Introduction
High-intensity exercise training leads to structural, functional, and electrical changes of the heart, which are referred to as ‘‘athlete’s heart’’, characterised by increased left ventricular mass (LVM), cavity dimensions and wall thickness [1]. Athletes with left ventricular hypertrophy generally have a normal ejection fraction (EF) and no evidence of either systolic or diastolic dysfunction [2]. Recent studies have documented decrements, especially in right ventricular (RV) function, during intense endurance exercise [3,4]. Actual evidence suggests that there may be some overlap between physiological and pathological hypertrophy, such that a small amount of fibrosis may accompany cardiac remodelling associated with lifelong endurance training and then acts as a substrate for arrhythmias [5].
Myocyte damage during high-intensity exercise
After intense exercise, acute increases in troponin (cTn) and B-type natriuretic peptide (BNP) have been detected in athletes [6]. These are specific markers of myocyte damage and strain, but none of these indicates a permanent injury. Potential mechanisms have been proposed to explain cTn elevations after prolonged or strenuous exercise, but actually the release of cTn by healthy individuals cannot be explained by any of these theories. It is possible that exercise induces an increase in myocardial sarcolemma permeability with passive diffusion of cTn from the intracellular to extracellular compartment. The increase of membrane permeability might be due to mechanical stress on the cardiomyocytes, increased production of oxidative radicals or exercise-induced altered acid-base balance [6].
Some individuals with post-exercise cTn elevation might have myocardial injury with necrosis, taking into account the case reports of myocardial fibrosis and late gadolinium enhancement (LGE) in a small number of veteran athletes, but the pathogenesis of these cases remains unclear. However, routine cTn testing is not indicated for the evaluation of individuals who present for medical attention after participation in exercise sessions, unless there are specific clinical data suggesting cardiac involvement [6].
Key point:
- Routine cTn and BNP testing is not indicated for the evaluation of athletes who present for medical attention after exercise sessions, unless there are specific clinical data suggesting cardiac involvement.
Exercise-related myocardial fibrosis
Despite the widely recognised benefits of regular physical activity, numerous observational studies have shown that high-level exercise training may be associated with an increased risk of cardiac arrhythmia and even primary cardiac arrest [7].
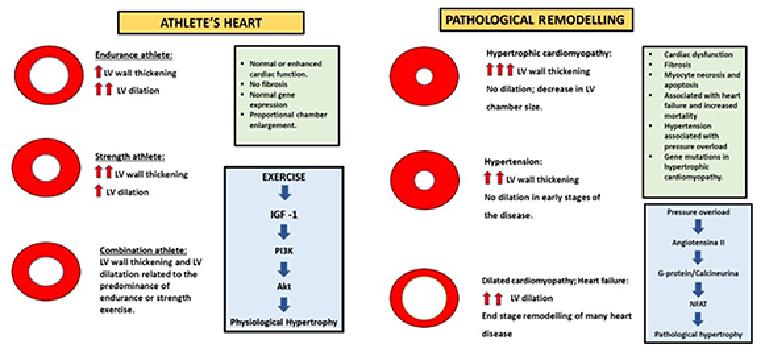
Athlete’s heart is a benign condition that represents a physiological adaptation to chronic training in which, usually, loss of myocytes and abnormal deposition of collagen do not occur. The fibrillar collagen network provides structural integrity of adjoining myocytes, facilitating myocyte shortening which translates into efficient cardiac pump function [8]. Pathological hypertrophy is associated with cell death (apoptosis, necrosis) and the loss of myocytes is replaced with excessive collagen. An excessive accumulation of collagen stiffens the ventricles, which impairs contraction and relaxation, damages the electrical coupling of cardiac myocytes with extracellular matrix proteins, and reduces capillary density. Fibrosis and reduced capillary density increase oxygen diffusion distances, leading to myocardial ischaemia. This is likely to contribute to the transition from hypertrophy to failure (Figure 1) [8]. Interestingly, recent studies have shown myocardial inflammation and fibrosis in animal models of long-term, intensive exercise. Chen et al [9] forced rats to swim strenuously, with histological evidence of localised myocyte damage, myocardial necrosis and inflammatory infiltrates. Benito et al [10] instituted an intensive treadmill running regime in young rats and showed that in the “marathon rats” there was an increase in atrial and ventricular inflammation/fibrosis. Most importantly, this was associated with a greater potential for inducible ventricular arrhythmias.
