is involved in nearly two thirds of the cases of mesothelioma [1,3]. It is also to be borne in mind that nearly half the patients with symptomatic pericardial disease may have a non-neoplastic aetiology, for example, secondary to radiation therapy, drug-induced, infection in immunocompromised, autoimmune disorders, and idiopathic pericarditis [1,2,4,5].
Clinical presentation and diagnosis
Pericarditis
Acute pericarditis is an inflammatory process, and may occur with or without effusion [2,6,7]. Pericarditis can be diagnosed clinically when any two of the following features are present [2,6,7,8].
- Chest pain – described as a sharp, retrosternal pain increasing with inspiration, eased by sitting up and leaning forward.
- Pericardial rub – a high pitched, scratchy friction sound best heard along the left sternal edge, usually triphasic and highly specific of pericarditis.
- ECG changes – widespread concave upward ST-segment elevation and PR segment depression are described in the acute phase.
- Presence of new or worsening pericardial effusion.
In addition, other signs and symptoms may also be present depending on the primary illness and accompanying comorbidities.
The clinical diagnosis is straightforward and most of the pericarditis is self-limiting. A detailed diagnostic work-up is only warranted in cases with suspicion of malignant involvement. It is essential to look for high-risk signs and symptoms which include high-grade fever (38° C), subacute course, large effusion (more than 20 mm), cardiac tamponade and absence of response to NSAIDs [2,8,9].
Pericardial effusion
Pericardial effusions in the majority of cases are clinically silent and asymptomatic. They are detected incidentally on chest X-ray and echocardiography. The most common symptoms of pericardial effusion are non-specific, and the slow accumulation of pericardial fluid results in delayed manifestations of signs and symptoms [2,4,10]. Rapidly accumulating effusions are more likely to cause symptoms [1,11]. Dyspnoea, cough and pleuritic chest pain are the most common symptoms. Compression of the adjacent anatomical structures, such as the oesophagus, may cause dysphagia, and recurrent laryngeal nerve and phrenic nerve compression may manifest as hoarseness and hiccups, respectively. Non-specific symptoms such as excessive tiredness and palpitation secondary to reduced cardiac output are not uncommon [1,2,12].
Pericardial effusions are difficult to diagnose, as the clinical examination might be essentially normal in haemodynamically stable patients and tachycardia may be the only sign [1,2]. Cardiomegaly with a flask-shaped heart and clear lung fields on chest X-ray is suggestive of significant pericardial effusion [1,8,10]. ECG may be normal or may show non-specific ST and T-wave abnormalities. Occasionally, low QRS voltage may be seen, but that is more specific for tamponade [1]. Two-dimensional transthoracic echocardiography is the preferred investigation for pericardial effusion as it can determine the size, location, and haemodynamic significance of a pericardial effusion and is also low cost and easily available [1,2,4,13,14]. Upon detection of the effusion, it is imperative to assess the size of the effusion, look for signs of tamponade and any evidence of associated illness [2]. Though echocardiography is fairly conclusive in diagnosing pericardial effusion (Figure 1, Figure 2), computed tomography (CT) or cardiac magnetic resonance (CMR) imaging is useful in providing a wider field, detecting loculated effusion, thickening of the pericardium, and detecting pericardial and myocardial malignant deposits. Cytology of the deposits may be helpful in the differentiation of malignant and non-malignant deposits (pericardial mesothelial cells could resemble malignant cells but could be identified by special stains) and also picks up associated thoracic abnormalities [1,2,12,13,14)
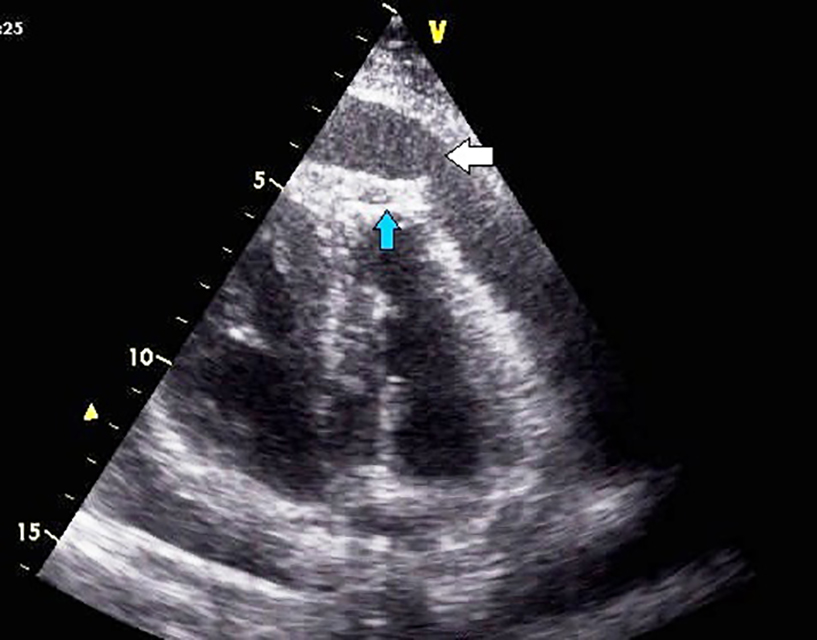
Figure 1. Two-dimensional transthoracic echocardiography of a patient with primary bronchogenic carcinoma, in apical four-chamber view showing pericardial effusion (broad white arrow) and malignant deposits in the left ventricular apex (blue arrow).
