Keywords
constrictive pericarditis, New York Heart Association, pericardiectomy
Abbreviations
CMR cardiac magnetic resonance
CP constrictive pericarditis
CT computed tomography
NYHA New York Heart Association
TTE transthoracic echocardiography
Introduction
The normal pericardium is composed of two layers, i.e., an inner serous layer and an outer fibrous layer. The pericardium has multiple functions to protect the heart and preserve its normal cardiac function including the maintenance of mechanical effects on the heart. The pericardium also has immunological, paracrine, vasomotor and, most importantly, fibrinolytic activity [1,2]. Constrictive pericarditis (CP) is a chronic inflammatory process involving the pericardium, which leads to fibrotic thickening, scarring and subsequently calcifications which could be quite extensive, extending into the myocardium [3,4,5,(6]. CP is characterised by abnormal diastolic filling patterns which may subsequently lead to impaired cardiac output and overt heart failure [3]. Although the pericardial constrictive syndromes could be related to a diverse spectrum of aetiologies, idiopathic, post-surgical and prior mediastinal radiotherapy are the three leading causes (in descending order) of CP in the western world [7,8]. Tuberculous pericarditis remains the leading cause of CP in underdeveloped countries and also in immunocompromised patients, particularly those infected with human immunodeficiency virus (HIV) or acquired immunodeficiency syndrome (AIDS) [7,9,10,11,12,13]. The primary causes of CP may be identified and treated to prevent the development of CP later on; however, in some cases CP may not be prevented.
As a result, we opted to review the prevention, suggested medical therapy or treatment modalities of the primary aetiologies for CP and currently recommended surgical options for constriction.
Prevention
Constrictive pericarditis may develop without an apparent primary cause and, in some cases, it is not preventable. However, the conditions which are primarily the leading causes of CP should be adequately managed to prevent the occurrence of CP (Figure 1) [7,14,15,16,17]. The following special circumstances, among others, should be addressed appropriately, as discussed below.
All forms of acute pericarditis.
Both acute and chronic infections including bacterial, viral, fungal and parasitic, or surgical infections that cause abnormal inflammatory response to the pericardium.
Chronic renal failure or end-stage renal disease is a well-documented cause of acute pericarditis which could in turn lead to CP.
Connective tissue disorders including systemic lupus erythematosus, rheumatoid arthritis, systemic sclerosis or scleroderma (and more) are important causes of pericardial diseases.
Acute pericarditis following an ST-elevation myocardial infarction, which is also called Dressler's syndrome, should be anticipated and managed accordingly. This warrants a close echocardiographic evaluation both acutely and during follow-ups.
Conditions associated with pulmonary diseases including pulmonary asbestosis, tumours including mesothelioma, and neoplastic pericardial infiltration are among important neoplastic conditions which could lead to constriction.
Incomplete drainage of purulent pericarditis could lead to recurrent pericarditis and subsequent constriction.
Post cardiothoracic surgery (any procedure where the pericardium is opened, damaged, or manipulated) including prior valve surgeries, congenital heart disease repair, etc., a proper echocardiographic evaluation needs to be implemented to rule out normal thickness CP which may be easily missed early on during the disease course [18].
Prior mediastinal radiation therapy, the most important cause of constriction in breast cancer patients or those who have received prior mediastinal radiotherapy.
Tuberculosis is the leading cause of CP in underdeveloped countries and particularly in immunocompromised patients.
