Introduction
A precise evaluation of the severity of aortic valve stenosis (AS) is crucial for patient management and risk stratification, and to allocate symptoms legitimately to the valvular disease. Echocardiography is the main method to assess AS severity. It relies on three parameters, namely the peak velocity (PVel), the mean pressure gradient (MPG) and the aortic valve area (AVA). The first two parameters are directly measured using continuous wave Doppler, while the last one is calculated based on the continuity equation and measurement of the left ventricular outflow tract (LVOT) diameter, LVOT time-velocity integral (TVI) and aortic TVI. Ideally, these parameters should be concordant, with severe AS being defined by a peak velocity >4 m/sec, an MPG >40 mmHg and an AVA <1 cm2 (Table 1). The last decade has seen this apparently easy and straightforward classification shaken up by the observation that up to one-third of patients present with discordant AS grading, and by the identification of a subset with paradoxical low-flow, low-gradient severe aortic stenosis despite preserved ejection fraction. In the present paper, we present pitfalls that should be avoided to ensure that the patient truly presents with discordant grading, we assess the prevalence and outcome of this entity, and finally we highlight the importance of computed tomography to assess AS severity independently. We will not discuss the assessment of AS severity in patients with depressed ejection, but will focus on patients with normal/preserved ejection fraction.
Table 1. Aortic Stenosis Grades of Severity as Assessed Using Echocardiography and Computed Tomography (calcium scoring)
Echo Parameters for Aortic Stenosis (AS):
- Sclerosis
- Mild AS
- Moderate AS
- Severe AS
Peak velocity (m/sec):
- Sclerosis: < 2.5
- Mild AS: 2.5–3
- Moderate AS: 3–4
- Severe AS: > 4
Mean gradient (mmHg):
- Sclerosis: Normal
- Mild AS: < 20
- Moderate AS: 20–40
- Severe AS: > 40
Aortic Valve Area (AVA, cm²):
- Sclerosis: Normal
- Mild AS: ≥ 1.5
- Moderate AS: 1–1.5
- Severe AS: < 1
Calcium scoring (Agatston Units, AU):
- Male: 2,065
- Female: 1,275
Pitfalls in the evaluation of aortic valve stenosis
Discordant grading is defined based upon the observation that one parameter suggests a moderate AS while the other suggests a severe AS. Discordant grading is defined either by an AVA <1 cm2 while MPG is ≥40 mmHg/PVel <4 m/sec, or by an AVA ≥1 cm2 and an MPG ≥40 mmHg/PVel ≥4 m/sec, the first situation being much more common. MPG and PVel are highly correlated (collinear) and can be used almost interchangeably. Thus, in the rest of the article we will use the MPG.
Calculation of the AVA relies on the measurement of three parameters; error measurement may occur in all three. Measurement of LVOT diameter is probably the main source of error for the calculation of the AVA. LVOT diameter should be measured in the parasternal long-axis view, using the zoom mode, in mid systole and repeated at least three to five times. There is still ongoing debate as to whether the LVOT diameter should be measured at the level of leaflet insertion – i.e. aortic annulus – or more apically, i.e. 5 to 10 mm below the annulus. The recent recommendation on echocardiographic assessment of AS from the European Association of Cardiovascular Imaging and the American Society of Echocardiography [1] does not provide a definite answer, but underlines the fact that measurement of the LVOT at the annulus level provides higher measurement reproducibility and ensures that diameter and pulse Doppler are measured at the same anatomical level. This is our usual practice and our personal recommendation. It is critical to underline that a 1 mm change in measurement of the LVOT diameter results in 0.1 cm2 difference in AVA calculation. Severe calcification and poor echogenicity are important challenges to measure the LVOT diameter accurately. LVOT, as with any anatomic structure, is correlated to body size. We previously established a safeguard formula using the body surface area (BSA) (theoretical LVOT diameter = 5.7*BSA + 12.1). [2] The standard deviation was 1 mm, meaning that 50% of the patients were 1 mm above or below this theoretical value and that 95% of patients were 2 mm above or below.