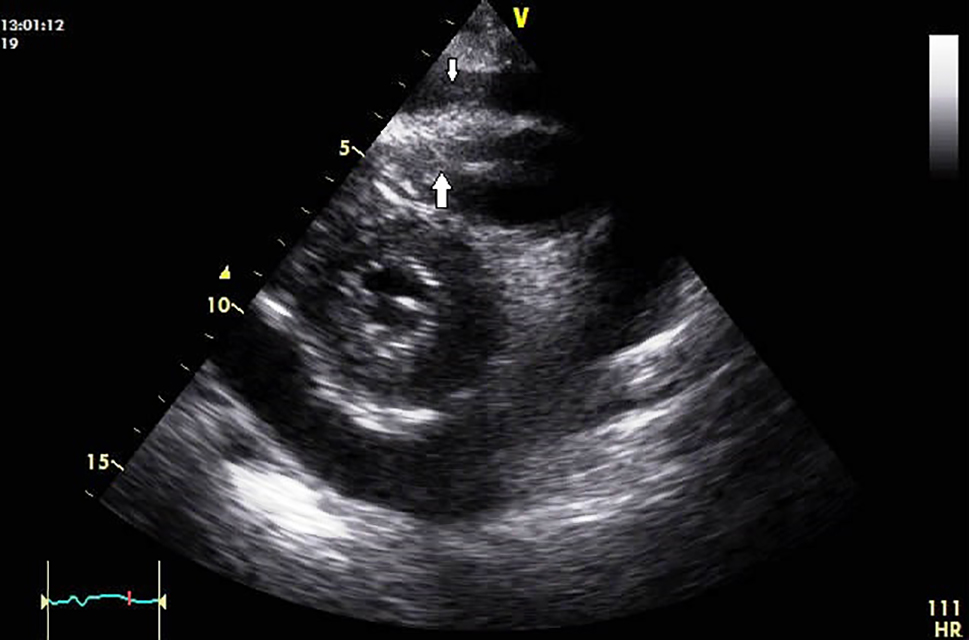
Figure 2. Two-dimensional transthoracic echocardiography of a patient with laryngeal carcinoma treated by surgery and with radiation in the past. Parasternal short-axis view showing pericardial effusion with tumour deposits at the apex of the right ventricular cavity (broad white arrow). Also, few deposits were seen over visceral pericardium (narrow white arrow).
Constrictive pericarditis
Constrictive pericarditis is usually seen after radiation therapy for breast malignancy or Hodgkin’s disease [2], leading to reduced filling of the ventricles due to the unyielding and scarred pericardium. This is characterised by signs and symptoms of systemic venous congestion with preserved ventricular systolic function [2,8]. Patients present with complaints of fatigue, shortness of breath, and palpitation. Clinical examination may reveal signs of right-sided heart failure [2,8,15].
Cardiac tamponade
Cardiac tamponade is a medical emergency, and may be fatal if emergent and appropriate intervention is not initiated. Tamponade develops when the pericardial fluid pressure is greater than the intracardiac filling pressures and compromises the cardiac filling which leads to reduced cardiac output and cardiogenic shock [8]. When tamponade develops, clinical signs to look for include distended neck vein with elevated jugular venous pressure with diminished or absent y descent, a paradoxical elevation in the jugular venous pressure during inspiration (Kussmaul sign), an exaggerated drop of greater than 10 mmHg in the systolic blood pressure during inspiration (pulsus paradoxus), and muffled heart sounds on cardiac auscultation [2,8,12]. The classic triad of hypotension, tachycardia and muffled heart sounds (Beck’s triad) is seldom seen in malignant pericardial tamponade as they are of subacute onset [1].
