Introduction
Mitral regurgitation (MR) has a variable prognosis depending on its severity and on its aetiology which includes organic disorders mainly due to structural or degenerative abnormalities of the different mitral valve components (leaflets, chordae tendineae, papillary muscles, mitral annulus), or a functional effect that occurs in the absence of organic mitral valve (MV) disease. Functional mitral regurgitation (FMR) is one of the most critical conditions in current clinical cardiovascular practice and is present in many patients with left ventricular (LV) systolic dysfunction contributing to heart failure (HF) and to its worsening. It is more frequent as a complication of ischaemic heart disease rather than of dilated cardiomyopathy (DCM). Due to the large number of patients with acute myocardial infarction (AMI), the prevalence of FMR is high. Following MI, nearly 50% of patients will develop different degrees of MR, and one in every four patients with MI will progress to at least a moderate grade of insufficiency [1]. FMR is strongly associated with unfavourable outcomes. Additionally, the degree of FMR relates to morbidity and mortality in patients with HF.
The high operative mortality risk of these patients, as well as the unclear strategy of surgery, more so in patients with severe MR and severe LV systolic dysfunction, has led to the search for other therapeutic approaches. Currently, the surgical gold standard treatment for FMR is down-sized ring annuloplasty; however, the recurrence of MR after two to four years may be high. Percutaneous mitral valve repair techniques have already been developed in an effort to solve FMR. Cardiac resynchronisation therapy (CRT) has appeared as a useful therapeutic tool in patients with HF, reduced LV ejection fraction (LVEF), QRS disturbances and limiting symptoms despite optimal medical treatment [2].
This article focuses specifically on FMR, the understanding of which requires a holistic comprehension of the LV and the mitral valve structure and their interdependence. Interest in CRT for the management of patients presenting with FMR and HF with reduced LVEF has increased in the last few years because CRT is capable of offsetting some of the pathophysiologic mechanisms of FMR due to the decrease in LV dyssynchrony.
Prevalence and prognosis of functional mitral regurgitation
An accurate estimation of the prevalence of FMR has been difficult due to the different criteria and techniques used in the diagnosis, the timing of that diagnosis and the characteristics of the patient population. Nevertheless, the estimated prevalence ranges from 8% to 74% in echocardiography studies [3], bearing in mind that the timing of diagnosis is crucial due to the dynamic characteristic of this disease. There is usually a more pronounced mitral deformation, and therefore more severe regurgitation, in patients with ischaemic heart disease (IHD) than in patients with DCM [4]. In clinical practice, the recognition of heart failure signs and symptoms in IHD is crucial to identifying it. FMR independently adds a worse prognosis (adverse outcome and even all-cause death [5]) in patients with LV systolic dysfunction, and it also implies a higher surgical risk [6]. The prognosis of MR in patients with chronic HF remains controversial. Several studies have reported unfavourable outcomes in patients with DMC and a strong association between FMR severity and all-cause mortality and hospitalisation [7]. In terms of quality of life, untreated FMR in patients with HF is correlated with an increased risk of hospital admission for acute decompensated HF.
A dilated and dysfunctional LV can imply FMR which itself contributes in turn to worsening LV remodelling. This issue raises a dilemma about treatment strategies.
Mechanisms of functional mitral regurgitation
Nowadays, FMR is understood to be a more complex pathology than initial theories explained, where only papillary muscles were involved; FMR occurs in valves without organic lesions. The changes in LV geometry secondary to remodelling, whether the cause is IHD or DCH, lead to a disruption in the normal relationships between the LV and the mitral apparatus: restriction in leaflet motion and incomplete mitral leaflet closure cause regurgitation. In the following paragraphs we explain the role of the different components which participate in the genesis of mitral regurgitation.
