Introduction
Transcatheter aortic valve implantation (TAVI) is now established as the treatment of choice for patients presenting with severe symptomatic aortic stenosis (AS) who are deemed inoperable or high surgical risk [1]. Within this manuscript, we consider the technical aspects of TAVI that should be considered in order to optimise clinical outcomes once a patient has been identified by the “Heart Team” as suitable for TAVI following clinical assessment and pre-procedural investigations. We highlight the limitations of current devices that should be taken into account when performing TAVI to optimise both procedural and longer-term clinical outcomes.
Environment
Greater operator and institutional experience coupled with technological advances have reduced procedure times and complications. However, TAVI remains a complex procedure in an elderly and frail population. As a consequence, the current European Society of Cardiology (ESC) guidelines [1] advise that all TAVI procedures should be carried out in hospitals with on-site cardiothoracic surgery to facilitate immediate access to the necessary expertise if required. Whilst this is imperative for procedures that are carried out via the transapical or other surgical access sites (see subsection on vascular access), availability of surgical expertise is also critical to manage complications that require emergency surgery (which can occur in up to 5% of all cases).
Anaesthesia
General anaesthesia (GA) is required for surgical access sites (e.g., transapical or transaortic). However, improvements in pre-procedural assessment (particularly using advanced imaging of the aorta and peripheral vasculature using computed tomography [CT] and echocardiography) and engineering advances in prosthesis delivery systems (e.g. smaller delivery sheaths) have enabled the increasing use of conscious sedation and local/regional anaesthesia. Whilst no randomized studies have been conducted to ascertain if these are superior to GA, the advantages include shorter procedure times, the elimination of risks associated with GA and faster patient recovery [2].
Vascular access
Transfemoral approach
If the peripheral vasculature is suitable, then the transfemoral vascular approach has a number of advantages, including shorter procedure times, faster patient recovery and superior outcomes when compared to the transapical approach (although outcomes are confounded by differences in baseline patient characteristics) [3], and is now the default access site. Furthermore, whilst initial TAVI devices required the use of large delivery sheaths that required surgical cut-down, new smaller sheaths have now enabled the procedure to become truly “percutaneous”. Other access sites should be considered when the transfemoral route is not thought to be suitable (e.g., minimal luminal diameter <5.5 mm, excessive tortuosity).
Transapical approach
This approach requires a small left thoracotomy and direct puncture of the left ventricular apex and avoids the requirement for large catheters to be advanced through the iliofemoral system and aortic arch. The main disadvantage is the requirement for general anaesthesia, a greater degree of myocardial injury and longer recovery times. “Percutaneous” transapical procedures are currently being evaluated with the advent of ventricular closure devices; however, these techniques are still in their infancy and require further clinical evaluation prior to wider adoption.
Transaortic approach
The use of the transaortic approach through a mini-sternotomy or right thoracotomy not only avoids the passage of large catheters through the iliofemoral system and aortic arch but also puncture of the left ventricle. Furthermore, since aortic cannulation is a routinely performed procedure this access route is potentially appealing to cardiac surgeons.
Other vascular access options with demonstrated clinical efficacy in the setting of TAVI include the transsubclavian, transaxillary and transcaval approaches for patients with anatomy that is unsuitable for the more traditional techniques.
Prosthesis selection
Whilst initial TAVI experiences were limited to a small number of devices, the rapid adoption of TAVI has generated a major impetus to refine and improve available devices, resulting in a wide selection now being available (Table 1). The principal advantages of the newer devices include the requirement for small delivery sheaths, the ability to reposition (or even completely recapture) the device should suboptimal positioning be an issue, and specific designs to reduce the frequency and severity of residual aortic regurgitation.
Data directly comparing individual device designs and specific prostheses are limited, and the choice of device is currently dependent on patient-specific anatomy (e.g., coronary height, dimensions of peripheral vasculature), operator preference and centre expertise. The CHOICE (A Comparison of Transcatheter Heart Valves in High Risk Patients with Severe Aortic Stenosis) trial [4] is the only randomized study comparing the balloon-expandable (BE) Edwards SAPIEN XT valve (Edwards Lifesciences, Irvine, CA, USA) and the self-expanding (SE) Medtronic CoreValve (Medtronic, Minneapolis, MN, USA). BE valve implantation was associated with a higher rate of success (p>0.001), a lower frequency of residual moderate aortic regurgitation (p<0.001) and a lower requirement for permanent pacing (p=0.001), but a slightly higher mean transvalvular gradient (p<0.001) and a trend towards more frequent stroke and coronary occlusion. Mortality at 30 days and 1 year were comparable between groups. However, a randomized comparison of contemporary devices is lacking.
