Causes
The pericardium is a double-walled sac containing the heart and the roots of the great vessels and is composed of a visceral and parietal component. The pericardial space enclosed between the two serosal layers normally contains up to 50 mL of serous fluid. It provides lubrication and protection from infection. Pericardial diseases may be isolated or part of a systemic disease. Cardiac tamponade is a life-threatening condition due to slow or rapid pericardial accumulation of fluid with subsequent compression of the heart [1].
The causes of pericardial fluid accumulation leading to cardiac tamponade are idiopathic, infectious, immune-inflammatory, neoplastic disease, post-cardiac surgery, trauma, renal failure, aortic dissection and miscellaneous (chronic renal failure, thyroid disease, amyloidosis) [2-4]. The most common causes of tamponade are pericarditis (infection and non-infection), iatrogenic (cardiac invasive procedures and post-surgery), and malignancy [5]. More rare causes are collagen diseases (systemic lupus erythematosus, rheumatoid arthritis, scleroderma), radiation, aortic dissection, uraemia, post-myocardial infarction and bacterial infection [2]. Causes of effusion with a high incidence of progression to tamponade include bacterial, fungal, human immunodeficiency virus-associated infections, bleeding, and cancer involvement [3]. For all patients, infectious diseases are still the most common cause of pericardial tamponade but, due to an increasing number of cardiac interventional procedures (cardiac ablation, device lead implantation and percutaneous coronary intervention), the incidence of haemopericardium seems to be increasing.
Signs
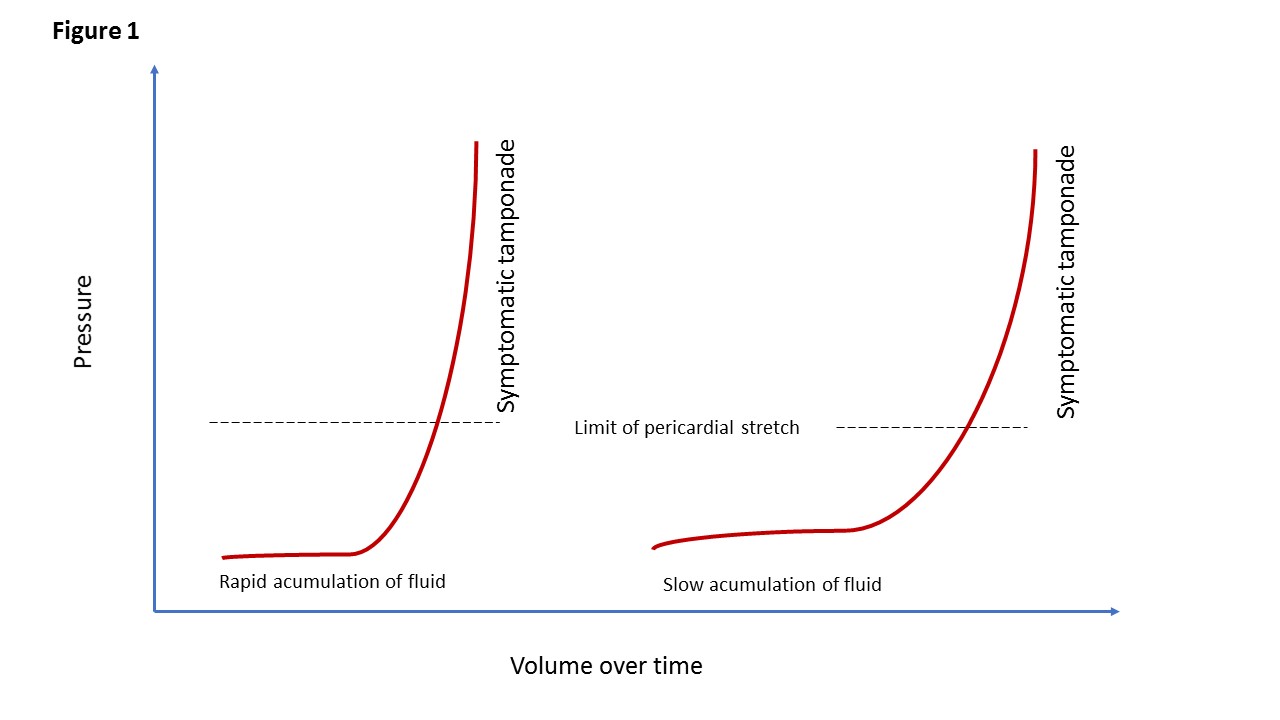
The key element which determines the clinical presentation is the rate of fluid accumulation relative to pericardial stretch and the effectiveness of compensatory mechanisms. Thus, cardiac tamponade comprises a continuum from an effusion causing minimal effects to one causing circulatory collapse. The stiffness of the pericardium determines fluid increments precipitating tamponade [6,7]. The pericardial strain-stress curve is depicted in Figure 1. There is a slow phase and a steep rise, leading to critical cardiac compression. Thus, rapid accumulation of as little as 150 mL of fluid can result in a marked increase in pericardial pressure and can severely impede cardiac output. In contrast, 1,000 mL of fluid may accumulate over a longer period without any significant effect on diastolic filling of the heart. This is due to adaptive stretching of the pericardium over time. A compliant pericardium can allow considerable fluid accumulation over time without haemodynamic compromise.
