Background
Arrhythmogenic right ventricular dysplasia (ARVD) is a genetically determined myocardial disease characterised by fibrous fatty replacement and ventricular arrhythmias, involving the right ventricle predominantly, starting at the epicardium and extending transmurally.(1) It usually shows an autosomal dominant pattern i.e possession of this non-sex gene places one at risk for having this disease and offspring of an affected parent will have a 50% chance of inheriting this disease. It has, however incomplete penetrance – i.e it can express itself or not, and furthermore, ARVD has variable expressivity, with genes expressing themselves to varying degrees. (2) Causative mutations in gene encoding desmosomal proteins (plakoglobin, desmoplakin)- i.e proteins that bind cells to one another, are found in approximately half of all ARVD patients. (2, 3) Left ventricular involvement becomes present in a majority of cases and a left dominant form has also been described. (4, 5)
Clinical presentations in patients with ARVD vary widely. Heart failure, ventricular arrhythmias and sudden cardiac death (SCD) are the most severe clinical manifestations of ARVD.
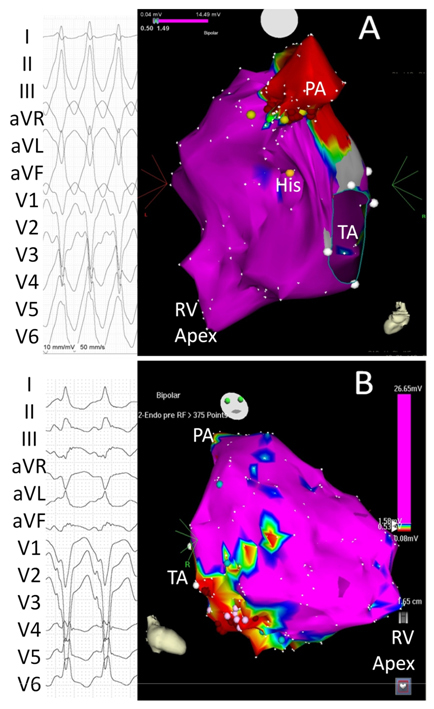
Early diagnosis appears crucial in preventing SCD. Furthermore, mortality in patients with diagnosed ARVD and an implantable cardioverter defibrillator (ICD) is relatively low. (6) Diagnosing ARVD is nevertheless difficult and is currently based on the presence of major and minor Task Force criteria, which include electrocardiographic, structural (cardiac imaging and histology), clinical and familiar factors. (7) Beyond a personal and family medical history, routine assessment includes electrocardiography, echocardiography, 24-hour Holter monitoring, stress testing, and cardiac magnetic resonance. In uncertain diagnoses, endocardial voltage mapping may help detect the presence of myocardial scar in the disease's early phases (figure 1).
Figure 1: Differential diagnosis between idiopathic right ventricular (RV) outflow tract tachycardia and ARVD/C.
Panel A: 12-lead electrocardiogram (left) in a patient with no structural heart disease and sustained VT and endocardial voltage mapping (right). Entire RV endocardium shows normal voltage (purple indicates>1.5 mV). Red dots located below the origin of the pulmonary artery (PA) indicate radiofrequency applications. Panel B: Endocardial voltage mapping of a patient diagnosed with ARVD/C showing a subtricuspid lateral low voltage area (red indicates <0.5 mV). The QRS morphology of the VT suggests that the exit of the reentry circuit is located on the RV lateral wall. TA, tricuspid annulus
Therapeutic armamentarium for treating ARVD includes antiarrhythmic drugs, the placement of an ICD, radiofrequency ablation and cardiac transplantation. The most important decision in the management of a patient with ARVD is whether to recommend an ICD for treatment of VA and prevention of SCD. Treating patients with an ICD in repeated VA is often difficult; Moreover, antiarrhythmic drugs are frequently ineffective. Although radiofrequency ablation has been relegated as a last therapeutic option, the development of epicardial mapping techniques and substrate-guided ablation have offered promising results.
