Mitral regurgitation
Surgery for heart valve disease follows coronary artery bypass grafting in terms of workload and even surpasses them in countries such as France and Spain. Organic mitral regurgitation (MR) is the leading pathology of the mitral valve in western countries. It is now the second most frequent valve disease after aortic stenosis. Left untreated, strain on the left ventricle (LV) from volume overload can lead to LV dysfunction, pulmonary hypertension, right ventricular dysfunction and ultimately, heart failure whichever valvular dysfunction mechanism is involved.
Surgical treatment is indicated in patients with severe symptomatic MR, left ventricular ejection fraction (LVEF) >30% and end-systolic dimension (ESD) <55 mm. Compared to valve replacement, surgical repair of the mitral valve has consistently lead to superior survival rates and better outcomes. This is basically due to preservation of the subvalvular apparatus which helps to maintain the shape and contractility of the left ventricle (1).
Mortality rates for mitral valve repair are approximately 2% as opposed to 6% for valve replacement (2). Accordingly, patient referral at stages when repair is no longer feasible decreases long term survival. This is particularly true for valve lesions induced by endocarditis (3).
Predicting factors in mitral valve surgery
Predictors of poor outcome in patients with MR include presence of symptoms, increased age, presence of atrial fibrillation, severe grades of MR (effective regurgitant orifice area ≥0.40 cm²), left atrial dilatation, LV dilatation (ESD >45 mm), and low EF ≤60% (4, 5).
Limits related to LV systolic failure
LV dysfunction is a particularly strong predictor of poor outcome and thus surgical management is also recommended in asymptomatic patients with LV dysfunction. In patients with poor LV function (EF<35%), both operative mortality (8%) and 5-year survival (54%) were significantly higher than those observed in patients with a preserved myocardial function 2% and 82%, respectively. Compared to replacement, mitral valve repair has a superior operative outcome and improves NYHA status in patients with low EF, but does not significantly influence long term survival or progression to implantation of LV assist devices or heart transplantation (6).
Ischaemic mitral regurgitation (IMR)
Chronic IMR is a major cause of congestive heart failure after myocardial infarction. There is no surgical consensus regarding the definite determination of operability. Factors such as severity of MR, life expectancy and comorbidities must be factored in. Valve repair seems to benefit patients with the best preoperative function and least comorbidities (7).
Mitral restrictive annuloplasty plus CABG are considered the best therapeutic strategies for chronic IMR even though adding valve surgery doubles operative death in patients with coronary artery disease (8). Age, congestive heart failure, severe LV dysfunction, preoperative intra-aortic balloon pump use and a high number of diseased coronary vessels, increases risk for in-hospital death (9). In these patients and in the absence of significant myocardial viability, the outcome is poor and surgery, whichever procedure is chosen, provides minimal benefit, if any.
Mitral stenosis
Surgical intervention is indicated in clinically significant MS (valve area<1.5 cm²), for symptomatic patients in cases when valve anatomy is unfavourable for percutaneous balloon commissurotomy (PMC) or in the presence of contraindications such as left atrial thrombus. The onset of pulmonary hypertension is associated with a significant increase in surgical mortality, and surgery should be performed as early as possible irrespective of the presence of symptoms. Nevertheless, pulmonary artery pressure usually decreases significantly following valve surgery and seems related to a fall in left atrial pressure and reversal of the severe spastic pulmonary vasoconstriction that accompanies left atrial hypertension in certain patients (10).
Although not an absolute contraindication for surgery, the presence of severe pulmonary hypertension, dilated right heart with right heart failure and tricuspid insufficiency are predictors of poor survival after surgery.
Limits to surgery related to technical factors
Technical considerations are limiting factors in patients requiring mitral valve surgery.
- The risk of damage to the grafts and issues related with cardioplegia especially with arterial grafts in redo surgery with patent coronary artery bypass grafts is also true for mitral as for any valve surgery.
- Severe calcification of the mitral annulus also renders mitral valve repair difficult and valve replacement hazardous from risk of atrioventricular rupture (11).
Minimally invasive surgery and transcatheter techniques
The use of video-assisted minimal access approach after previous cardiac surgery does not obviate the need for cardiopulmonary bypass but has shown lower than predicted mortality and strong patient satisfaction in reoperative mitral valve surgery (12).
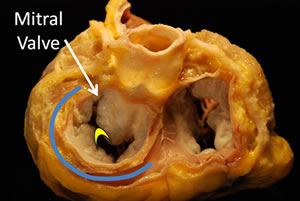
Transcatheter techniques for the mitral valve are in the early stages of development. Attempts to repair the valve by placing a suture between the leaflets to create a double orifice mitral valve by co-apting the central leaflet scallops (figure 1) have been evaluated by the Endovascular Valve Edge-to-Edge Repair Study (EVEREST) with an 85% acute procedural success (13).
Annuloplasty based the principle of correction of leaflet coaptation by decreasing the size of the annulus has been performed with transcatheter techniques. Anatomical studies (14) have evaluated the procedure with devices introduced into the coronary sinus; this has been termed “indirect annuloplasty”. Experimental work has demonstrated the feasibility of transcatheter implantation of the mitral valve within an annuloplasty ring through a transatrial approach (15).
Figure 1. Cross section of the heart at the level of the atrioventricular valves. The blue line shows the site for implantation of coronary sinus devices. The yellow curve shows the site for clip placement - between the 2 leaflets.


 Our mission: To reduce the burden of cardiovascular disease.
Our mission: To reduce the burden of cardiovascular disease.