The second source of error is the measurement of the aortic valve TVI obtained using continuous Doppler. PVel and MPG are obtained on the same image acquisition. With the improvement in echocardiographic systems and combined two-dimensional/Doppler probe, the crystal probe tends to be disused and may appear outdated. However, even using the most recent materials, it is crucial to record the highest aortic velocity in multiple incidences, namely the apical view but also the right parasternal view, the suprasternal view and the subcostal view. Not using other views leads to the underestimation of AS severity in 20% or more of patients. [3] If the crystal probe is unavailable, the regular two-dimensional probe can be used in the right parasternal view, providing similar results to the crystal probe in our experience.
The importance of the third parameter, the LVOT TVI, is often underestimated. If the Doppler sample is positioned too far from the aortic orifice, it will be responsible for an overestimation of AS severity. In contrast, if positioned too close, within the flow acceleration, it will be responsible for an underestimation of AS severity. The recommendation is to move the Doppler sample up and down in order to obtain a nice Doppler trace with a closure click (possibly missing in very severe AS) without the opening click.
Finally, an AVA below 1 cm2 may also be observed in small-sized patients. In these circumstances, AVA should be adjusted for BSA, with the threshold being 0.6 cm2/m2. It is important to keep in mind that BSA correction should be only undertaken in patients with small and large stature (small, elderly lady or male, professional basketball player), and should be avoided in those who are obese. Thresholds adjusted to height are currently missing.
Incidence and prognosis of discordant AS grading
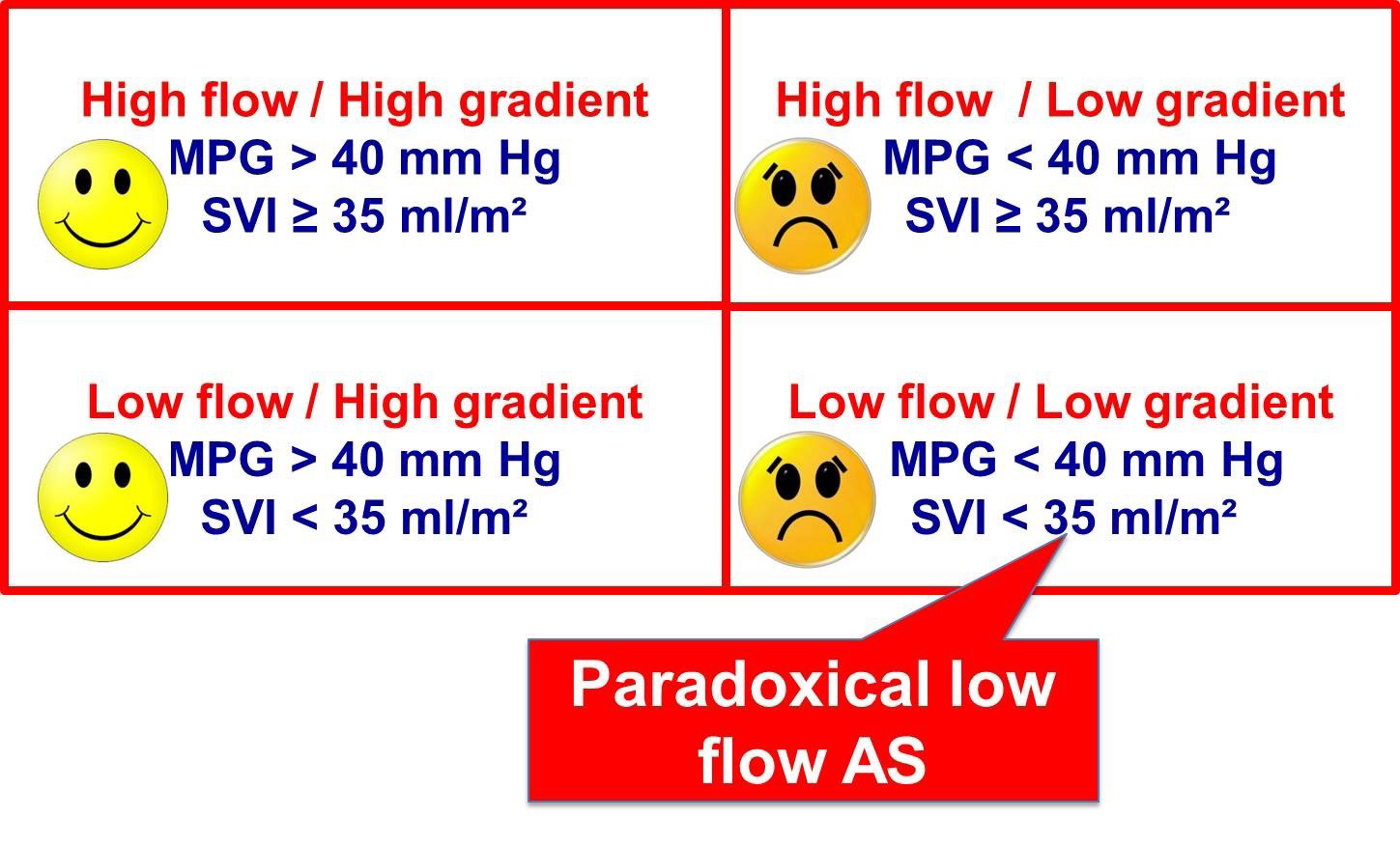
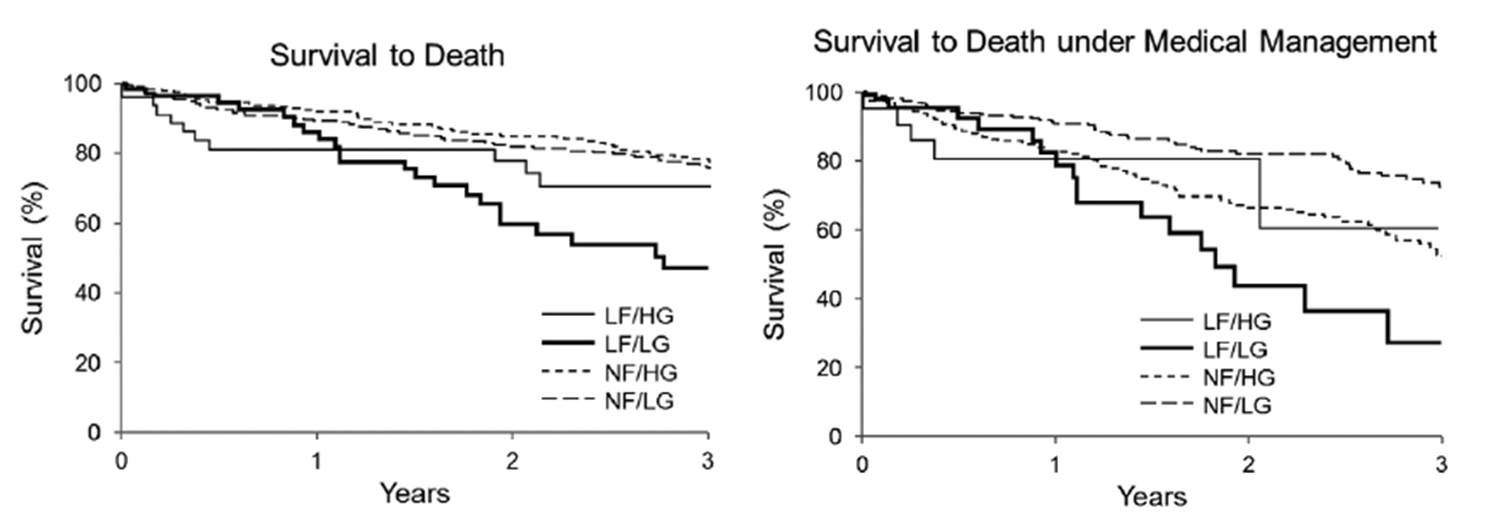