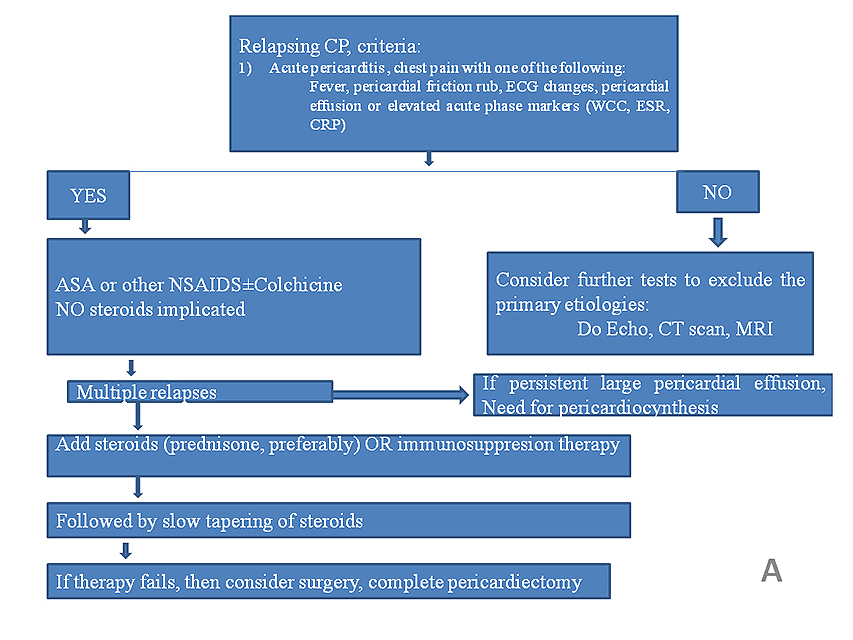
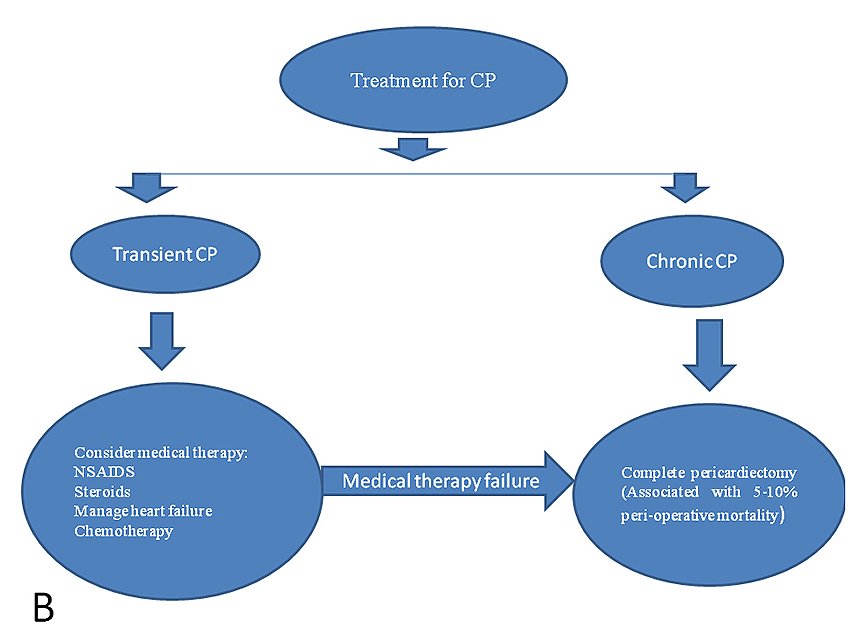
Figure 1. Flow charts showing (A) diagnosis and therapeutic approach for relapsing pericarditis, and (B) therapeutic overview for constrictive pericarditis. [17].
ASA: aspirin; CP: constrictive pericarditis; CRP: C-reactive protein; CT: computed tomography; ESR: erythrocyte sedimentation rate; MRI: magnetic resonance imaging; NSAIDS: non-steroidal anti-inflammatory drugs; WCC: white cell count
The overall general approach in the management of constriction
The general management of constriction is shown in Figure 1 [7,14,15,16,17]. The primary goal in CP is to improve cardiac function and it is crucial that the primary aetiology be identified and managed accordingly. The definitive cure for CP is primarily surgical in the form of complete pericardiectomy, where feasible [19,20,21,22]. Pericardiectomy typically leads to rapid haemodynamic and symptomatic improvements in most patients; however, in a subset of patients, particularly those with left ventricular systolic dysfunction, advanced New York Heart Association (NYHA) functional class, concomitant myocardial disease and also comorbidities, symptoms may persist after surgery. Medical management, such as careful observation or symptomatic treatment, has been suggested in less severe cases; however, this option remains controversial.
Diuretics have been used in the early stages of the disease to improve pulmonary and systemic congestion. However, diuretics should be used cautiously as any drop in intravascular volume may lead to a significant drop in cardiac output. A strict fluid restriction and low sodium diet may be recommended in some patients. It is important to anticipate complications which may arise due to failure for early diagnose of CP or improper management of constriction, as well as complications related to the primary aetiology of constriction or related to any existing pathology.
What are important additional general measures for outpatients?
Outpatient care may be appropriate during the early stages of the disease, particularly when the diagnosis is still uncertain and symptoms are relatively stable. It is important to emphasise and encourage a low salt and fluid-restricted diet as these are probably beneficial in order to avoid congestion and exacerbations of heart failure symptoms. Although no specific restrictions are needed, activity can often be severely limited by symptoms. However, decreased daily activities may be recommended in some patients.