Figure 1. Cardiac tamponade.
Pericardial strain-stress curves illustrating that the pericardium has some degree of elasticity. When the elastic limit is reached the intrapericardial pressure rises. The left-hand panel shows the pressure-volume curve with rapidly increasing pericardial fluid whereas the right-hand panel shows a slower rate of pericardial effusion.
Several signs may be present during examination depending on the time of fluid accumulation. The classic signs in cardiac tamponade are included in Beck’s triad of hypotension, jugular venous distension, and muffled heart sounds. Other clinical signs in a patient with cardiac tamponade include tachycardia, pulsus paradoxus, decreased electrocardiographic voltage with electrical alternans and an enlarged cardiac silhouette on chest X-ray with slow-accumulating effusions [8-10].
The pathophysiological and haemodynamic explanation for the above-mentioned findings is as follows.
During fluid accumulation, left- and right-sided atrial and ventricular diastolic pressures rise, and equalise the pressure similar to the pericardial sac (20-25 mmHg). The equalisation is closest during inspiration. Thus, pericardial pressure dictates intracavitary pressure leading to a progressive decline in cardiac volumes. The decreased preload accounts for the reduced stroke volume and compensatory increased contractility, and tachycardia is not enough to maintain stroke volume, thus leading to reduced cardiac output. Since the filling pressure in the right side of the heart is lower than in the left side of the heart, filling pressure increases more rapidly in the right than in the left side of the heart. Pulsus paradoxus is an abnormal decline (>10 mmHg) in systemic arterial pressure during inspiration. Normally, the intrathoracic pressure decreases during inspiration which allows blood to flow easily into the right heart. Conversely, the left heart filling decreases during inspiration, as the intrapericardial volume is fixed. During expiration, the intrathoracic pressure increases which leads to less right heart filling and augments filling of the left heart chambers. When fluid accumulates in the pericardial space, the intrapericardial pressure increases. This leads to a compression of the right heart, increasing the right heart pressure. Thus, the right heart filling is now relying more heavily on the decreased intrathoracic pressures during inspiration to fill, exaggerating the blood pressure change. The interventricular septum shifts to the left during inspiration and encroaches on the left ventricle, leading to a further reduction in stroke volume of the left ventricle [11].
The underlying process for the development of tamponade is a marked reduction in diastolic filling, which results when transmural distending pressures become insufficient to overcome increased intrapericardial pressures. Tachycardia is the initial cardiac response to these changes to maintain the cardiac output.
Systemic venous return is also altered during tamponade. Because the heart is compressed throughout the cardiac cycle due to the increased intrapericardial pressure, systemic venous return is impaired and right atrial and right ventricular collapse occurs. Because the pulmonary vascular bed is a vast and compliant circuit, blood preferentially accumulates in the venous circulation, at the expense of left ventricular filling. This results in reduced cardiac output and venous return.
The amount of pericardial fluid needed to impair diastolic filling of the heart depends on the rate of fluid accumulation and the compliance of the pericardium.
Small effusions are seen posteriorly and are typically less than 10 mm in thickness (pericardial pressure <10 mmHg). Moderate effusions tend to be posterior and may be circumferential and are usually 10-20 mm in thickness (pericardial pressure <10 mmHg), whereas large effusions tend to be circumferential and greater than 20 mm in thickness (pericardial pressure >15 mmHg).
Symptoms
The symptoms of cardiac tamponade vary with the length of time over which pericardial fluid accumulates. As depicted in Figure 1, a rapid accumulation of fluid in the pericardium quickly leads to a steep rise in pericardial pressure, whereas a slower accumulation of fluid takes longer to reach critical or symptomatic pericardial pressure [12,13]. Thus, the haemodynamic impact of an effusion ranges from none or mild to cardiogenic shock which leads to a clinical presentation ranging from acute to subacute. Acute or rapid cardiac tamponade is a form of cardiogenic shock and occurs within minutes. The symptoms are sudden onset of cardiovascular collapse and may be associated with chest pain, tachypnoea, and dyspnoea. The decline in cardiac output leads to hypotension and cool extremities. The jugular venous pressure rises which may show as venous distension at the neck and head. Acute cardiac tamponade is usually caused by bleeding due to trauma, aortic dissection or is iatrogenic.