Left ventricular remodelling
LV remodelling occurs after a local (acute myocardial infarction) or global (DCM, volume or pressure overload) myocardial injury. The key element in the development of FMR is LV remodelling, which means changes in the geometry, volume and function of the LV. In the case of IHD, it not only affects the infarcted region but also remote areas and the left ventricle overall. The mechanisms of remodelling are complex and inter-related: reorganisation of myocytes, intercellular matrix and microvessels produce stiffness and impaired contractility; there are changes in diastolic and systolic properties; the scar or more affected regions tend to be more liable to deformation. During systole the closure of the mitral leaflets is arbitrated by the interaction of closing forces represented by the LV-left atrium (LA) pressure gradient and the opposing tethering forces determined by LV intracavitary systolic pressure. The latter are transmitted by papillary muscles (PM) through the chordae to prevent mitral leaflets from prolapsing and they are dependent on normal LV-MV spatial relationships. The remodelled LV can change the normal site of the papillary muscles, increasing the distance between them and the mitral leaflets. As a result, the tethering forces increase too. All these factors cause a restriction in systolic leaflet movement and a displacement of the coaptation point apically [3]. The presence of LV dysfunction alone has not been tested as being the only factor responsible for mitral regurgitation. On the other hand, it is also important to understand that FMR is not only due to LV dysfunction but that the key is an imbalance of forces [8]. FMR is an active condition that is affected significantly by changes in loading, pressure or volume situations because they potentiate that imbalance.
Mitral apparatus
Two phenotypes of tenting have been described: asymmetric and symmetric. The asymmetric pattern is characterised by posterior displacement of the posterior leaflet following the papillary muscle, leading to an increase of posterior traction forces restricting its systolic movement and displacing the coaptation point posteriorly [3]. This pattern is more frequent in inferolateral myocardial infarctions because the inferior papillary muscle is predominantly affected (the anterior leaflet may also be affected because it also receives chords from that muscle but this is more rare; in that case it acquires a hockey-stick morphology). The jet direction is opposite the affected leaflet, it is usually not significant and it is related to a lesser degree of LV dysfunction. In the symmetric pattern, both leaflets are similarly affected due to the apical displacement of both papillary muscles. In this case the coaptation point is set apically and the resulting regurgitant jet is central. In this group, the LV overall remodelling is more evident as is the dilatation and dysfunction. It presents typically in patients with more extensive IHD.
The identification of the tethering is very helpful in choosing the surgical strategy.
Role of the annulus
Patients with FMR present a larger annulus size and flattened shape. The changes in geometry are more evident in anterior myocardial infarctions. The mechanism of why the annulus dilation worsens FMR remains unclear. Isolated annulus dilation does not lead to mitral regurgitation in the absence of mitral leaflet tethering. However, if the annulus dilation is combined with the latter, they both contribute to FMR and, moreover, to the perpetuation and deterioration of FMR. The left atrium may play a role in this phenomenon because its enlargement due to chronic mitral regurgitation relocates the posterior mitral annulus portion to the top of the LV inflow tract, resulting in tethering of the posterior mitral leaflet.
Cardiac mechanical dyssynchrony
Cardiac dyssynchrony can be categorised into atrioventricular (AV), interventricular and intraventricular dyssynchrony. An increase in atrioventricular conduction time entails the delay of the systolic ventricular contraction, which hinders diastolic LV filling that leads to an increase of diastolic pressure. Interventricular dyssynchrony means an LV asynchronous contraction in respect to the right ventricle. Intraventricular dyssynchrony refers to differences in the timing of contraction in distinct myocardial segments following an ineffective LV overall contraction which reduces the stroke volume. Intraventricular dyssynchrony may be caused principally by ischaemia (IHD) and LV remodelling due to pressure/volume overload because of the coexistence of a delay in the electrical depolarisation.
Dyssynchrony is an electrical condition that has several mechanical consequences. When dyssynchrony occurs, regions that are electrically activated earlier contract first, while at the same time other regions with electrical delay are pre-stretched and will contract later. This reduces the efficiency of LV pump action by increasing oxygen consumption and reducing the stroke volume [9].