Pharmacologic therapy
In the majority of cases, medical therapy is deemed ineffective unless there is prominent evidence suggestive of an inflammatory component which makes cardiac magnetic resonance (CMR) imaging very important in the detection of pericardial enhancement as part of the initial diagnostic modalities in patients suspected of pericardial disease, in particular CP. The management of CP differs from that of acute pericarditis, where the use of non-steroidal anti-inflammatory agents (NSAIDs), cyclooxygenase (COX)-2 inhibitors, colchicine, corticosteroids, or a combined therapy may be beneficial (Figure 1) [7,14,15,16,17]. Although reports have documented the benefit of optimal medical therapy for acute pericarditis, constriction may still develop over time after the initial insult. As a result, these patients need a close and regular follow-up, where echocardiography will be one non-invasive imaging modality to evaluate the patient, in addition to other modalities. Transient CP has been described, so those individuals with the diagnosis of constriction who are medically stable may be given a trial of conservative treatment for a period of two to three months (using NSAIDs and/or steroids) (Figure 1) [7,14,15,16,17]. This should only be considered in individuals with optimised volume status and controlled symptoms.
The following specific treatment modalities should be considered for each of the special situations highlighted.
- Subacute CP may respond to steroids if treated before pericardial fibrosis develops.
- Diuretics, particularly loop diuretics, are the mainstay to relieve congestion and to optimise clinical volume status; however, they may decrease preload to the point of reducing cardiac output and thus require closer monitoring.
- Any other medications used to treat patients with CP should be specifically directed to the underlying aetiology of the pericardial disease.
- Any therapy directed towards the causative disease, e.g., antituberculosis therapy, is appropriate.
- CP-related complications require their own specific therapy as appropriate, e.g., atrial arrhythmias.
- In general, beta-blockers and calcium channel blockers should be avoided, because the sinus tachycardia that commonly occurs in constriction has a compensatory function, maintaining cardiac output in the setting of fixed stroke volume (secondary to fixed diastolic filling).
- Patients with advanced cardiac symptoms related to constriction may derive little benefit from surgery, as these are often the high surgical risk group.
Pericardiectomy
Pericardiectomy was first performed and reported in 1913, and is regarded as a curative measure for CP [20,22]. Although partial pericardiectomy has been recommended in some patients, complete pericardiectomy remains the definitive therapy and a potential cure in most CP patients [17,19]. If pericardiectomy is performed earlier in the course of CP, when the pericardium is less calcified and no element of myocardium fibrosis/dysfunction or advanced heart failure is evidenced, the outcomes are generally acceptably good. However, some judgement must be exercised because patients who are in NYHA Class I or early NYHA Class II may be clinically stable for many years. Pericardiectomy may not be recommended very early on during the course of the disease or in patients with advanced disease (Class IV symptoms), when the surgical risk is high. The surgical risk in patients with advanced diseases or those with LV systolic dysfunction is very high, with an estimated 40% to 60% perioperative mortality rate [2].
Partial pericardiectomy or less invasive options (i.e., pericardial window) should be considered whenever fluid reaccumulates, becomes loculated or biopsy material is required in the setting of a large or recurrent pericardial effusion, particularly in effusive constriction. This approach should also be considered in patients with advanced disease, myocardial dysfunction or anticipated heart failure, particularly in those at high risk of acute ventricular dilatation after surgery, with subsequent acute heart failure (right ventricular failure [RVF]).
Pericardiectomy can be a long, horrendous and often technically complex procedure, requiring extensive surgical experience. The two recommended standard approaches are via an anterolateral thoracotomy and median sternotomy [2,19,20]. Pericardial decortication should be as extensive as possible, especially at the diaphragmatic-ventricular contact regions. An excimer laser is recommended and should be considered, particularly when severe adhesions occur between the pericardium and epicardium to prevent perioperative mediastinal bleeding and maintain haemodynamic stability throughout the surgical procedure. Complications related to pericardiectomy may include excessive bleeding, atrial and ventricular arrhythmias, ventricular wall ruptures, phrenic nerve injury and even CP recurrence.
In the recent past, published reports have indicated that the surgical mortality is estimated to range between 5 and 15%, with one of the reports indicating a 30-day perioperative mortality of 6.1% [2,7,16]. Furthermore, other studies have demonstrated a perioperative mortality of between 5% and 10%, and a further increased later mortality rate of up to 70% in the same study population; however, these poorer outcomes were also exacerbated by other comorbidity factors [2,16]. Generally, poor prognostic markers associated with these detrimental outcomes include progressive heart failure or advanced heart failure symptoms (NYHA III-IV), sepsis, renal failure, respiratory failure, arrhythmia, prior mediastinal radiotherapy as the primary cause of constriction, and other concomitant severe comorbidities [7]. Reports have indicated that an estimated 80% to 90% of CP patients who undergo a successful pericardiectomy, particularly in the hands of extensive expertise, achieve significant symptomatic improvement to NYHA Class I or II postoperatively and live potentially normal or near normal lives afterwards [9].
