Background
In the battle against sudden cardiac death (SCD), an implantable cardioverter-defibrillator, with the treatment of coronary artery disease, risk stratification and pharmacological interventions is a therapeutic cornerstone for the management of ischemic and nonischemic cardiomyopathy or impaired ventricular function in the prevention of potentially lethal ventricular tachycardia.
Today's devices are miniaturised pectoral systems capable of defibrillation and pacing with leads inserted into venous circulation through cephalic, axillary or subclavian veins whereas in 1980, surgically-applied pericardial patches required a large abdominally-placed generator (1,2). Great progress in this area has been made.
Several large randomised trials have demonstrated reduction in total mortality in primary and secondary prevention with the implantable cardioverter-defibrillator (3).
However, insertion of the electrode into the central venous circulation and inside cardiac chambers can cause vascular obstruction, thrombosis, infection and cardiac perforation (4,5). Moreover, lead failure has been estimated at 0.58%/year and up to 20% at 10 years; failures may require the leads to be extracted, however this is a highly challenging procedure: major complications rates are about 1% and mortality is at 0.3%, even in experienced centers (6,7,8,9). Some leads have even necessitated recall due to increased mortality.
The development of the subcutaneous defibrillator was initially motivated by cases of children with congenital heart diseases or patients with who were unsuitable for transvenous ICD due to impaired venous access. The result has been the development of a device with no leads within the heart and preservation of the central venous circulation.
1 - Description
The subcutaneous implantable cardioverter defibrillator consists of a 3-mm tripolar parasternal lead (12 French, 45 cm) connected to an electrically active pulse generator. The lead is vertically positioned in the subcutaneous tissue of the chest, parallel to and 1-2 cm to the left sternal midline followed by a horizontal segment, at the level of the 6th rib, until it reaches the left anterior axillary line. The lead has a 8-cm shock coil, flanked by two sensing electrodes - the distal one positioned adjacent to the manubriosternal junction and the proximal one adjacent to the xiphoid process.
Compared to a traditional ICD, the pulse generator is double in size. It measures 78x65x15 mm with a volume of 69 cc and a mass of 145 grams. Its estimated longevity, defined as 3 full-energy capacitor charges per year, is of nearly 5 years (which is half that of the traditional device).
The generator is positioned in the subcutaneous tissue of the chest over the 6th rib between the left midaxillary and the left anterior axillary lines (Fig.1).
Fig. 1: External appearance of an implanted device.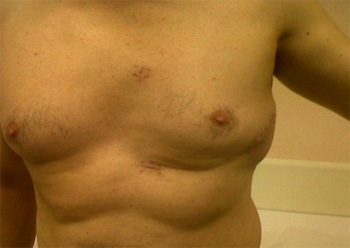
The sensing and detection function identifies subcutaneous signals (“subcutaneous ECG”) and cardiac rhythm from the two sensing electrodes or from electrodes and a pulse generator. There are three available sensing vectors: primary - from proximal electrode ring to can -, secondary - from distal electrode ring to can- and alternate - from distal to proximal electrode. (Fig.2).
Fig. 2: The three available sensing vectors of S-ICD.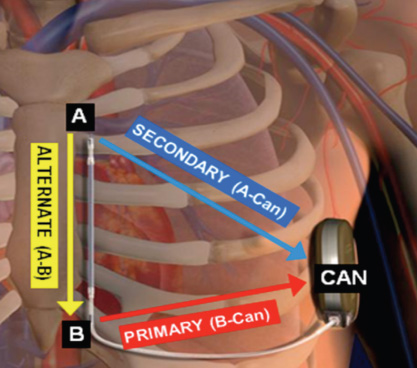
The system automatically selects the optimal vector for detection to prevent inappropriate therapy due to myopotentials, noise or multiple counting (in particular double QRS and T-wave oversensing). A dedicated algorithm or template matching is used for the discrimination of supraventricular arrhythmias. Its system is conceptually similar to that used by transvenous devices, but the S-ICD evaluates a greater number of points (up to 41) of ventricular complex arrhythmias to improve signal resolution.
2 - Performance
In the START study, a recent head-to-head comparison between the S-ICD and the ICD showed that the S-ICD was superior to the transvenous ICD with regard to specificity for supraventricular arrhythmia discrimination (10). In this context, compared to late stage morphology discriminators in transvenous ICD, earlier use of morphology discriminators may represent an advantage.