Prosthesis implantation
When the transfemoral route is chosen, a “crossover” technique (involving a wire placed in the contralateral artery to allow delivery of a balloon or stent to treat the access vessel in case of injury) is usually performed to “protect” the artery in case of vascular complication [5].
A temporary pacing wire (TPW) is positioned in the right ventricle via the jugular or femoral vein and may be required during balloon aortic valvuloplasty (BAV), implantation of the TAVI prosthesis, post-dilatation or if the patient develops significant conduction disturbance following valve deployment.
The aortic valve is usually crossed with the aid of a Judkins right 4 (JR4) or Amplatz left 1 (AL1) diagnostic catheter and a soft straight-tipped wire. This is then exchanged for a stiffer wire taking care to ensure that this is free of the mitral valve apparatus. Originally, wires (e.g., Amplatz Super StiffTM, Boston Scientific, Marlborough, MA, USA) were manually shaped to create a curve at the end to reduce the risk of ventricular injury during valve deployment. However, dedicated pre-shaped wires (e.g., the SafariTM pre-shaped TAVI guidewire; Boston Scientific)) have more recently been developed that have better memory (and therefore maintain their shape) to further reduce the risk of ventricular injury (Figure 1). Stiffer wires, including the Lunderquist® Extra Stiff wire (Cook Medical, Bloomington, IN, USA) or the Back-up MeierTM guidewire (Boston Scientific), may be used when greater support is required to deliver the TAVI device (e.g., in the setting of severe aortic tortuosity).
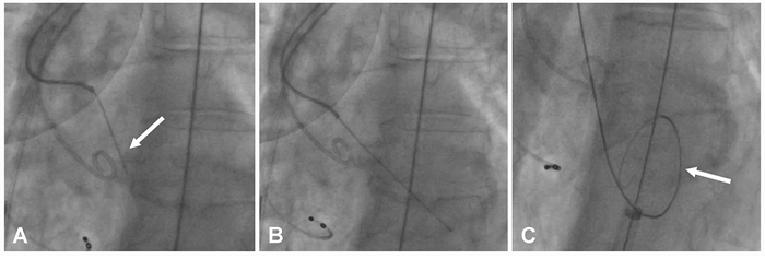
Figure 1. Technique to cross the aortic valve
The aortic valve is initially crossed with a soft-tipped straight wire (A, white arrow) with the aid of an Amplatz left 1 catheter (A). Once the valve is crossed (B), the wire is exchanged for a stiff wire with a curved tip (white arrow) to minimise ventricular injury (C) over which the TAVI device is then advanced.
As summarized in Table 1, each of the current TAVI devices has different mechanisms for implantation. With first-generation devices, BAV was systematically performed prior to valve implantation to facilitate device delivery and valve expansion. Whilst this is still mandatory for some devices (e.g., the Direct Flow valve, Direct Flow Medical®, Santa Rosa, CA, USA), lower profile contemporary devices and greater operator experience now allow routine implantation without prior BAV [6].
Table 1. Currently available TAVI devices.