Fulfilling the precise and rigorous methodology presented above, the rate of patients with discordant grading is still between 20% and 30%, thus representing a common clinical problem. Uncertainties regarding incidence and outcome of these patients are the consequence of the use of a different nosology between papers and possibly error measurements. Thus, in the seminal paper from the Quebec team [4], the criterion used to differentiate groups was the stroke volume index. The proposed threshold of 35 ml/m2 is now widely accepted, even if its validation has never been carried out properly. In contrast, in the SEAS trial [5], the authors considered the discordance between AVA and MPG independently of any flow consideration. The most appropriate way of classifying patients is first to consider whether AVA and MPG are concordant, and secondly to consider the flow (stroke volume index). Thus, among patients with an AVA below 1 cm2, four groups can be identified (Figure 1). Patients on the left part of the figure are easily classified as severe AS, whereas rest echocardiography remains inconclusive in the other two groups, namely patients with low gradient and normal or low flow. The latter group is close to the low flow paradoxical severe AS described by the Quebec team. [4] The Mayo Clinic group has provided us with important data regarding the prevalence of the different subsets. [6] Among 1,704 patients with a valve area below 1 cm2, 24% presented with discordant grading (AVA <1 cm2 and MPG <40 mmHg). In the vast majority (21% of the overall population), the flow was normal, while low flow was observed in only 3% of the total population. This study confirms the high prevalence of patients with discordant grading and also shows that most often these patients presented with normal flow. Importantly, this study also showed that the subset of patients with discordant grading (AVA <1 cm2, MPG <40 mmHg) and a low flow had the worst prognosis (Figure 2). Our understanding of the literature is that flow is a prognostic factor, whatever the reason or the cause of the depressed flow. The typical phenotype initially proposed of an old lady often in AF with preserved ejection fraction but important left ventricular hypertrophy responsible for the low flow is thus more the exception than the rule. Of note, the rare cases of discordant grading with an AVA >1 cm2 and an MPG >40 mmHg are often observed in patients with a bicuspid aortic valve and a large LVOT/annulus size. They are usually classified as having severe AS.