Moreover, the ultra far field signal recorded by S-ICD lead mimics the surface ECG more closely, theoretically improving diagnostic capability. A shock zone, at 240 beats per minute and a conditional one between 170 and 240 beats per minute can be set to deliver therapy. These zones are to distinguish supraventricular from ventricular tachyarrhythmias.
Testing during implantation is performed with a 65-J shock on induced VF, however after implantation the device delivers only up to five consecutive biphasic 80-J shocks per episode. It does not deliver anti-tachycardia pacing however it can automatically reverse shock polarity if initial shock is unsuccessful.
Charge time is approximately 15” to maximum output, which is longer than transvenous systems.
Demand transthoracic pacing (200-mA biphasic pulse) at a fixed rate of 50 beats per minute is available for 30” after a shock, activated only after >3500 msec of post-shock asystole. Programming is simple as all device settings are automated apart from shock therapy (on/off), pacing after a shock (on/off), conditional discrimination of supraventricular tachyarrhythmias (on/off) and upper-rate cut off for the conditional shock zone (170 to 240 beats per minute). Implanting the system is relatively simple and guided only by anatomical landmarks and with no fluoroscopy required; however a learning curve, consisting of at least 3-4 cases, is necessary for an operator to become familiar with the procedure (11,12).
3 - Approval
A number of pivotal studies initiated more than 15 years ago demonstrated the ability of the subcutaneous system to terminate ventricular arrhythmias using different shock vectors and with variable energy requirements. The first human study provided information regarding optimal lead configuration, lead and generator placement and shock efficacy on both induced and spontaneous arrhythmias (13). After obtaining CE marking in 2009, several single-center and multicenter experiences across Europe demonstrated safety and efficacy of the S-ICD in a “real world” scenario (14,15,16,17). Finally the first prospective, single-arm, non-randomised, multicenter trial was completed in the U.S. It showed that both effectiveness and safety endpoints were met compared with traditional devices; ventricular fibrillation (VF) was detected without delay in >99% of cases and all spontaneous VT/VF episodes were terminated by the device with a first shock efficacy >92% which is comparable to the efficacy of transvenous ICD. These results led to the US Food and Drug Administration approval in late 2012.
4 - Advantages
The advantages of the S-ICD are:
- The absence of leads within the heart and the preservation of central venous circulation is the main advantage making it a great alternative for children with congenital heart diseases or patients with no venous access who were unsuitable for transvenous ICD.
- There is no risk of vascular injury or pneumothorax.
- The risk of systemic infections appears to be very low, best in high risk cases of previous device infection, hemodialysis, chronic immunosuppression therapy immunodeficiencies, or artificial heart valves.
- Although pocket infections can occur with the S-ICD (5-10%, similar to transvenous devices) infection resolves with antibiotics in the majority of cases (18).
- Explantation is rarely necessary however explanting an S-ICD is much simpler and safer than endovascular lead extraction. Young patients with high lead failure rates due to an active lifestyle may especially benefit (19).
- The simplified implant procedure, with no need for fluoroscopy favors a more widespread use of a life-saving therapy, even in those centers without great experience in traditional surgical procedure, with less risk of serious complications.
- The S-ICD has cosmetic advantages despite its larger size, owing to the anatomic location in the lateral axilla: this location, rather than the anterior infraclavicular position is particularly appreciated by women.
- The device is well tolerated, no explantation due to patient discomfort has been noted.
- Another benefit is the lack of apparent myocardial damage despite greater shock intensity (80 J). Energy is actually distributed more evenly throughout the myocardium, with only 10% of delivered energy reaching cardiac myocytes: elevation of serum bio-markers of myocardial damage is lessened (20), and this may be particularly important in patients with ventricular dysfunction.
5 - Limitations
Described further are the limitations of the device:
- Pre-implantation screening is mandatory. Using a customised transparent plastic tool provided by the manufacturer, a modified “ad hoc” ECG skin electrode positioning is carried out to verify adequate sensing of subcutaneous signals, avoiding double counting of QRS or T wave oversensing (TWOS) in particular (Fig.3). Up to 7.4% of possible candidates would not be suitable for implant: hypertrophic cardiomyopathy, heavy weight, prolonged QRS duration and a R/T ratio <3 were independently associated with screening failure (21).