|
Type of device |
Release |
Valve Size (mm) |
Sheath access route |
Repositionable? |
Retrievable? |
PVL* (>2) |
PPM (30 days)* |
Stroke* (30 days) |
|---|---|---|---|---|---|---|---|---|
|
LotusTM Valve |
M |
23, 25, 27 |
18 Fr TF |
Yes |
Yes |
1-4% |
23.4-28.6% |
1.7-4% |
|
CoreValve Evolut R |
SE |
23, 26, 29, 31 |
14 Fr TF, TS |
Yes |
No |
3.4-5.7% |
8.3 -11.7% |
0-2.7% |
|
SAPIEN 3 |
BE |
20,23,26,29 |
14 Fr, TF, TA |
No |
No |
1.8-4.8% |
8.2-10.1% |
1.4-2.6% |
|
CENTERA |
SE |
23, 26, 29 |
14 Fr TF |
Yes |
No |
8% |
27% |
0% |
|
Direct Flow Medical |
IR |
23, 25, 27, 29 |
18 Fr TF |
Yes |
Yes |
1-2.6% |
13 -17% |
2.4-4% |
|
ACURATE |
SE |
23, 25, 27 |
Sheathless 28 Fr, TA |
No |
No |
7.7% |
2.5% |
5% |
|
ACURATE neoTM |
SE |
23,25,27 |
18Fr TF |
No |
No |
0% |
6.7% |
0% |
|
JenaValve |
SE |
23, 25, 27 |
Sheathless 32 Fr, TA |
Yes |
No |
0% |
12.1% |
3% |
|
Engager |
SE |
23, 26 |
30 Fr TA |
Yes |
No |
0% |
28.5% |
1.7% |
|
Portico |
SE |
23, 25 |
18 Fr TF |
Yes |
No |
4% |
9.7% |
2.9% |
*reported from largest available registries or trials. BE: balloon-expandable; IR: inflatable rings; M: mechanical; mm: millimetre; PPM: permanent pacemaker; PVL: paravalvular leak; SE: self-expanding; TA: transapical; TAVI: transcatheter aortic valve implantation; TF: transfemoral
Transcatheter valves are positioned prior to deployment with the aid of aortography, fluoroscopy and, in some instances, transoesophageal echocardiographic guidance. Balloon expandable valves require rapid ventricular pacing (180-220 beats per minute [bpm]) for deployment to reduce cardiac output and avoid inaccurate valve implantation. Other devices may not routinely require ventricular pacing, although this may still be useful in instances when valve positioning is challenging (e.g., horizontal aorta). Figure 2 illustrates implantation of the more commonly used TAVI prostheses.
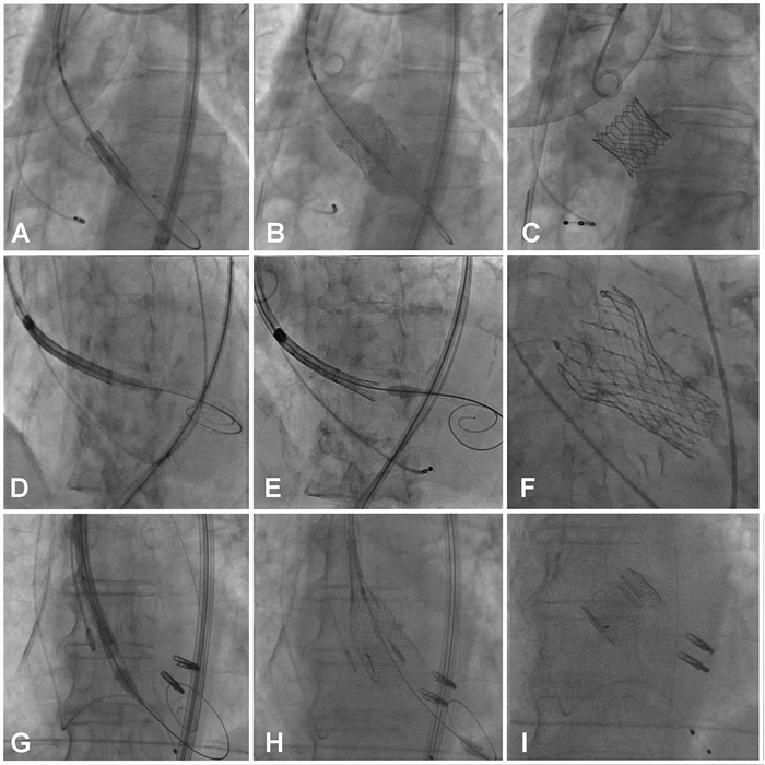
Figure 2. Mechanisms of deployment of commonly used TAVI devices.
Initial position of the balloon-expandable Edwards SAPIEN 3 valve (A), deployment (B) and final appearance (C). Initial position of the self-expanding Medtronic Evolut R valve (D), deployment (E) and final appearance (F). Appearance of the mechanically deployed Boston Scientific Lotus valve (G), deployment (H) and final appearance (I).
Post-implantation assessment
Aortic regurgitation
Trivial or mild aortic regurgitation (AR) is commonly observed following TAVI. However, moderate and severe AR are associated with worse outcomes and independently predict mortality [7]. Possible causes of post-TAVI AR include incorrect valve sizing, suboptimal positioning, eccentric calcification of the aortic root, or incomplete valve expansion. A combination of aortography, echocardiography and invasive haemodynamic assessment (including the AR index [8] and heart rate-adjusted scores [9]) are used to determine the presence and severity of AR.