Acute myocardial ischaemia or its sequelae are one of the main causes of intraventricular dyssynchrony because regional wall motion abnormalities appear. Several studies have shown that the dyssynchrony of myocardium surrounding the papillary muscles is related to the development of FMR [10]. Also, the uncoordinated regional LV activation causes an imbalanced systolic force in the left ventricle, decreasing MV systolic closing forces [11].
Left bundle branch block (LBBB), primary or secondary to ischaemia, also implies a dyssynchrony in LV contraction: the segment next to anterolateral papillary muscle contraction is delayed with respect to the inferior segment. The different time-to-peak contraction time between both papillary muscles will result in changes in the normal mitral tethering forces which, linked to difficulty in mitral closure because of geometrical changes, exacerbates the LV remodelling and a vicious circle is established. This time interval has been identified as the principal factor of mitral regurgitation in patients with heart failure [12]. Besides, LBBB by itself in patients with DCM aggravates the MR duration by increasing the LV isovolumetric contraction and relaxation periods.
Finally, mechanical dyssynchrony and the subsequent LV remodelling lead to an increase in wall stress and LV dilation which is a major contributor to FMR. An improvement of MR is associated with reverse LV remodelling, a decrease in LV volume, and outcome improvement in patients with HF presenting with FMR.
Role of cardiac resynchronisation to manage mitral regurgitation
CRT is a well-established recommended therapy for patients with HF, depressed LV ejection fraction and intraventricular conduction disturbance but its therapeutic benefits have been extended to a wider range of clinical situations than previously reported [2]. CRT reduces morbidity and mortality rates as well as improving symptoms and quality of life in patients with HF. CRT devices commonly have three leads that are placed, respectively, in the right atrium, in the right ventricle (RV) and in the LV (it is usually implanted through the coronary sinus and placed into a target vein to pace the epicardial LV free wall) (Figure 1). The role of CRT is to detect atrial activity (if not, it can stimulate too) and accordingly to stimulate both ventricles so that their contraction occurs at once; moreover, the tardy contraction of some LV segments due to QRS prolongation can be corrected. In other words, during CRT pacing, the simultaneous biventricular stimulation decreases the electrical delay, improving the coordinated overall contraction between different segments of the heart, increasing global LV contraction efficiency. Finally, if the LV has a better stroke volume, it can become a normal sized and a normally functioning LV (inverted remodelling).
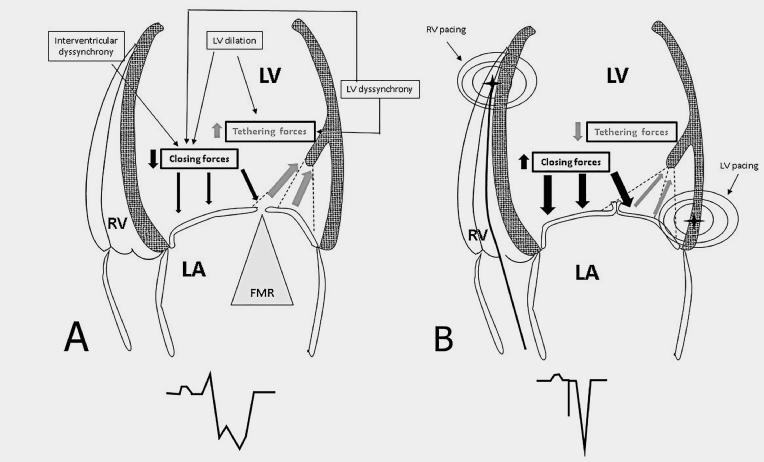
Figure 1. Functional mitral regurgitation (FMR) mechanisms.
- A) Illustration showing functional mitral regurgitation (FMR) mechanisms on the right.
- B) Effects of simultaneous biventricular pacing with a lead located at the right ventricle (RV) apex and a left ventricle (LV) lead.
The lower part of the panel shows the differences in QRS duration.