Figure 1. Classification of Patients with an Aortic Valve Area <1 cm2 (and preserved ejection fraction) into Four Groups according to Mean Pressure Gradient (MPG) and Stroke Volume Index (SVI)
Figure 2. Prognosis of the Four Subsets as Defined in Figure 1. The Patients with Low Flow (stroke volume index <35 ml/m2) and Low Gradient (<40 mmHg) Incurred the Worst Prognosis (from reference [6])
Use of computed tomography in the assessment of AS severity
The fact that discordant grading is common and that low flow is rare but impacts on prognosis is of no help in assessing whether these patients truly presented severe AS. One main debate of recent years in the domain of valvular heart disease has, indeed, been whether these patients with discordant grading should be managed according to the valve area (thus as severe AS) or according to MPG (usually moderate AS). Flow consideration has added a supplementary level of confusion. As resting echocardiography is inconclusive, it requires the use of additional methods.
With the use of computed tomography in the workup evaluation before TAVI, the anatomy of the aortic annulus has been well described. More specifically, CT has clearly demonstrated that the LVOT and the aortic annulus are not circular but oval. For the calculation of the AVA, a diameter is measured and the LVOT area calculated assuming that the LVOT is circular, introducing an obvious error. This is why some have suggested combining CT (for the measurement of the LVOT area) and echocardiography for LVOT and aortic TVI in the calculation of the AVA. [7] Although attractive, such methodology suffers from important bias. First, it is well established that echocardiography underestimates the measurement of the LVOT annulus by 1 to 2 millimetres. Thus, it is expected that the AVA will increase and the number of patients with MPG <40 mmHg and AVA <1 cm2 will mathematically decrease. Second, the prognostic value of the AVA has been established using echocardiographic evaluation, while the prognostic value of combined AVA calculation is uncertain. Third, in no study combining CT measurement of the LVOT area was a reference (if not a gold standard) method used.