1) Recommendations according to risk
Although data on risk stratification and management of ARVD is lacking, broad agreement exists that ICD implantation is recommended in patients with documented sustained VA due to high incidence of arrhythmia recurrences. (8, 9) Implantable cardioverter-defibrillator therapy occurs in approximately 10% of ARVD cases per year, and estimated survival benefit is around 25-30% at 3-years follow-up. (9)
On the other hand, there is no clear consensus regarding primary prevention of SCD in ARVD patients without documented sustained VA. Several risk markers have been identified, from retrospective studies mostly, as ones of adverse prognosis: syncope, proband status, young age, sporting activity, familial history of SCD, QRS and QT dispersion, severe right ventricular dysfunction, left ventricular involvement or inducible VT. (9-11) ICD implantation is also reasonable in patients with a history of undiagnosed syncope. (8)
A cohort of patients with ARVD from a recent multicenter observational study received an ICD for primary prevention based on 1 or more arrhythmic risk factors: syncope, nonsustained ventricular tachycardia, familial SCD, and VA inducibility. Study showed that syncope was an important predictor that ICD therapy will be life-saving. (12) In another recent study involving ICD recipients without prior sustained VA, proband status, presence of nonsustained ventricular tachycardia and inducibility at electrophysiologic study were identified as predictors that ICD therapy is appropriate. (13)
The predictive value of programmed ventricular stimulation has shown conflicting results in various studies in the literature and an uncertain role in risk stratification. (1, 12-14) Patients with extensive disease, including left ventricular involvement should be considered for primary prevention of SCD. (8)
Sporting activity has been related to increased risk of SCD in patients with ARVD. Furthermore, the overload resulting from training can accelerate the progression of the disease. (15) Indeed, since ARVD is a disease of desmosomal dysfunction, circumstances that increase myocardial strain worsen the mechanical coupling between cardiac myocytes and promote myocardial replacement fibrosis. Indeed, when placed under mechanical stress (exercise) the defective desmosomes, detach from each other, leading to cell death. This cell death causes inflammation with scar formation and fat deposition. Therefore, patients with ARVD - asymptomatic mutation carriers included, should avoid strenuous physical exercise. Competitive sports are altogether contraindicated and recommendations on recreational sport are very restrictive. (16)
Recommendations for risk stratification and clinical management are summarised in Table 1.
Table 1. Proposed recommendations for clinical management and prevention of sudden cardiac death in patients with ARVD
| Subgroups | Risk markers | Recommendations | Follow-up | ICD indication |
|---|---|---|---|---|
| Definite ARVD High risk |
Aborted SCD Sustained VT Unexplained syncope |
Reduce physical exercise Avoid competitive sport β-blockers |
Annually including: Electrocardiography Cardiac imaging (echocardiography vs. CMR) Holter Exercise stress testing |
Recommended |
| Definite ARVD Moderate risk |
Extensive disease (severe RV dysfunction, large LV involvement) Nonsustained VT |
Reduce physical exercise Avoid competitive sport β-blockers |
Annually including: Electrocardiography Cardiac imaging (echocardiography vs. CMR) Holter Exercise stress testing |
Consider |
| Definite ARVD Low risk |
Remaining patients with definite diagnosis of ARVD | Reduce physical exercise Avoid competitive sport β-blockers |
Annually including: Electrocardiography Cardiac imaging (echocardiography vs. CMR) Holter Exercise stress testing |
Not recommended |
| Asymptomatic mutation carriers | Asymptomatic mutation-carrying relatives of ARVD | Reduce physical exercise Avoid competitive sport |
Annually including: Electrocardiography Cardiac imaging (echocardiography vs. CMR) Holter Exercise stress testing |
Not recommended |
ARVD: Arrhythmogenic right ventricular dysplasia; CMR: Cardiac magnetic resonance; LF: left ventricle; RV: right ventricle; SCD: Sudden cardiac death; VT: ventricular tachycardia
2) Pharmacological treatment
No clinical trial has studied the efficacy of antiarrhythmic drugs in the treatment of ventricular arrhythmias in patients with ARVD. Other data available however, suggest that β-blockers, sotalol and amiodarone are effective to this end.(17)
Antiarrhythmic drugs are useful in ICD recipients with sustained ventricular tachycardia and patients with nonsustained or frequent premature ventricular complexes. The impact of medical therapy on mortality however is not established and in patients with documented sustained VA, medical therapy can only be considered a substitute for an ICD in cases where device implantation is not feasible. As demonstrated in other clinical settings, the role of antiarrhythmic drugs in preventing SCD is probably very limited. Its use should be directed to the prevention of ventricular arrhythmias and their appropriate ICD therapies.(18)
Current practice in selection of antiarrhythmic therapy is mostly drawn from a study that compared the efficacy of different drugs based on suppression of ventricular tachycardia in programmed ventricular stimulation.(17, 19) In a recent prospective observational study involving ARVD patients with ICD, in contrast to amiodarone neither β-blockers nor sotalol seemed to be protective against VA. (20) The doses of sotalol administered however (mean 240 mg/day) in this study were lower than those described in studies with positive results (320-480 mg/day). Moreover, side effects of prolonged therapy with amiodarone should be carefully considered, especially in young patients.