Chronic fluid accumulation or subacute cardiac tamponade is characterised by the patients being more asymptomatic in the early phase but, when the pressure rises above the pericardial stretch point (Figure 1), they complain of dyspnoea, chest discomfort, peripheral oedema, fatigue, or tiredness, all symptoms attributable to increased pericardial pressure and limited cardiac output.
Diagnosis
Prompt diagnosis is the key to reducing the mortality risk for patients with cardiac tamponade. Although cardiac tamponade is a clinical diagnosis, echocardiography (Figure 2) provides useful information and is the cornerstone during evaluation (availability, bedside, and treatment). However, cardiac tamponade is associated with a variety of abnormalities that lead to changes on the electrocardiogram (ECG), chest X-ray, and on echocardiography. Abnormalities of tamponade on the ECG are typically low voltage and electrical alternans. However, reduced voltage can also be seen among other conditions such as infiltrative myocardial disease and emphysema, whereas electrical alternans characterised by beat to beat alterations in the QRS complex caused by swinging of the heart is specific, but not sensitive for tamponade. The chest X-ray reveals a normal cardiac silhouette until the effusions are at least moderate in size (~200 mL). In general, an enlarged cardiac silhouette is neither sensitive nor specific for the diagnosis of cardiac tamponade.
Echocardiographic techniques remain the standard non-invasive method to establish the diagnosis and can be used to visualise ventricular and atrial compression abnormalities as blood cycles through the heart [14]. An effusion appears as a transparent separation between the parietal and visceral pericardium during the cardiac cycle. Physiologic pericardial fluid may only be visible during ventricular systole, whereas effusions exceeding 75-100 mL are visualised throughout the cardiac cycle.
Figure 2. Two-dimensional echocardiogram illustrating cardiac tamponade with right atrium collapse or indentation (arrow).
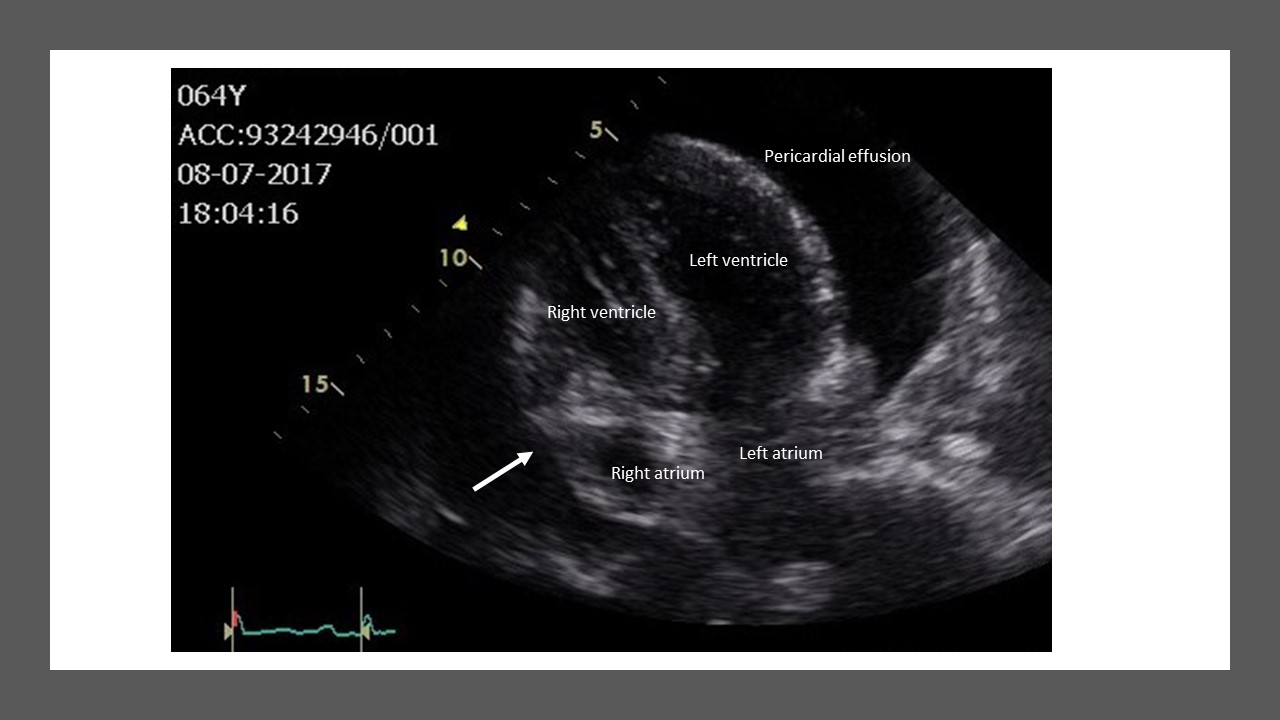
The following may be observed with two-dimensional echocardiography:-
1) Early diastolic collapse of the right ventricular free wall
2) Late diastolic compression/collapse of the right atrium
3) Swinging of the heart in the pericardial sac
4) Dilated inferior vena cava with minimal or no collapse with inspiration
5) A greater than 40% relative inspiratory augmentation of blood flow across the tricuspid valve
6) A greater than 25% relative decrease in inspiratory flow across the mitral valve
7) Septal bounce into the left ventricle during inspiration
Transoesophageal echocardiography is also useful in patients where transthoracic echocardiography is non-diagnostic. It is typically used in post-cardiac surgery patients suspected of having loculated effusions containing clots. Additional imaging with magnetic resonance or computed tomography is normally not necessary to diagnose cardiac tamponade, but can be used as second-line imaging in cases of complex or loculated effusions and evaluation of associated or extracardiac diseases or findings. Depending on the underlying cause of the pericardial fluid, lab test and analysis of the pericardial fluid can supply diagnostics. However, most effusions are transudate and do not yield the underlying cause, but it is good practice to analyse the pericardial fluid for white blood cell count, haematocrit, malignant cells and protein content [14,15].