Although symptomatic improvement is evident after pericardiectomy, evidence of abnormal diastolic filling patterns often persist for months. These can be correlated with symptoms or clinical status [3]. Furthermore, reports have indicated that a significant number of patients, in excess of 60% of CP patients, demonstrate complete normalisation or resolution of cardiac haemodynamics after pericardiectomy [2,7,16]. Although some patients improve with time, persistent diastolic filling abnormalities tended to occur in those who had a longer history of preoperative symptoms, supporting the view that early operation is advisable in symptomatic patients [3]. Those patients who have symptoms that persist even after successful pericardiectomy may have a mixed constrictive-restrictive picture or so-called constrictive cardiomyopathy, which warrants thorough clinical and imaging modalities including advanced echocardiographic parameters, cardiac computed tomography scan or even CMR [7,16].
One study reported 58 CP patients who underwent total pericardiectomy and were then followed up for a total of four to five years after surgery [1]. About 30% of these patients had persistently severe heart failure symptoms during the follow-up period after pericardiectomy; some had persistent evidence of a restrictive or constrictive pattern [2,7,14,16]. The overall assumption was that the persistent symptoms could be related to the restrictive or constrictive changes demonstrated in most of their Doppler signals [2,7,14,16].
Additional prognostic parameters
Cardiac morbidity and mortality seem to be related to preoperative myocardial atrophy or fibrosis, which can be detected by means of advanced cardiac imaging modalities including computed tomography. This prolonged myocardial atrophy or fibrosis and reduced ventricular mobility, particularly the RV free wall, are associated with acute RV dilatation related to subsequent increased preload to the RV, to increased venous return which then leads to acute RV failure and decreased cardiac output, function and subsequent death. Inadequate responses after surgery could also be attributed to the chronicity of constriction which is associated with prolonged myocardial immobility due to longstanding disease. Apart from myocardial atrophy or fibrosis, other important factors associated with prolonged recovery after pericardiectomy include incomplete decortication, and also recurrent constriction which could lead to prolonged cardiac compression related to persistent mediastinal inflammation and fibrosis. Although there is controversy regarding the impact of pericardial calcification on perioperative or post-surgical survival, some reports indicate that extensive pericardial calcification might have prognostic implications. Most importantly, these calcifications could extend into the myocardium, making pericardiectomy more extensive and complex, leading to perioperative bleeding, haemodynamic instability and subsequent death [7,16]. Excluding patients with pericardial calcifications from studies reduces the reported mortality rate to the lower end of the spectrum.
Precautionary measures
Postoperatively, low cardiac output may occur in patients who are severely symptomatic due to advanced disease or being debilitated and also in those who have ascites or other findings of fluid retention. Patients with a low cardiac output state need stringent perioperative measures and an extensively experienced surgeon to avoid imminent death. These types of patients may require maintenance of high left atrial pressure, sympathomimetic infusions, or both to maintain their cardiac output during this critical period. Mechanical support of the circulatory system, such as extracorporeal membrane oxygenation, intra-aortic balloon counterpulsation, or an LV or RV assistance device system should be used in patients who are critically ill. Acute RV failure should be anticipated and adequate measures in the form of supportive parameters should be in place to avoid early mortality after pericardiectomy.
Surgical approaches
First and foremost, different surgical approaches and techniques have been the subject of long debate which includes mediastinal versus lateral thoracotomy, and partial versus complete pericardiectomy [19,20,22]. In simple terms the median sternotomy enables a more radical pericardial removal along the right pericardium and venae cavae; this could be horrendous when using the lateral approach. The left anterolateral approach is generally preferred in patients with purulent pericarditis and effusive CP as a precautionary measure to avoid pyothorax and risk of sternal infection [19,20,22].
Secondly, cardiopulmonary bypass (CPB) as an adjunctive measure has been extensively debated [19,20,22]. CPB is recommended in CP patients with prior cardiac injuries or haemodynamic instability related to significant mediastinal bleeding, patients with previous pericardial manipulation or any form of cardiac surgery, those with extensive pericardial calcifications, or with a concomitant cardiac or cardiovascular surgical procedure [2,19,20].
Conclusion
The primary causes of CP may be identifiable and treated in some cases which could be helpful to prevent recurrences or development of constriction later on; however, in some cases CP may not be preventable. Pericardiectomy remains the only definitive treatment for constrictive pericarditis. Pericardiectomy, particularly complete pericardiectomy, is indicated once the diagnosis of CP has been confirmed. Most importantly, pericardiectomy is recommended early on during the course of constriction while patients still have a favourable functional status, as this approach is associated with favourable outcomes both in the short and long term. Medical therapy should be directed to the primary aetiology of constriction, where feasible. An identifiable cause should be managed accordingly.