- Rates of inappropriate shocks are similar to those occuring with the transvenous ICD and range from 4 to 25% however the mechanisms are different. WIth the S-ICD, eighty per cent of inappropriate shocks are caused by TWOS, especially in congenital heart disease, Brugada and long QT syndromes, while in transvenous ICD, TWOS is involved in about 20% of cases. Accurate screening is possible even with acquisition of templates during exercise. Moreover about 5 to 10% of inappropriate shocks can be caused by muscular noise due to myopotentials (lead is placed over intercostal and pectoral muscles) but this problem may be overcome by reprogramming the sensing vector. Explantation can become necessary (5% in one study) when repeated shocks due to TWOS or noise occur despite different vectors programming.
- The risk for undersensing of true arrhythmias (i.e. VF with very low amplitude waves)
- Prolonged time to therapy compared with transvenous ICD (14-18” vs 7-8”)
- Inadequate shock efficacy on spontaneous clinical VT/VF (22). Indeed, there are still relatively few data regarding long term performance of the S-ICD in a “real world” scenario therefore larger experience and longer follow-up are required, nevertheless initial results are encouraging (no death can be attributed to device or lead failure in published studies).
- Other limitations are no pacing capability (with the exception of post-shock pacing) that also means no ATP nor resynchronisation therapy delivery (CRT).
- Remote monitoring and atrial tachyarrhythmias monitoring are not yet available.
- Pulse generator is larger, with anticipated life battery shorter than traditional devices (about 5 years).
- Costs are still high: The S-ICD in Italy costs 18.000 € compared to 4-7000 € for a single chamber transvenous ICD.
Fig. 3: Two examples of oversensing during premature ventricular contractions. Top: double QRS counting. Bottom: T wave oversensing. S indicates a sensed event.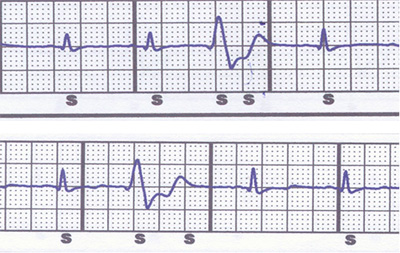
6. Patient selection
In as much as there are patients needing an ICD but in whom a transvenous ICD is not feasible, the S-ICD may be the only option.
- The pediatric or GUCH (Grown-Up Congenital Heart disease) population with no venous access due to congenital anomalies is concerned.
- Children with preserved venous access factors favoring S-ICD owing to the effect of growth on endocardial leads and the possibility of saving venous vasculature
- Acquired stenosis or obstruction of central veins that may make the placement of a lead difficult or even impossible; such venous anomalies can be found in 7% of cases during pre-implant contrast venography (23).
- A history of previous endocarditis or device infections: the risk of relapse is very high in these cases, with potential catastrophic consequences.
- Other situations with a very high risk of infection of endovascular leads are: dialysis, immunodeficiencies, cancer with the need of a chronic indwelling catheter for drug infusion.
- An S-ICD should be considered in patients anticipating cardiac transplantation by avoiding endovascular fibrosis that may render surgical lead extraction very difficult at the time of transplantation.
- An S-ICD appears suited to young patients with an active lifestyle and a long life expectancy. This is particularly true for inherited genetic arrhythmogenic syndromes (Brugada, Long and Short QT, Early Repolarisation) where clinical arrhythmias are polymorphic VT or VF (not treatable with ATP) and the risk of bradycardia and monomorphic VT is very low.
- In Brugada patients the incidence of endocardial lead dysfunction was reported to be as high as 2% per year versus 0.58%/year of the general ICD population and the overall risk of device-related complications can be as high as 8-9%/year, so they would benefit from a therapeutic option not requiring endocardial lead implantation (24). However the issues of S-ICD therapy efficacy and detection accuracy are particularly relevant in Brugada Syndrome: the unique QRS to T wave ratio and morphology, carries the risk of double QRS/T wave counting and inappropriate therapy. Moreover low amplitude wave VF may result in tachyarrhythmias undersensing, with delayed detection and intervention. Until now very few patients with Brugada syndrome were included in clinical trials and published series, but initial reports are encouraging with evidence of correct detection and shock efficacy even when low amplitude waves VF occurred (Fig. 4).
- The S-ICD would also represent a valuable option in hypertrophic cardiomyopathy, as traditional transvenous ICD is associated with a high complication rate.
- Other potential indications for this new device are: patients with prosthetic heart valves (infection risk) and women – for cosmetic reasons especially.