Treatment
When tamponade is present or threatened, clinical decision making requires urgency, and the threshold for pericardiocentesis should be low. The treatment of cardiac tamponade is drainage, preferably by needle paracentesis with the use of echocardiographic or another type of imaging, such as fluoroscopy [14,16]. The needle tip is evident on imaging, and imaging can thus be used to identify the optimal point to perform the centesis [17]. Imaging guidance allows the operator to select the shortest and safest route to the effusion. Open surgical drainage is normally not necessary, but based upon local preference and experience, and is desirable if intrapericardial bleeding is present, when there is a clotted pericardium and if needle centesis is difficult or ineffective. Treatment should be individualised, and thoughtful clinical judgement is essential. Patients with large effusions with minimal or no evidence of haemodynamic compromise may be treated conservatively with careful follow-up and monitoring, and therapy aimed towards the underlying cause. Thus, patients with apparently idiopathic pericarditis and mild tamponade could be treated for a period with non-steroidal anti-inflammatory drugs (NSAID) and colchicine in the hope that the effusion will shrink. The same approach or strategy could be performed in patients with connective tissue or inflammatory diseases. Unfortunately, there are no proven effective medical therapies to reduce an isolated effusion. In the absence of inflammation, NSAID, colchicine and corticosteroids are generally not effective [14]. Pericardiocentesis alone may be necessary for the resolution of large effusions, but recurrences are also common, and surgical pericardiectomy or less invasive options (i.e., pericardial window) should be considered whenever fluid reaccumulates, becomes loculated, coagulopathy is present, or biopsy material is required [14]. Loculated effusions due to bleeding are difficult to drain sufficiently with a closed approach, whereas surgical drainage affords the opportunity to correct the source of the bleeding.
Once tamponade is diagnosed, management should be orientated toward urgent pericardiocentesis. Preparing the pericardiocentesis, intravenous hydration and positive inotropes can be used temporarily, but should not be allowed to substitute for or delay pericardiocentesis. The risk and benefits of needle centesis should be considered in patients with anticoagulation therapy or if coagulopathy is present. Mechanical ventilation should be avoided due to a further decrease in cardiac output [16]. In case of cardiac arrest due to tamponade, external cardiac compression has limited or no value because there is little room for additional filling [18].
A triage system has been proposed by the ESC Working Group on Myocardial and Pericardial Diseases in order to guide the timing of the intervention and the possibility of transferring the patient to a referral centre [1]. This triage system is essentially based on expert consensus and requires additional validation in order to be recommended in clinical practice.




 Our mission: To reduce the burden of cardiovascular disease.
Our mission: To reduce the burden of cardiovascular disease.