ECG may show low voltage complexes and electrical alternans, and cardiomegaly on chest X-ray [2,12,16]. Echocardiographic features of tamponade include late systolic collapse of the right atrium (RA) and early right ventricle (RV) diastole collapse, bulging of the interventricular septum towards the left ventricle (LV) during inspiration and reversal of the movement during expiration, increased variability in the transmitral and tricuspid flow gradient with respiration and pulmonary venous diastolic flow reversal. Dilated inferior vena cava (IVC) with absent collapse during inspiration and diastolic flow reversal in hepatic veins are other signs of cardiac tamponade [2,8,12,13,14]. The clinical severity and the haemodynamic status are determined by the pace of collection and the volume of the pericardial fluid in addition to the compliance of the pericardium and the cardiac chambers [2]. Transthoracic echocardiography is the preferred imaging tool in suspected pericardial diseases. Echocardiography along with Doppler is the most economical and accurate method for detecting pericardial effusion, assessing haemodynamic status and early diagnosis of tamponade. In addition, it can be used to look for any underlying myocardial disease leading to systolic dysfunction [2,4,13,14]. CT and CMR may be of use in diagnosing constrictive pericarditis and in differentiating it from restrictive cardiomyopathy [2,13,14]. CT is a valuable and complementary imaging modality and is best for delineation of calcified structures and measuring pericardial thickness, treatment planning of neoplastic effusions and also during follow-up [2,13,14]. For complete assessment of pericardial disease CMR is the best choice [2,17].
Demonstration of uptake of 18F-fluorodeoxyglucose (FDG) tracer in the pericardium by either positron emission tomography (PET), or in combination with CT (PET/CT) in patients with solid tumours and lymphoma may indicate malignant pericardial involvement and aids in the diagnosis, staging and assessment of response to treatment [2,18]. To increase the diagnostic yield and for early and complete assessment and better management of pericardial diseases, it would be prudent to integrate and optimise the use of all imaging modalities [13,14].
In order to improve the diagnostic yield and optimise the management, an integrated approach using different imaging modalities as appropriate should be the ideal strategy [2,13,14]. However, the analysis of the pericardial fluid and the pericardial biopsy specimen is central to the diagnosis of malignant effusion; the diagnostic yield from the tumour markers in the pericardial fluid remains inconclusive to differentiate malignant and benign pericardial effusion [2,19].
Treatment
Medical treatment
Not all pericardial involvement in neoplastic disease is malignant. Acute pericarditis secondary to an underlying inflammatory process responds to medical treatment. The first-line treatment in acute pericarditis is aspirin or NSAIDS [7,9]. In patients with a non-viral identifiable aetiology, the underlying disease also needs to be treated appropriately [2,20].
Colchicine in reduced doses is recommended to enhance the response to medical treatment and reduce recurrence rates; tapering is not mandatory [2,21]. Corticosteroids are not favoured owing to the increased risk of recurrence and are second-line mainly for patients with contraindications or failure of adequate response to the first-line agents. Low to moderate doses are recommended and are given along with colchicine [2,8].
In the majority of cases pericardial effusion has an underlying disease; hence, the treatment should be directed towards the underlying cause for complete resolution and to prevent recurrence [2,4,16]. In addition, the extent and prognosis of the underlying disease along with the patient’s general condition should be considered before devising a treatment strategy [1,4].
In an asymptomatic, haemodynamically stable patient with a small pericardial effusion, careful monitoring, avoiding dehydration and regular follow-up with echocardiography may suffice [2,8]. In symptomatic cases with a non-inflammatory aetiology and with failed medical therapy, pericardial drainage is suggested.
Pericardiocentesis
Pericardiocentesis is the treatment of choice for cardiac tamponade. Needle pericardiocentesis under echocardiographic or fluoroscopic guidance is preferred [22]; blind procedures are only attempted in dire emergencies as a life-saving procedure [2]. The surgical approach is reserved to certain situations such as persistence of symptoms or bleeding into the pericardium [2].