- In terms of primary prevention in patients with ischemic/non ischemic dilated cardiomyopathy, the S-ICD can be a reasonable choice for two main reasons: 1) pacing indication for bradycardia is relatively rare when ICD is implanted (less than 10% of patients); 2) the likelihood of developing first time monomorphic VT is not so high (only 15-20% of patients experience high rate monomorphic VT during the first year after the implant) and subsequent risk is 1.8%/year. However a cause for caution exists in patients with left ventricular dysfunction following myocardial infarction; a long term study (2 years follow up) recording cardiac rhythm with implantable loop monitor (25) showed a non negligible incidence of bradycardic events in patients without previous pacing indication: 10% high-degree AV block, 7% sinus bradycardia <30 bpm and 5% sinus arrest.
- Finally, regarding secondary prevention patients a S-ICD can be considered if they are survivors of an out-of-hospital VF episode (with no evidence of monomorphic VT as index arrhythmia).
Fig. 4: VF episode in Brugada syndrome treated by the subcutaneous defibrillator. S-ICD electrogram of spontaneous ventricular polymorphic tachycardia degenerating into ventricular fibrillation and treated with a shock in a patient with Brugada Syndrome. C= capacitor charging; S= sensing of an event not classified as tachycardia; T= sensing of an event classified as tachycardia. A dot indicates sensing of an unclassifiable event that is discarded. Black lightning symbol indicates a shock. Red arrows indicate post shock transthoracic pacing (reproduced with permission from reference 24).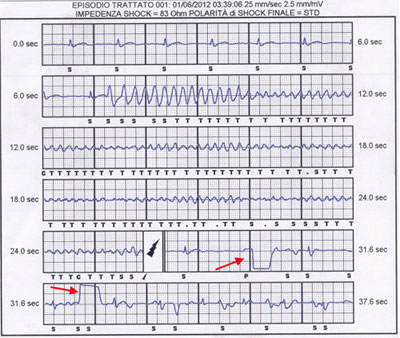
Contraindications
Contraindications of the ICD are the following:
- The S-ICD should not be implanted when the pre-implant screening test has failed (up to 7% of cases).
- Also, S-ICD is contraindicated in patients with: symptomatic bradycardia requiring permanent pacing; previously implanted unipolar pacemaker (because of sensing/detection pitfalls); systolic heart failure and left bundle branch block indicated for CRT; recurrent sustained monomorphic VT treatable with ATP.
- Moreover some anatomic characteristics may increase the risk of complications following the implant: for example very thin patients with poor subcutaneous tissue (risk of skin erosion over the generator) or patients with abnormalities of chest wall such as “pectus excavatum” (risk of skin erosion along the lead).
A summary of proposed recommendations for patient selection is as follows:
S-ICD as a first choice:
- Pediatric or GUCH patients with no venous access
- Acquired stenosis or obstruction of central veins
- Previous endocarditis or device infection
- Patients at very high risk of infection of endovascular leads: dialysis, immunodeficiencies, cancer, need of a chronic indwelling catheter
- Patients candidates to cardiac transplantation.
S-ICD as a reasonable choice:
- Young patients with an active lifestyle and a long life expectancy
- Inherited genetic arrhythmogenic syndromes (Brugada, Long and Short QT, Early Repolarisation)
- Hypertrophic cardiomyopathy
- Prosthetic heart valves (infection risk). Women (“cosmetic” issue).
- Primary prevention patients with ischemic/non ischemic dilated cardiomyopathy
- Secondary prevention patients survivors of out-of-hospital VF
When to avoid the S-ICD:
- Failed pre-implant screening (up to 7% of cases)
- Symptomatic bradycardia requiring permanent pacing
- Previously implanted unipolar pacemaker (sensing/detection pitfalls)
- Systolic heart failure and left bundle branch block indicated for CRT
- Recurrent sustained monomorphic VT treatable with ATP
- Anatomic characteristics: thin patients with poor subcutaneous tissue, “pectus excavatum”.
8 - Future perspectives
Approximately 55% of patients in routine clinical practice needing an ICD are potential candidates for a subcutaneous device despite current limitations. Nevertheless, to maximise clinical outcome and cost/benefit ratio, it is fundamental to choose candidates that can benefit the most.
Next generation devices might address many of the current limitations by offering improvement of battery technology, a downsized generator; improved algorithm design, remote monitoring and pacing capability with perhaps leadless ultrasound-based cardiac stimulation.
Currently there are still no clinical data that demonstrate S-ICD can prevent sudden death or improve survival in the same way as traditional ICD. The possible use of S-ICD as a first-line strategy or as an alternative approach for specific populations will require the confirmation of large clinical trials and registries that are currently ongoing (26,27).


 Our mission: To reduce the burden of cardiovascular disease.
Our mission: To reduce the burden of cardiovascular disease.