Background
Hyperkalemia is defined as a serum potassium level greater than 5.5 mmol/L [1]. Chronic kidney disease is the most common cause inducing hyperkalemia. The prevalence is 2-3% in the general population but is as high as 40-50% in the elderly population with chronic kidney disease [2]. The renal adaptive system disrupts the acid-base balance and urinary potassium excretion when the glomerular filtration rate declines to less than 15 ml/min/1.73 m2 [3]. Ninety percent of potassium is excluded from healthy kidneys, but in patients experiencing end-stage renal disease the potassium excretion from the intestine increases [4]. In such a condition, potassium excretion is largely dependent on the delivery of sodium and aldosterone secretion to collecting tubules as well as colonic mucosa.
Medications can affect the serum potassium level, and patients who have taken angiotensin converting enzyme inhibitors/angiotensin receptor blockers, and/or aldosterone antagonists are susceptible to the risk of hyperkalemia [2]. Specifically, diabetic patients or elderly patients with a decreased glomerular filtration rate need to have their potassium level monitored frequently [5]. Clinical manifestation due to hyperkalemia may be vague (i.e., muscular weakness of a non-specific nature, nausea, vomiting and diarrhea), and hyperkalemia is often revealed in incidental laboratory findings. Hospitalized patients with hyperkalemia have a higher mortality rate than those without hyperkalemia, and it is noted that renal insufficiency significantly contributes to the confounding variables [2,6].
What are the clinical consequences of hyperkalemia on the heart?
Potassium is a main intracellular cation (100-150 mmol/L), and the inward potassium current is responsible for the maintenance of resting membrane potential (Vm) [7]. A small increase in the extracellular potassium level leads to a great change in the ratio of intracellular ([K+]i) to extracellular potassium ([K+]o), resulting a decrease in the resting membrane potential (Vm= -61.5 log [K+]i/[K+]o).
The cardiac muscle is the most prominent organ subjected to hyperkalemia, and the increased serum potassium ion leads to the conduction abnormality, and impaired myocardial contraction [8]. Atrial tissues are more sensitive to hyperkalemia than the sinus node, ventricular tissues or the bundle of His [9-11]. As the potassium ion level increases, the sinus node continues to propagate electrical activity to the ventricles without depolarizing the atrial muscle, called sino-ventricular rhythm. In addition, hyperkalemia causes implanted pacemaker malfunctioning [12]: 1) widening of the paced QRS complex, 2) failure to capture the atrial and ventricular pacing, and 3) delay from the pacing stimulus to the onset of ventricular (or atrial) depolarization.
What is the use of the electrocardiogram in hyperkalemia?
An increase of serum potassium levels in the blood is manifested by the surface electrocardiogram. Classically, the electrocardiogram reveals peaked and symmetric T-waves (serum potassium 5.5-6.5 mmol/L), to a loss of P-waves (serum potassium 6.5-7.5 mmol/L), widened QRS complex (serum potassium 7-8 mmol/L), and ultimately a sine wave pattern (serum potassium >8 mmol/L) (Figure 1 A-C) [1]. It is also documented that sinus arrest, marked intraventricular conduction delay, ventricular tachycardia and ventricular fibrillation can develop at potassium ion levels greater than 10 mmol/L [11].
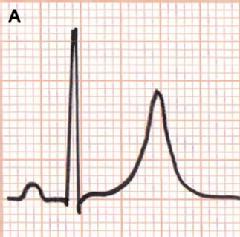
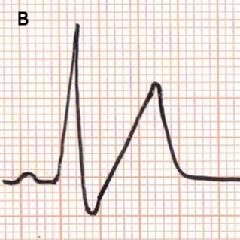
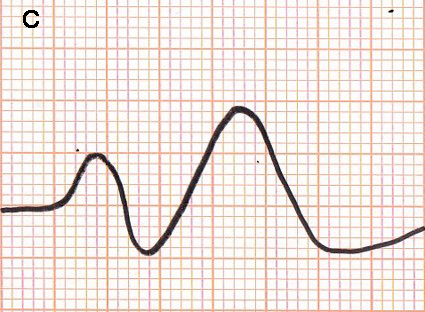
Figure 1. Typical changes in electrocardiograms in the development of hyperkalemia. Serum potassium ion, 6.5-7.5 mmol/L (A); 7-8 mmol/L (B); > 8 mmol/L (C).
Other arrhythmias associated with hyperkalemia include right or left bundle branch block, hemi-block and advanced atrioventricular block [11,13,14]. Figure 2A is an example of continued stimulation of the ventricles without atrial electrical activity in severe hyperkalemia: an 82-year-old man was admitted to our hospital due to general edema with progressive renal dysfunction within a short period. His blood test demonstrated that urea nitrogen was 94 mg/dL, creatinine was 10 mg/dL, sodium ion was 119 mmol/L and potassium ion was 7.7 mmol/L. A simultaneous 12-lead electrocardiogram presented the widened QRS tachycardia in the absence of a P-wave. He was treated with an infusion of gluconate calcium and glucose/insulin, followed by hemodialysis. His serum potassium ion level declined to 4.7 mmol/L, and the electrocardiogram showed recovery of sinus rhythm. Figure 2B shows an 83-year-old man admitted to our hospital due to dimmed vision. A surface 12-lead electrocardiogram presented sinus arrest with peaked T-wave. His blood test revealed that urea nitrogen was 72 mg/dL, creatinine was 3.67 mg/dL, sodium ion was 137 mmol/L, chloride ion was 104 mmol/L and potassium ion was 6.0 mmol/L. After an intravenous infusion of gluconate calcium and subsequent hemodialysis, his potassium ion level dropped to 4.2 mmol/L and the electrocardiogram showed a return to normal sinus rhythm.