Therefore, the best way to address this issue is to use a quantitative and reliable flow-independent method for the assessment of AS severity, which is the remarkable characteristic of calcium scoring. Aortic valve calcification is the leading process of AS. [8] In contrast to what is observed in the vasculature, hydroxyapatite deposition and leaflet infiltration are the main mechanisms for leaflet restriction and haemodynamic obstruction. The degree of aortic valve calcification can be quantitatively and accurately assessed in vivo using computed tomography. [9] The methodology is simple and widely available. Gated computed tomography is performed from the apex to the base of the heart, including the aortic valve. There is no need for contrast injection. Using semi-automatic software, areas that are considered as calcification (defined by a tissue density >130 Hounsfield units) are highlighted in red. The operator 'just' has to select the area that is considered as belonging to the aortic valve. Avoiding simple pitfalls such as mitral annular, aortic wall and coronary ostia calcifications, the method is highly reproducible. Quantification is performed based on the Agatston score (expressed in arbitrary units [AU]) which rely on the area of calcification and of peak density. We have shown that calcium scoring is highly correlated to echocardiographic haemodynamic severity and have validated its diagnostic value for the diagnosis of severe AS. [10] Interestingly, thresholds for severe AS were different between females and males. [11] For the same degree of aortic valve calcification, females experienced a higher haemodynamic obstruction or, put another way, a mean gradient of 40 mmHg is associated with a lower calcium load in females than in males. In addition to the fact that thresholds are different in males and females (approximately 2,000 and 1,250 AU, respectively), these results show that AS pathophysiology is different in males and females and, indeed, female leaflets are more fibrotic than those of males. [12] Importantly, these thresholds are not valid for rheumatic disease and deserve specific validation in the bicuspid aortic valve. We have used this methodology in 646 patients with moderate/severe AS and normal ejection fraction. [13] Confirming the findings of other papers, a discordant grading (AVA <1 cm2 and MPG <40 mmHg) was observed in 27% of the population; most of them (85%) presented with normal flow. As expected, computed tomography and calcium scoring accurately classified patients with concordant grading, but more importantly 50% of the patients with discordant grading could be considered as having true severe AS, whereas 50% did not fulfil the criteria for severe AS, irrespective of flow calculation.
Calcium scoring measurements and the above-mentioned thresholds have recently been implemented in the latest version of the ESC/EACTS guidelines on valvular heart disease. [14] In case of discordant grading, after verification of potential error measurements, calcium scoring should be performed as the first-line test. If the diagnosis of severe AS is established (and if the patient is symptomatic), intervention should be promptly considered. If calcium scoring is below the threshold, AS is more likely to be non-severe and probably conservatively managed, although whether an intervention may provide a benefit still needs to be evaluated. It is also worth noting that the proposed thresholds are not 'magic numbers', but provide a probability of having or not having severe AS. Thus, a woman with a score of 3,000 is very likely to present with severe AS, whereas a man with a score of 700 is very unlikely to present with severe AS.
Take-home message
The important points discussed in the present paper can be summarised as follows:
Discordant grading is common in clinical practice.
The first step is to look for error measurements.
Adjust for BSA in patients with extreme body size (but this should be avoided in obese patients).
Among patients with discordant grading (AVA <1 cm2 and MPG <40 mmHg), those with low flow are much less frequent than those with normal flow.
Flow does not provide any diagnostic information regarding AS severity, but provides prognostic information.
Transthoracic echocardiography cannot help you solve the problem of AS severity in most cases of discordant grading.
Aortic valve calcium scoring is a quantitative and flow-independent method of assessing AS severity (recommended thresholds are 2,000 in men and 1,250 in women).
Intervention is recommended in symptomatic patients with proven severe AS, as in classic severe AS.