Figure 1. The different morphological and molecular characteristics between physiological remodelling (athlete’s heart) vs. pathological remodelling.
Fibrosis and inflammatory infiltrates have been identified in well-trained athletes who underwent a cardiac biopsy for high pre-test probability of identifying a cardiac pathology [11]. Histology provides the only direct evidence of fibrosis but, most commonly, inflammatory infiltrates and fibrosis are non-specific and their aetiology can be deduced only by other clinical factors. Furthermore, a cardiac biopsy is an invasive procedure with significant risks; it is not applicable in the absence of a strong suspicion of heart disease.
An accurate, non-invasive surrogate tool for detecting fibrosis is cardiac magnetic resonance (CMR) imaging with gadolinium contrast. Gadolinium-based extracellular paramagnetic contrast agents accumulate in areas of extracellular expansion and thus can be used to delineate areas of injured myocardium. Typically, areas of gadolinium accumulation relate to areas of scar expansion due to focal myocardial replacement fibrosis. Using gradient-echo inversion recovery imaging, fibrosis appears as a bright signal, with a prolonged wash-out time for the gadolinium (delayed gadolinium enhancement [DGE]), contrasting with the normal myocardium, which looks black [5]. Several studies have identified DGE in extensively trained veteran athletes. In most cases, the patches of DGE were very small and sited around the septum and RV insertion points, regions subjected to local mechanical stresses during exercise [5].
More recently, La Gerche et al have shown myocardial fibrosis with CMR, and a reduction in RV function occurred in those athletes competing for a longer duration, suggesting that the heart has a finite capacity to maintain the increased work demands of exercise. The patches of cardiac fibrosis may be the substrate for ventricular tachycardia and sudden death in predisposed individuals [3]. Based on this finding, some authors have recently proposed a new type of cardiomyopathy, the so-called Phidippides cardiomyopathy: repetitive sustained exertion can induce dilation of the cardiac chambers and also stimulates resident macrophages, pericytes, and fibroblasts, resulting in the deposition of collagen-causing patchy fibrosis [12,13]. CMR can also specifically detect intramyocardial fibro-fatty infiltration of the RV wall, typical of arrhythmogenic right ventricular cardiomyopathy (ARVC). This often leads to ventricular arrhythmias and usually appears in young adulthood, and in affected asymptomatic or minimally symptomatic individuals [13].
In conclusion, it is possible that the right ventricle is more susceptible to fatigue than the left ventricle (LV) after prolonged exercise. More studies are required to identify probable effects of exercise “dose” and their implication in the development of heart failure. Furthermore, it is important to underline that some banned athletic performance-enhancing drugs might have cardiac toxic effects, such as anabolic-androgenic steroids (AAS). Healthy athletes, abusing AAS, may exhibit LV hypertrophy with impairment of both systolic and diastolic myocardial function and focal areas of DGE, with a pattern of non-ischaemic distribution [14].
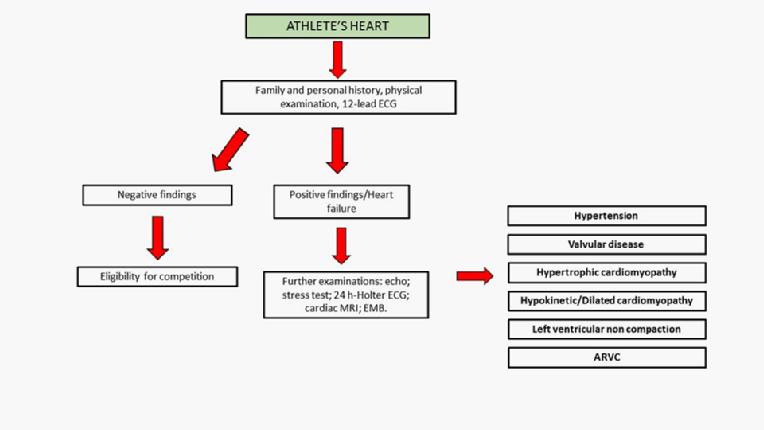
Figure 2 presents an algorithm for the differential diagnosis between an athlete’s heart and pathologic conditions.
Figure 2. Heart failure and athlete’s heart: a flow chart for a differential diagnosis.
Key points:
- It is possible that the right ventricle is more susceptible to fatigue than the left ventricle after prolonged exercise.
- Recent studies have shown fibrosis in athletes competing for a longer duration, sited around the septum and RV insertion points, regions subjected to local mechanical stresses during exercise.
