Sudden cardiac death (SCD) is the most frequent mode of death due to cardiac disease. In older individuals (>35 years old), the main cause is ischemic heart disease, while in younger individuals (<35 years old) the main cause are genetic cardiomyopathies or channelopathies.1 In this age group, the incidence of SCD is reported to be around 1-3 per 100 000 individuals.
Genetic testing for SCD cases aims to identify the rare genetic variant with high effect size considered as the main causal factor for the sudden death.2 Two different situations can be present, either the individual suffering from SCD is deceased or successfully resuscitated.
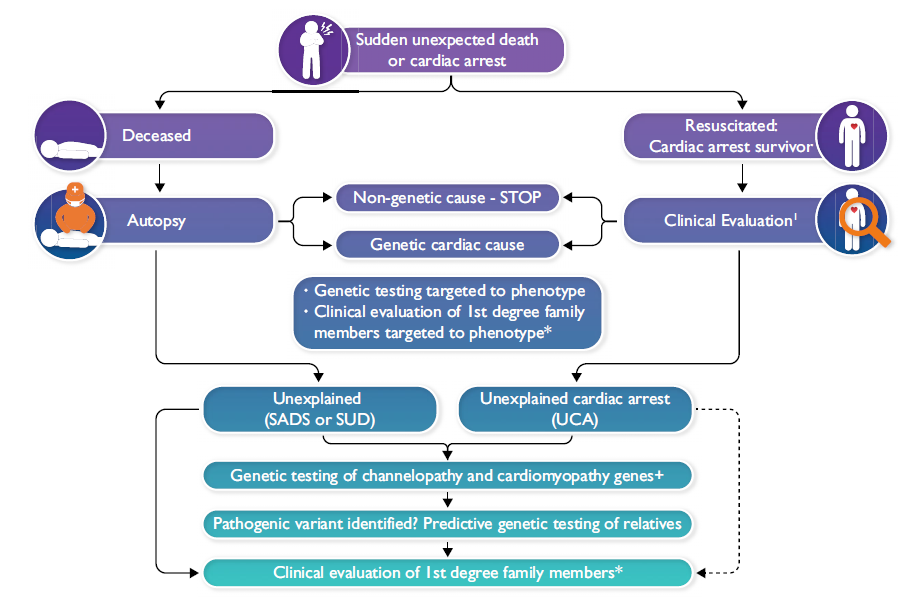
Genetic testing for deceased individuals with SCD – the ‘molecular autopsy’
Whenever SCD happens, a thorough examination is warranted including an autopsy and toxicology. If the autopsy points in the direction of an underlying cardiomyopathy, phenotype directed gene panel testing is advised. If SCD remains unexplained after autopsy and toxicology (so called sudden arrhythmic death syndrome; SADS), and the circumstances of the death are suggestive of an underlying channelopathy (young age, exertional death, familial history) post-mortem genetic testing targeted to channelopathy genes should be performed. If this comes back negative, cardiomyopathy genes may be evaluated as well taking into account so-called ‘concealed’ cardiomyopathy.
Along with postmortem genetic testing, it is advised to screen first degree relatives for underlying cardiac disease focusing on channelopathies and cardiomyopathies. This includes at least a detailed personal and family history, resting ECG, exercise ECG and echocardiography.1
The yield of postmortem genetic testing in SADS is around 13% according to the largest case series published using the more stringent ACMG/AMP guidelines for variant interpretation.4 This yield can be increased to about 40% if combined with cardiac screening of first degree family members.
Genetic testing in survivors of SCD
Survivors of cardiac arrest should undergo a thorough clinical screening to identify an underlying cause including ischemic heart disease, coronary anomaly, cardiomyopathy and channelopathy. Genetic testing should be offered whenever indicated in the specific phenotype. If the detailed clinical screening turns out negative, the condition is termed unexplained cardiac arrest (UCA) or idiopathic ventricular fibrillation (IVF) if VF was documented. IVF is present in about 5-7% of all out of hospital cardiac arrests. The yield of genetic testing with whole exome sequencing (WES) and a virtual panel has recently shown to result in a genetic diagnosis in 10% of UCA (CASPER registry).5
Possible genetic findings in UCA are the following:
- - Concealed cardiomyopathy: pathogenic variants in genes linked to arrhythmogenic cardiomyopathy with normal structural findings at TTE and MRI. Genes most commonly involved are either desmosomal (PKP2, DSP) or non-desmosomal (FLNC, RBM20, DES).
- - Channelopathy: pathogenic variants in genes linked to long QT syndrome, Brugada syndrome, catecholaminergic polymorphic ventricular tachycardia and short QT syndrome.
- - Short coupled VF: DDP6 Dutch founder haplotype and gain of function mutations in RYR2 causing calcium release deficiency syndrome (CRDS)
Advantages of a positive genetic result
A positive genetic test result can have impact both on the treatment and prognosis of a survivor of UCA, as on family members of an individual suffering from SCD.
- In case of SADS, a positive genetic test leads to a definite cause of death, which can help family members in the grieving process
- In case of UCA or IVF, a positive genetic test results in diagnosis of a particular disease leading to disease specific treatment and follow-up
- A positive genetic test makes cascade screening of family members possible. Variant carriers should undergo detailed phenotyping leading to personalized treatment and follow up.
- A positive genetic test opens the possibility of pre-implant genetic testing
- A negative genetic test in a family member results in dismissal of follow-up and inability to pass on the genetic defect