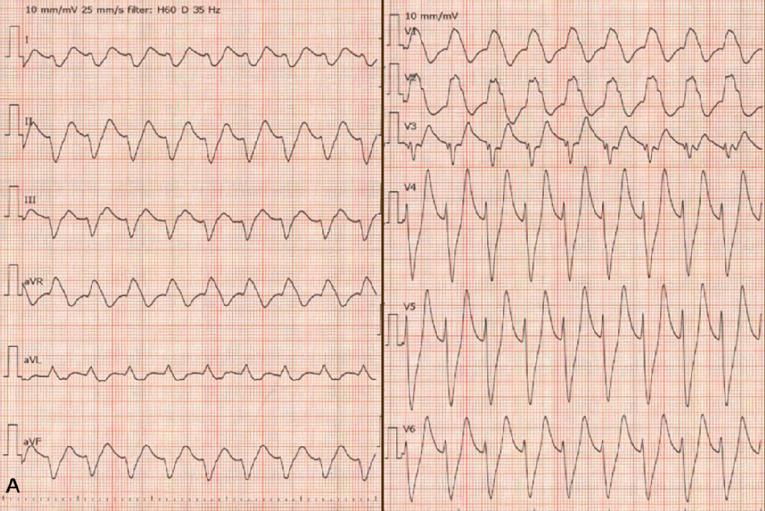
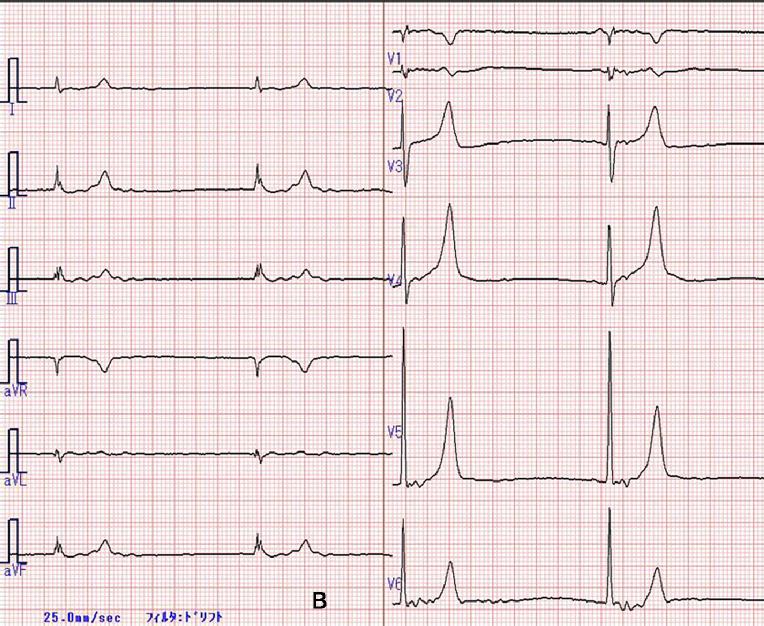
Figure 2. Two examples of electrocardiograms. A) An 82-year-old man admitted to our hospital due to general edema with progressive renal dysfunction within a short period (serum potassium ion was 7.7 mmol/L). B) An 83-year-old man admitted to our hospital due to dimmed vision (serum potassium ion was 6.0 mmol/L).
Thus, diagnosis of hyperkalemia through the electrocardiogram is challenging, because the visual information about the morphology of P-wave amplitude, QRS prolongation and T-wave changes is not sensitive and specific enough to identify the magnitude of hyperkalemia; no diagnostic threshold of T-wave amplitude has been found [15], and the sensitivity of diagnosis of hyperkalemia is as low as 0.34 to 0.43 for physician readers of the electrocardiogram, whereas the specificity is higher at 0.85. Specifically, missed diagnosis has been shown to occur with potassium levels between 5.0 and 6.4 mmol/L [16]. In accordance with our experience, a modest elevation of potassium ion levels, such as 6.4 mmol/L [13] and 5.5 mmol/L [17], can lead to sinus arrest. On the other hand, Martinez-Vea et al [18] reported that electrocardiograms did not reveal any typical changes even the potassium ion level was greater than 8 mmol/L. Thus, it is likely that the serum potassium ion level itself cannot explain the electrocardiographic changes. It is important to note that multiple factors, such as acid-base imbalance, rapid rise of extracellular potassium ion level, coexisting low serum sodium and calcium ion concentrations, latent sinus node dysfunction and pre-existing myocardial ischemia, may influence the electrophysiological effect of hyperkalemia in cardiac tissues [17,19,20]. These results suggest that the management of hyperkalemia should not rely on the electrocardiogram, but be guided by the clinical scenario and repeated potassium ion measurements [15].
Conclusion
It is difficult to diagnose the magnitude of hyperkalemia based electrocardiogram at presentation; on the contrary, it is impossible to predict the development of a fatal arrhythmia based on the serum potassium level. Therefore, physicians should start the treatment promptly by considering the clinical scenario of the patient (i.e., underlying renal dysfunction, and medications taken potentially to increase the potassium ion level) and any electrocardiographic changes suggesting the suspicion of hyperkalemia while awaiting the chemical results.