For echocardiography-guided pericardiocentesis, the point of needle entry corresponds to the area of maximum fluid collection and closest to the body surface. Care should be taken to avoid injuring the nearby vital visceral organs and the vascular bundles. The puncture site is marked on the skin before the pericardiocentesis; alternatively, the procedure can be carried out under echocardiographic monitoring [2,22]. For fluoroscopy-guided pericardiocentesis, a blunt tip needle with a saline-filled syringe under negative pressure is introduced from the subxiphoid area, and advanced with the needle pointing to the left shoulder. The lateral angiographic plane is preferred for monitoring. Once pericardial fluid has been aspirated, a multi-hole pigtail is introduced via the Seldinger technique and fluid is drained [2,22].
Pericardiocentesis with prolonged catheter drainage, until the drain is less than 30 ml/day, would significantly reduce the recurrence rates by promoting adherence of pericardial layers [1,2,4,9,12,16]. Large effusions have high recurrence rates and pericardial drainage is recommended in such cases.
Intrapericardial antineoplastic treatment
Depending on the underlying neoplasm, sclerosing and cytotoxic agents are instilled intrapericardially to prevent recurrence [2,5]. Cisplatin (lung cancer) and thiotepa (breast cancer) are the most effective cytotoxic agents. Tetracycline as a sclerosing agent has very good results but has a high incidence of atrial arrhythmias, chest pain and recurrent fever [1,2,5,23].
Radiation therapy
In pericardial effusions secondary to radiosensitive tumours such as lymphomas and leukaemia, radiation therapy is the therapy of choice but has the risk of causing pericarditis and myocarditis [2,5].
Pericardial window
As malignant pericardial effusions have high recurrence rates, pericardiocentesis alone may not be sufficient. Some cases may require surgical intervention in the form of either a less invasive pericardial window or pericardiectomy in cases of loculated effusion or when biopsy samples are needed [2,4]. For malignant cardiac tamponade, creating a pleuropericardial window by percutaneous balloon pericardiotomy, allowing fluid to drain into the pleural space and relieving the tension and giving symptomatic relief [2,23], and a pericardial window by a minimally invasive surgical approach are safe and effective treatment options [2,24].
A pericardial window is the surgical creation of a channel between the pericardial and pleural cavity, in order to facilitate free flow of fluid from the pericardial to the pleural cavity to give symptomatic relief to patients with large pericardial effusion and to prevent cardiac tamponade. A pericardial window can also be created by either video-assisted thoracoscopy or, percutaneously, by balloon pericardiotomy [2]. This is a palliative procedure in patients with malignant pericardial diseases with a grave prognosis and with high risk for more complex pericardiectomy. The disadvantage is that this is not a definitive treatment, and failure rates are high in the long run and may lead to loculated effusions [2].
Pericardiectomy
Constrictive pericarditis secondary to pericardial inflammation may resolve with supportive and medical therapy [8]. Pericardiectomy is the treatment of choice in chronic constrictive pericarditis. Complete pericardiectomy is favoured over partial, as constrictive symptoms may linger on after partial pericardiectomy [8,25,26]. Care is required to ensure that the right-sided cardiac chambers along with the venae cavae are free of pericardial attachments [26]. Piecemeal decortications are carried out only when the pericardium is adherent or calcified [2]. Pericardiectomy carries a high operative mortality risk, and the long-term outcome is dependent on the comorbidities and overall medical condition of the patient [2,25].
The approach to malignant pericardial disease management is usually based on the type and stage of the primary illness and the general medical condition of the patient. In those with advanced stages of the disease with a grave prognosis, the treatment is usually palliative and is aimed at symptom relief and improving the quality of life; in those with non-malignant pericardial involvement, aggressive treatment as per guidelines is advocated.
Conclusion
Pericardial involvement in malignant diseases is relatively frequent. A large proportion of this pericardial involvement is non-malignant. The spectrum of clinical presentation includes pericarditis, pericardial effusion, constrictive pericarditis and cardiac tamponade. Primary pericardial involvement is not common; secondary tumour spread to the pericardium from lung and breast carcinoma, lymphomas and leukaemia is more common. In patients with malignancy presenting with shortness of breath, a low threshold to investigate for possible pericardial involvement with an integrated systematic approach and optimum utilisation of multimodality imaging and pericardial biopsy will have a greater diagnostic accuracy. Therapy is dependent on the underlying aetiology, and is aimed at providing symptomatic relief and attaining haemodynamic stability.