If AR is more than mild, attempts should be made to reduce its severity. If a repositionable valve has been used (e.g., LotusTM [Boston Scientific], CoreValve Evolut R [Medtronic], or Direct Flow Medical [Direct Flow Medical]), then it may be realigned to achieve a more optimal position. Alternatively, the valve may be post-dilated to improve the apposition of the valve with the aortic annulus [10].
Conduction disturbances
The requirement for permanent pacemaker implantation following TAVI is common and the incidence varies with different prostheses (3.4% for the BE SAPIEN valve [11], 22% for the SE CoreValve [12], 29.3% for the Lotus valve [13] and 16% for the Direct Flow valve [14]). Explanations for these findings include trauma to the conduction system by the catheter and guidewires used during the procedure [15] and aspects of device design, with greater protrusion into the left ventricular outflow tract being associated with a higher incidence of conduction abnormality.
If there has been no observed conduction abnormality during the procedure then the TPW can be removed immediately following BE valve implantation. However, the TPW is routinely kept in situ for a minimum of 24 hours following implantation of other devices (especially SE) due to the risk of late conduction disorders.
Limitations of TAVI
Contemporary TAVI devices have other limitations that should be considered. These are described in more detail below together with possible preventive strategies.
Vascular complications
Vascular complications occur in up to 25% of all patients undergoing TAVI, and major events are associated with adverse clinical outcomes, including an increase in mortality [16]. Small vessel dimensions, moderate or severe calcification and tortuosity of the peripheral vasculature are associated with an increased incidence of complications.
Whilst the traditional treatment for injured arteries has been surgical reconstruction, newer endovascular techniques may allow non-invasive management of arterial complications. Furthermore, newer devices have been developed to enable delivery through lower profile sheaths to further reduce the incidence of vascular complications.
Acute kidney injury
Acute kidney injury (AKI) is a common occurrence following TAVI (when severe it is an independent predictor of adverse outcomes) presumably as a consequence of numerous factors including impaired baseline renal function in this elderly comorbid patient group, contrast nephropathy, and peripheral embolization.
Strategies to reduce its incidence include minimal (or no) contrast administration during TAVI implantation and use of the RenalGuard® system (RenalGuard Solutions Inc., Milford, MA, USA) which may have a potential role [17].
Stroke
The incidence of clinical stroke following TAVI is approximately 3-4% [18], although numerous imaging studies have demonstrated the occurrence of new cerebral ischaemic defects in a larger proportion of patients (the clinical relevance of which is currently unclear). Anatomical features such as the presence of a severely stenotic and calcified aortic valve, use of large delivery systems, and multiple manipulations during device implantation have been associated with an increased risk of stroke. Furthermore, new onset atrial arrhythmias (which are noted in up to one third of patients undergoing TAVI) may also be important [19].
A number of strategies are under investigation to reduce stroke, including aggressive anticoagulant and antiplatelet strategies and the use of dedicated cerebral protection devices (e.g., Claret dual-filter cerebral protection system (Claret Medical, Santa Rosa, CA, USA) and Embrella device [Edwards LifeSciences]). Use of these devices has been shown to significantly reduce the number of ischaemic cerebral lesions in the protected brain regions, although the clinical relevance of these findings remains unclear. Longer follow-up in larger numbers of patients is required to demonstrate efficacy.
Post-procedural care
Patients are routinely admitted to a critical care environment following TAVI to ensure optimal haemodynamic monitoring and nursing care. This may be required for only the immediate post-procedural period following an uncomplicated transfemoral procedure carried out under conscious sedation but may be much longer in a frail elderly patient requiring surgical access site under GA.
Patients are then transferred to a general ward for ongoing care, including optimisation of medication and rehabilitation. Patients are routinely discharged 2-3 days following their procedure, although a considerably longer hospital stay may be required in the setting of other medical problems or when prolonged rehabilitation is necessary. Amazingly, same-day discharge has recently been demonstrated to be feasible in highly-selected uncomplicated patients [20].
Conclusion
In conclusion, in spite of greater operator experience and improvements in TAVI technology, patients treated with this revolutionary procedure remain a high-risk, fragile patient group. A number of technical considerations and an appreciation of the limitations of current devices (and techniques) need to be recognised in order to optimise procedural and longer-term outcomes.