FMR: functional mitral regurgitation
When the pathophysiology of the FMR is understood, the fact that one therapy could contain all the implicated factors (and the vicious circle) is encouraging:
- AV dyssynchrony
- LV dyssynchrony
- LV function
- LV dimensions
- PM-bearing segments dyssynchrony
The benefit of CRT is derived from the LV dyssynchrony (which implies the segments where the papillary muscles are set) overall improvement. If a synchronous and effective LV contraction is achieved, LV volumes (inverted remodelling) and ejection fraction return to normal limits and therefore: 1) closing forces are restored, 2) tethering forces diminish, 3) interpapillary muscle-bearing segments have the same time to peak maximum contraction, and 4) the mitral annulus gets smaller.
All the effects do not occur at the same time, but one leads to the other:
- All the LV segments contracting at the same time increases the closing forces.
- Synchronous contraction of the papillary muscle-bearing segments diminishes the tethering forces.
Then, a nearly immediate MR response is obtained which is directly related to the electric cardiac stimulation. Following that, there is less LV volume overload which, in addition to the increased LV contraction efficiency, participates in the restoration of the LV size. Once the LV is reshaped, the tethering forces also diminish, the closing forces also get better and better, and the mitral annulus geometry is in better condition to act as a sphincter.
Another potential mechanism of CRT is the restitution of the atrioventricular synchrony: when the LV contraction follows the atrial activity, the LV contraction effectiveness increases the transmitral closing force.
In summary, CRT has a double effect on MR: the rapid effect has been related to the decrease of interpapillary dyssynchrony, leading to an increase in closing forces; the later effect is due to the better contractile recruitment and LV systolic function.
Knowing that CRT performs its function by electrical myocardium activation through a cell-to-cell mechanism, the presence of viable myocardium is very important: the extent of the scar is connected with non-responders [13], in which case it is very important to choose the best segment to place the LV lead. The whole clinical evaluation must include the viability assessment by cardiac magnetic resonance, nuclear medicine or stress echocardiogram. Placing the LV electrode in regions with longer electrical delay may achieve better results in FMR (this fact must be considered because it is of major importance for CRT optimal response).
In addition, the mitral valve response has been identified as a prognostic factor in patients under CRT: the persistence of mitral regurgitation represents a notable predictor of arrhythmias, hospitalisation and mortality. Only three trials have included MR in their outcomes evaluation, CARE-HF [14,15], MIRACLE [16] and MUSTIC [17]. They found that MR was reduced by CRT (worse results in ischaemic patients) and that the persistence of MR was related to morbidity and higher mortality. It should be pointed out that those trials were carried out in patients with heart failure, not FMR specifically. In this regard, Upadhyay et al [18] showed that, in terms of LV function improvement, even the LV function responders had a poorer outcome if the mitral regurgitation persisted (non-responders had higher tethering forces characterised by augmented tenting areas). Cabrera-Bueno et al [19] related the persistence of MR to adverse arrhythmic events.
Finally, CRT is recommended over simple RV pacing in patients with heart failure and reduced ejection fraction who need a pacemaker with a high percentage of ventricular pacing [20] in order to avoid the whole cascade of dyssynchrony and MR appearance [2].
The final decision concerning the best approach to FMR should be obtained by a comprehensive and multidisciplinary evaluation.
Conclusion
Mitral regurgitation is a frequent complication of IHD and DCM. Its presence overshadows the prognosis of these patients. Its development is the result of the imbalance between closure forces and tethering forces secondary to a disruption in the normal LV geometry and to a dyssynchronous LV contraction.
Medical treatment reduces the regurgitant fraction and improves heart failure symptoms and exercise capacity. There is no consensus about the best repair strategy (surgical or percutaneous). CRT appears to be a growing therapy for patients with moderate to severe FMR, LV dysfunction, QRS disturbances and at high surgical risk because it can control and even improve the pathologic base of the disease.