3) Catheter ablation
Radiofrequency catheter ablation is an option for the treatment of VA in patients with ARVD. This therapeutic option has often been restricted to patients with refractory ventricular tachycardia (11), as a last resort therapy due to limited experience, variable acute and chronic success rates and fear of complications, especially cardiac perforation. However, with the development of new ablation techniques and recent results, we think that catheter ablation can be offered to patients unresponsive or intolerant to antiarrhythmic drugs, and also to avoid the potential occurrence of drug side effects.
Catheter ablation can be directed to the clinical or induced ventricular tachycardia. Mapping and ablation techniques are comparable to those used in patients with remote myocardial infarction. (21) The presence of multiple circuits, hemodynamic instability or non-inducibility nevertheless limits this ablation approach. More recently, following the emergence of electroanatomical navigation systems, techniques for the characterisation of the arrhythmia substrate have been developed. Substrate-based mapping and ablation aim to identify and abolish areas that are likely to support reentrant circuits during sinus rhythm. (22) Substrate guided ablation eliminates multiple potential circuits that could result in ventricular tachycardia, regardless of the fact that the arrhythmia may not have been clinically documented or induced during electrophysiological study. Complex arrhythmia substrate of ARVD can justify the better results observed in substrate-guided ablation. (23, 24)
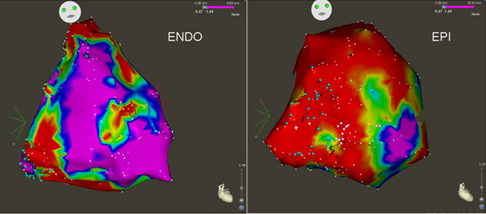
Epicardial mapping and ablation have been used in patients with ARVD. (25) Epicardial scar is wider than the endocardial scar in ARVD patients with sustained VA, and the majority of potential isthmuses for ventricular tachycardia are identified on the epicardial surface (Figure 2).
Figure 2: Endocardial (ENDO) and epicardial (EPI) voltage maps of a patient submitted for ventricular tachycardia ablation and definite diagnosis of ARVD. Patchy non-confluent scars (< 0.5 mV, in red) are identified at subtricuspid, free wall and outflow levels in the endocardial mapping. An extensive low-voltage area is present in the epicardial surface. The epicardial scar is wider than the endocardial, with higher density of isolated potentials (blue dots). Normal myocardium (>1.5mV) is coded in purple. Red dots: radiofrequency applications.(26)
In our experience, a first-line combined endocardial and epicardial substrate guided catheter ablation resulted in a very good short- and mid-term success rate. The high recurrence rate published in earlier series has been attributed to the progressive nature of the disease. However, the known epicardial nature of ARVD should play a key role in the results of conventional endocardial approach.
Despite the extensive and complex arrhythmogenic substrate and concerns about cardiac perforation, no study using radiofrequency has reported severe complications from catheter ablation in ARVD patients. The procedure appears to be safe, - in high volume centers at least.



 Our mission: To reduce the burden of cardiovascular disease.
Our mission: To reduce the burden of cardiovascular disease.