- The existence of Phidippides cardiomyopathy is uncertain.
Cardiovascular drugs in athletes
Athlete’s heart is a physiological condition and does not require a specific treatment. The athletic population and professional athletes are not risk free in terms of developing cardiovascular disease, despite the fact that regular exercise is a healthy habit and commonly protects against cardiac injury. Although the prevalence of high blood pressure (BP) is approximately 50% lower in athletes compared with the general population, it is also the most common cardiovascular condition in athletes. Therapy in this population can be difficult because of competition regulations and the potential adverse effects. Hypertension is associated with an increased risk of developing heart failure (HF); antihypertensive therapy markedly reduces the incidence of HF [15]. Unfortunately, few studies have evaluated the effects of antihypertensives in athletes and their consequences on exercise performance (Table 1) [16].
|
Drug |
Side effects |
Effects on exercise |
|---|---|---|
|
Diuretics |
Dehydration Electrolyte abnormalities |
“Masking agents” for doping substances. Diuretics are prohibited in competitive athletes. |
|
α-blockers |
Dizziness Orthostatic hypotension Nasal congestion Headache Fluid retention Reflex tachycardia |
No change in energy metabolism. No change in VO2 max. |
|
CCBs |
Headache Constipation Fluid retention Drowsiness Hypotension Dizziness |
Minimal effect on exercise heart rate and VO2 max. Dihydropyridines or long-acting diltiazem-like CCBs are well tolerated and effective in athletes. |
|
ACEi |
Cough and rash Headache Drowsiness Weakness |
No effects on heart rate and minimal effects on VO2 max. First choice in athletes. |
|
β-blockers |
Fatigue Cold hands Headache Constipation Dizziness Trouble sleeping Loss of sex drive/erectile dysfunction Depression |
Important effects on heart rate and on VO2 max. They are avoided in athletes. |
CCBs: calcium channel blockers; ACEi: angiotensin-converting enzyme inhibitors.
Table 1. The side effects and the consequences on exercise of the most common cardiovascular drugs.
Diuretics are restricted in athletes, because their use may result in dehydration and electrolyte abnormalities, such as hypokalaemia. In addition, diuretics are prohibited in this population by the National Collegiate Athletic Association (NCAA) and the World Anti-Doping Agency (WADA), because they can be regarded as "masking" agents due to the dilution of urine, which results in lower levels of doping substances being excreted from the body [17].
Alpha-blocker drugs, blocking the effect of sympathetic nerves on blood vessels by binding to α-adrenoceptors located on the vascular smooth muscle, do not cause major changes in energy metabolism during exercise, and maximum oxygen uptake (VO2 max) is preserved. However, they are not widely used in athletes, given the associated side effects such as dizziness, orthostatic hypotension, nasal congestion, headache, fluid retention, and reflex tachycardia [18].
Beta-blockers in general have an adverse effect on exercise heart rate, VO2 max, when compared with placebo, and they are avoided in athletes [18,19].
Calcium channel blockers (CCBs) appear to have a minimal effect on exercise heart rate and VO2 max. Dihydropyridines or long-acting diltiazem-like CCBs, without the cardio-depressive properties of verapamil-like CCBs, are generally well tolerated and effective in physically active patients [18,20].
Angiotensin-converting enzyme inhibitors (ACEi) appear to be ideal agents for antihypertensive therapy in athletes. Several small studies have shown evidence that ACEi have no effect on exercise heart rate and a minimal effect on VO2 max, and are well tolerated [18]. Unfortunately, data regarding ACEi and their action on athletes are lacking. The activation of the renin-angiotensin-aldosterone system is responsible for pathological cardiac hypertrophy and fibrosis, whereas in physiologic hypertrophy there is activation of the IGF1-PI3K-AKT1 pathway that protects the heart against failure, fibrosis and apoptosis, and inhibits the pathological signalling cascade. Therefore, the induction of physiological cardiac hypertrophy may be a potential therapeutic strategy for the treatment of heart failure [8].
Key points:
- Data regarding the treatment of hypertension and the effects of cardiovascular drugs on exercise are lacking.
- The first-line drugs in athletes for the treatment of hypertension are ACEì. They are well tolerated and have no effect on exercise.
Conclusions
The exact clinical significance and prognostic value of cardiac injury and fibrosis in athletes are unknown. More large prospective studies are needed to provide a better understanding of these conditions and to investigate the effects of cardiovascular drugs in this population.